Abstract
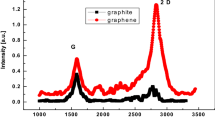
Graphene nanosheets are prepared from H2 thermal reduction of graphite oxide at 300 °C. The graphite oxide interlayer has readily been expanded through chemical oxidation of meso-carbon micro-beads graphite raw material. After H2 reduction, the carbon/oxygen ratio of graphene is increased from that of graphite oxide due to the removal of oxygen-containing functional groups as it is demonstrated from IR spectra. The d-spacing of resulting graphene nanosheets is increased to 0.37 nm, which facilitates lithium intercalation. Such synthesized graphene nanosheet material as anode of lithium-ion battery has exhibited high reversible discharge capacity of 1,540 mAh g−1 at a current density of 50 mA g−1, and the coulumbic efficiency was 97% over 50 cycles. The discharge curve of the anode material shows a continuously increased voltage profile, which is a characteristic of a capacitive material.




Similar content being viewed by others
References
Kasavajjula U, Wang C, Appleby AJ (2007) J Power Sources 163:1003–1039
Chan CK, Peng H, Liu G, McIlwrath K, Zhang XF, Huggins RA, Cui Y (2008) Nat Nano 3:31–35
Ng S-H, Wang J, Wexler D, Konstantinov K, Guo Z-P, Liu H-K (2006) Angew Chem Int Ed 45:6896–6899
Novoselov KS, Geim AK, Morozov SV, Jiang D, Zhang Y, Dubonos SV, Grigorieva IV, Firsov AA (2004) Science 306:666–669
Novoselov KS, Geim AK, Morozov SV, Jiang D, Katsnelson MI, Grigorieva IV, Dubonos SV, Firsov AA (2005) Nature 438:197–200
Hernandez Y, Nicolosi V, Lotya M, Blighe FM, Sun Z, De S, McGovern IT, Holland B, Byrne M, Gun'Ko YK, Boland JJ, Niraj P, Duesberg G, Krishnamurthy S, Goodhue R, Hutchison J, Scardaci V, Ferrari AC, Coleman JN (2008) Nat Nano 3:563–568
Yoo E, Kim J, Hosono E, Zhou H-s, Kudo T, Honma I (2008) Nano Lett 8:2277–2282
Paek S-M, Yoo E, Honma I (2008) Nano Lett 9:72–75
Wang G, Shen X, Yao J, Park J (2009) Carbon 47:2049–2053
Novoselov KS, Jiang Z, Zhang Y, Morozov SV, Stormer HL, Zeitler U, Maan JC, Boebinger GS, Kim P, Geim AK (2007) Science 315:1379
Stoller MD, Park S, Zhu Y, An J, Ruoff RS (2008) Nano Lett 8:3498–3502
Geng Y, Wang SJ, Kim J-K (2009) J Colloid Interface Sci 336:592–598
Stankovich S, Piner RD, Nguyen ST, Ruoff RS (2006) Carbon 44:3342–3347
Brodie BC (1860) Ann Chim Phys 59:466–472
Hummers WS, Offeman RE (1958) J Am Chem Soc 80:1339
Staudenmaier L (1898) Ber Dtsch Chem Ges 31:1481–1487
Stankovich S, Dikin DA, Piner RD, Kohlhaas KA, Kleinhammes A, Jia Y, Wu Y, Nguyen ST, Ruoff RS (2007) Carbon 45:1558–1565
Pan D, Wang S, Zhao B, Wu M, Zhang H, Wang Y, Jiao Z (2009) Chem Mater 21:3136–3142
Sun H, He HM, Ren JG, Li JJ, Jiang CY, Wan CR (2007) Electrochim Acta 52:4312–4316
Nagao M, Pitteloud C, Kamiyama T, Otomo T, Itoh K, Fukunaga T, Tatsumi K, Kanno R (2006) J Electrochem Soc 153(5):A914–A919
Acknowledgments
Support of this work by Natural Science Foundation of China (No.20663005) is greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, T., Gao, L. A high-capacity graphene nanosheet material with capacitive characteristics for the anode of lithium-ion batteries. J Solid State Electrochem 16, 557–561 (2012). https://doi.org/10.1007/s10008-011-1384-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-011-1384-x