Abstract
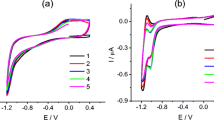
[Tetraphenylporphyrin]Co(II) (CoTPP), has been thought to be impossible to polymerize, is deposited on glassy carbon electrode (GCE) by anodic oxidation. The poly{[tetraphenylporphyrin]Co(II)} (pCoTPP) films are obtained through aryl–aryl couplings. Compared with monomeric CoTPP, the polymers provide higher density of active sites for oxygen-reduction reaction (ORR) in 0.5 M H2SO4. A synergistic effect between cobalt and porphyrin rings is observed. Results of the rotating disk electrode measurement indicate that ORR in the pCoTPP film mainly occurs through a four-electron pathway to form H2O. The pCoTPP-modified GCE exhibits good stability in an acidic medium.









Similar content being viewed by others
References
Jasinski R (1964) A new fuel cell cathode catalyst. Nature 201:1212–1213
Vengatesan S, Cho E, Oh IH (2012) Development of non-precious oxygen reduction reaction catalyst for polymer electrolyte membrane fuel cells based on substituted cobalt porphyrins. Korean J Chem Eng 29:621–626
Zhang H, Ren QZ, He SY (2011) High-temperature pyrolysis of ketjen black EC 300J supported cobalt porphyrin complexes as electrocatalysis for oxygen reduction. Chem J Chin Uni 32:344–349
Savastenko NA, Brüser V (2011) Plasma modification of self-assembled structures of CoTMPP molecules. Appl Surf Sci 257:3480–3488
De Paz HH, Médard C, Morin M (2010) An electrochemical and spectroelectrochemical study of pentacoordinated CoTPP on glassy carbon electrode. J Electroanal Chem 648:163–168
Shawn S, Allison E (2013) Enhanced O2 electrocatalysis by a highly conjugated cobalt(II) porphyrin. Inorganic Chem Commun 29:14–17
Matson BD, Carver CT, Ruden AV, Yang JY, Raugei S, Mayer JM (2012) Dostamt protonated pyridine groups in water-soluble iron porphyrin electrocatalysts promote selective oxygen reduction to water. Chem Commun 48:11100–11102
He QG, Mugadza T, Kang XW, Zhu XB, Chen SW, Kerr J, Nyokong T (2012) Molecular catalysis of the oxygen reduction reaction by iron porphyrin catalysts tethered into Nafion layers: an electrochemical study in solution and a membrane-electrode-assembly study in fuel cells. J Power Sources 216:67–75
Zhou Q, Ming C, Li J, Lu J (2008) Electrocatalysis of template-electrosynthesized cobalt-porphyrin/polyaniline nanocomposite for oxygen reduction. J Phys Chem C 112:18578–18583
Umasankar Y, Shie JW, Chen SM (2009) Electrocatalytic activity of oxygen and hydrogen peroxide reduction at poly(iron tetra(o-aminophenyl) porphyrin) coated multiwalled carbon nanotube composite film. J Electrochem Soc 156:K238–K244
Jafarian M, Haghighatbin MA, Gobal F, Mahjani MG, Rayati S (2011) A comparative investigation of the electrocatalytic oxidation of methanol on poly-NiTCPP and poly-TCPP/Ni modified glassy carbon electrodes. J Electroanal Chem 663:14–23
Wang J, Golden T (1989) Metalloporphyrin chemically modified glassy carbon electrodes as catalytic voltammetric sensors. Anal Chim Acta 217:343–351
Widelor A, Larsson R (1992) ESCA and electrochemical studies on pyrolysed iron and cobalt tetraphenylporphyrins. Electrochim Acta 37:187–197
George RC, Mugadza T, Khene S, Egharevba GO, Nyokong T (2011) Porphyrin nanorods modified glassy carbon electrode for the electrocatalysis of dioxygen, methanol and hydrazine. Electroanal 23:1699–1708
Deronzier A, Moutet JC (1996) Polypyrrole films containing metal complexes: syntheses and applications. Coord Chem Rev 147:339–371
Chen SM, Chen YL, Thangamuthu R (2007) Electropolymerization of iron tetra(o-aminophenyl)porphyrin from aqueous solution and the electrocatalytic behavior of modified electrode. J Solid State Electrochem 101:441–1448
Lin CY, Hung YC, Liu CM, Lo CF, Lin YC, Lin CL (2005) Synthesis, electrochemistry, absorption and electro-polymerization of aniline-ethynyl metalloporphyrins. Dalton Trans 2:396–401
Chen W, Akhigbe J, Brückner C, Li CM, Lei Y (2010) Electrocatalytic four-electron reduction of dioxygen by electrochemically deposited poly{[meso-tetrakis(2-thienyl)porphyrinato]cobalt(ii)}. J Phys Chem C 114:8633–8638
Kuester SN, McGuire MM, Drew SM (1998) Electrochemically initiated polymerization of zinc(II)5-vinyl- 10,15,20-triphenyl- porphyrin. J Elecctroanal Chem 452:13–18
Chang CJ, Deng YQ, Shi CN, Chang CK, Anson FC, Nocera DG (2000) Electrocatalytic four-electron reduction of oxygen to water by a highly flexible cofacial cobalt bisporphyrin. Chem Commun 2000:1355–1356
Liu RL, Malotki CV, Arnold L, Koshino N, Higashimura H, Baumgarten M, Müllen K (2011) Triangular trinuclear metal-N4 complexes with high electrocatalytic activity for oxygen reduction. J Am Chem Soc 133:10372–10375
Holmes-Smith AS, Hamill A, Campbell M, Uttamlal M (1999) Electropolymerised platinum porphyrin polymers for dissolved oxygen sensing. Analyst 124:1463–1466
Christine PR, Joёlle RB, Gérard S, Cyril P, Mohammed A, Julien L (2006) Electroactive films of poly (tetraphenylporphyrins) with reduced bandgap. J Elecctroanal Chem 597:19–27
Christine PR, Joёlle RB, Gérard S, Letessier J, Bergamini JF (2007) Selective anodic preparation of 1D or 2D electroactive deposits from 5,15-bis-(9H-fluoren-2-yl)-10,20-diphenyl porphyrins. J Elecctroanal Chem 606:103–116
Adler AD, Longo ER, Shergalis W (1964) Mechanistic investigations of porphyrin syntheses. I. preliminary studies on mes-tetraphenylporphin. J Am Chem Soc 86:3145–3149
Li D, Shi YY, Fa HB, Zheng WQ, Shan N, Yang M, Wang DJ, Cheng XL, Wang XQ (2006) Synthesis and properties of dimeric porphyrin based on the hydroxyphenylporphyrin scaffold. J Porphyrins Phthalocyanines 10:1392–1397
Maruyama H, Segawa H, Sotoda S, Sato T, Kosai N, Sagisaka S, Shimidzu T, Tanaka K (1998) Electrochemical construction of ultrathin film composed of quasi two-dimensional porphyrin polymers. Synth Met 96:141–149
Shimidzu T, Segawa H, Wu F, Nakayama N (1995) Approaches to conducting polymer devices with nanostructures: photoelectrochemical function of one-dimensional and two-dimensional porphyrin polymers with oligothienyl molecular wire. J Photochem Photobiol A 92:121–127
Song C, Zhang J (2008) Electrocatalytic oxygen reduction reaction. In: Zhang JJ (ed) PEM Fuel Cell Electrocatalysts and Catalyst Layers. Springer, New York, pp 89–134
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31101284), the Natural Science Foundation of Chongqing (CSTC, 2010BB1209, 2009BB6213), and the Fundamental Research Funds for the Central Universities (no. CQDXWL-2012-034, CQDXWL-2012-035).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yin, W., Chen, C., Fa, H. et al. Electropolymerization of CoTPP and its catalytic performance for oxygen-reduction reaction in an acid medium. J Solid State Electrochem 17, 3095–3099 (2013). https://doi.org/10.1007/s10008-013-2208-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-013-2208-y