Abstract
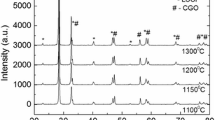
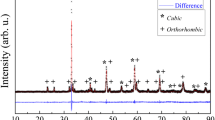
SrCo0.8Fe0.2O3-δ (SCF), as a promising cathode material for intermediate temperature solid oxide fuel cells, possesses a high catalytic activity for the reduction of O2 to 2O2−. The SCF powder was successfully synthesized by the solid state reaction method and Pechini method and characterized using XRD, particle analysis, and electrochemical performance measurements. Smaller-particle-size SCF materials (SCF-P) with single phase are obtained at lower synthesis temperature by the Pechini method and possess better electrochemical performance as compared with those prepared by the solid state reaction method. The reason is that the Pechini method involves the mixing of elements at atomic level, so pure SCF phase formation can be accelerated and showed high electrocatalytic activity. The preparation procedure of SCF cathode was firstly investigated using electrochemical impedance spectroscopy. Results show that the total polarization resistance and the low-frequency resistance decrease gradually with the reduction of the calcination temperature for the SCF cathodes. The SCF-P cathode sintered at 1,000 °C possesses the highest porosity and the best electrochemical performance. It is the result of a comprehensive function of three-phase boundary length, porosity of cathode, and the adhesion between cathode and electrolyte. The charge-transfer process, together with the adsorption, dissociation, and diffusion of oxygen, has a strong influence on the whole reaction process of the cathode. The influence of binder amounts on the performance of the SCF-P cathodes was also studied.







Similar content being viewed by others
References
Murray EP, Tsai T, Barnett SA (1999) Nature 400:649
Flytzani-Stephanopoulos M, Sakbodin M, Wang Z (2006) Science 312:1508
Park EW, Moon H, Park MS, Hyun SH (2009) Int J Hydrocarb Eng 34:5537
Steele BCH (2001) Nature 414:345
Zhu XD, Sun KN, Le SR, Zhang NQ, Fu Q, Chen XB, Yuan YX (2008) Electrochim Acta 54:862
Hibino T, Hashimoto A, Inoue T, Tokuno JI, Yoshida SI, Sano M (2000) Science 288:2033
Huang B, Wang SR, Liu RZ, Ye XF, Nie HW, Sun XF, Wen TL (2007) J Power Sources 167:39
Ishihara T, Matsuda H, Takita Y (1994) J Am Chem Soc 116:3801
Feng M, Goodenough JB (1994) Eur J Solid State Inorg Chem 31:663
Takeda Y, Sakaki Y, Ichikawa T, Imanishi N, Yamamoto O, Mori M, Mori N, Abe T (1994) Solid State Ionics 72:257
Jiang SP, Love JG (2003) Solid State Ionics 158:45
Huang K, Goodenough JB (2001) J Electrochem Soc 148:E203
Huang K, Wan J, Goodenough JB (2001) J Mater Sci 36:1093
Huang KQ, Wan JH, Goodenough JB (2001) J Electrochem Soc 148:A788
Adler SB (1998) Solid State Ionics 111:125
Adler SB, Reimer JA (1996) Solid State Ionics 91:175
Zhu XD, Zhang NQ, Wu LJ, Sun KN, Yuan YX (2010) J Power Sources 195:7583
Gosavi PV, Biniwale RB (2010) Mater Chem Phys 119:324
Sasaki K, Wurth JP, Gschwend R, Gödickemeier M, Gauckler LJ (1996) J Electrochem Soc 143:530
Jørgensen MJ, Primdahl S, Bagger C, Mogensen M (2001) Solid State Ionics 139:1
Baumann FS, Fleig J, Konuma M, Starke U, Habermeier HU, Maier J (2005) J Electrochem Soc 152:A2074
Zhu XD, Sun KN, Zhang NQ, Chen XB, Wu LJ, Jia DC (2007) Electrochem Commun 9:431
Yang YL, Chen CL, Chen SY, Chu CW, Jacobson AJ (2000) J Electrochem Soc 47:4001
Murray PE, Sever MJ, Barnett SA (2002) Solid State Ionics 148:27
Kek D, Panjan P, Wanzenberg E, Jamnik J (2001) J Eur Ceram Soc 21:1861
Fu CJ, Chan SH, Liu QL, Ge XM, Pasciak G (2010) Int J Hydrocarb Eng 35:301
Shao J, Tao YK, Wang JX, Xu C, Wang WG (2009) J Alloys Compd 484:263
Leng YJ, Chan SH, Liu QL (2008) Int J Hydrocarb Eng 33:3808
Acknowledgements
This work was supported by the Natural Scientific Research Innovation Foundation in Harbin Institute of Technology (No. HIT. NSRIF. 2008. 22), the China Postdoctoral Science Foundation (Nos. 20080430134 and 200902384), and Open Project of State Key Laboratory of Urban Water Resource and Environment, Harbin Institute of Technology (No.QA201027).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, X., Chen, X., Yuan, Y. et al. Influences of synthesis route and preparation process on the electrochemical properties of Fe-doped strontium cobaltite. J Solid State Electrochem 16, 313–319 (2012). https://doi.org/10.1007/s10008-011-1329-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-011-1329-4