Abstract
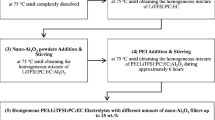
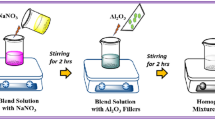
Ionic conductivity, differential scanning calorimetry (DSC), and 1H nuclear magnetic resonance (NMR) measurements have been performed on (PEO)9Mg(ClO4)2 and (PEO)9Mg(ClO4)2 + Al2O3 (neutral, nanoporous) polymer electrolyte systems. It is observed that the conductivity enhances due to the presence of filler up to 15 wt.% and then decreases. The NMR results are consistent with the idea that the conductivity enhancement is mainly due to the increase in chain mobility and ionic mobility of the solid polymer electrolyte caused by increased amorphocity of the electrolyte due to the presence of filler. DSC results also demonstrate that the fraction of the amorphous phase has increased due to the addition of the filler.




Similar content being viewed by others
References
Blumberg AA, Pollack SS, Hoere CAJ (1964) J Polym Sci Part Polym Chem 2:2499
James DB, Wetton RE, Brown DS (1979) Polym J 20:187. doi:10.1016/0032-3861(79)90220-9
Perera K, Dissanayake MAKL, Bandaranayake PWSK (2004) Mater Res Bull 39:1745. doi:10.1016/j.materresbull.2004.03.027
Liebenow C (1998) Electrochim Acta 43:1235. doi:10.1016/S0013-4686(97)10026-3
Kumar GG, Munichandraih N (2000) Solid State Ion 128:203. doi:10.1016/S0167-2738(00)00276-9
Reddy MJ, Chu PP (2002) J Power Sources 109:340. doi:10.1016/S0378-7753(02)00084-8
Armand M, Chabagno JM, Duclot M (1979) Fast ion transport in solids. North Holland, New York
Mac Callum JR, Vincent CA (1987) Polymer electrolyte reviews. Elsevier, London
Gray FM (1997) In: “Polymer electrolytes”. Royal Soc Chem Materials Monographs, Cambridge
Tsunemi K, Ohno H, Tsuchida E (1983) Electrochim Acta 28:833. doi:10.1016/0013-4686(83)85155-X
Weston JE, Steele BCJ (1982) Solid State Ion 7:75. doi:10.1016/0167-2738(82)90072-8
Croce F, Appetechchi GB, Persi L, Scrosati B (1998) Nature 394:456. doi:10.1038/28818
Marcinek M, Bac A, Lipka P, Zaleska A, Zukowska G, Borkowska R, Wieczorek W (2000) J Phys Chem B 104:11088. doi:10.1021/jp0021493
Dissanayake MAKL, Jayathilaka PARD, Bokalawala RSP, Albinsson I, Mellander BE (2003) J Power Sources 119–121:409. doi:10.1016/S0378-7753(03)00262-3
Croce F, Persi L, Scrosati B, Serraino-Fiory F, Plichta E, Hendrickson MA (2001) Electrochim Acta 46:2457. doi:10.1016/S0013-4686(01)00458-3
Stoeva Z, Martin-Litas I, Staunton E, Andreev YG, Bruce PG (2003) J Am Chem Soc 125:4619. doi:10.1021/ja029326t
Golodnitsky D, Livshits E, Ulus A, Peled E (2002) Polym Adv Technol 13:683. doi:10.1002/pat.266
Chung SH, Wang Y, Persi L, Croce F, Greenbaum SG, Scrosati B, Plichta E (2001) J Power Sources 97–98:644. doi:10.1016/S0378-7753(01)00748-0
Jayathilaka PARD, Dissanayake MAKL, Albinsson I, Mellander BE (2002) Electrochim Acta 47:3257. doi:10.1016/S0013-4686(02)00243-8
Jayathilaka PARD, Bokalawela RSP, Bandaranayake PWSK, Bandara LRAK, Dissanayake MAKL (2003) Solid state ionics: trends in new millennium. World Scientific, Singapore, pp 337–343
Freibolin H (1993) Basic one and two dimensional NMR spectroscopy. VCH, Weinheim
Acknowledgments
W.D.D. Wijesiri is acknowledged for her help during this work. Authors wish to thank the International Programmes in the Physical Sciences (IPPS), Uppsala University, Sweden for research support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ekanayake, P., Dissanayake, M.A.K.L. Effect of nanoporous alumina filler on conductivity enhancement in PEO9(MgClO4)2 polymer electrolyte: a 1H NMR study. J Solid State Electrochem 13, 1825–1829 (2009). https://doi.org/10.1007/s10008-008-0768-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-008-0768-z