Abstract
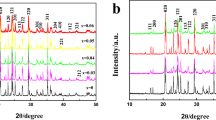
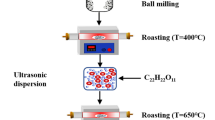
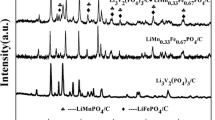
Nano-crystalline LiFePO4 and LiMg0.05Fe0.95PO4 cathode materials were synthesized by sol–gel method in argon atmosphere using succinic acid as a chelating agent. Physico-chemical characterizations were done by thermogravimetric and differential thermal analysis, X-ray diffraction, scanning electron microscopy, transmittance electron microscopy, and Raman spectroscopy. Electrochemical behavior of the cathode materials were analyzed using cyclic voltammetry, and galvanostatic charge/discharge cycling studies were employed to characterize the reaction of lithium-ion insertion into and extraction from virginal and magnesium-doped LiFePO4, in the voltage range 2.5 to 4.5 V (Vs Li/Li+) using 1 M LiPF6 with 1:1 ratio of ethylene carbonate and dimethyl carbonate as electrolytes. LiMg0.05Fe0.95PO4 exhibits initial charge and discharge capacities of 159 and 141 mAh/g at 0.2 C rate respectively, as compared to 121 and 107 mAh/g of pristine LiFePO4. Furthermore, LiMg0.05Fe0.95PO4 has retained more than 89% of the capacity even after 60 cycles. Hence, LiMg0.05Fe0.95PO4 is a promising cathode material for rechargeable lithium-ion batteries.








Similar content being viewed by others
References
Padhi AK, Nanjundaswamy KS, Goodenough JB (1997) J Electrochem Soc 144:1188
Padhi AK, Nanjundaswamy KS, Masquelier C, Okada S, Goodenough JB (1997) J Electrochem Soc 144:1609
Wang GX, Bewlay S, Yao J, Ahn JH, Dou SX, Liu HK (2004) Electrochem Solid State Lett 7:A506
Yamada A, Chung SC, Hinokuma K (2001) J Electrochem Soc 148:A224
Delacourt C, Poizot P, Morerette M, Tarascon J, Masquelier C (2004) Chem Mater 16:93
Takahashi M, Tobishima S, Takei K, Sakuai Y (2002) Solid State Ionics 148:283
Chung SY, Bloking JT, Chiang YM (2002) Nat Mater 1:123
Yang SF, Zavalij PY, Whittingham MS (2001) Electrochem Commun 3:505
Yang SF, Song YN, Zavalij PY, Whitingham MS (2002) Electrochem Commun 4:239
Shiraishi K, Dokko K, Kanamura K (2005) J Power Sources 146:555
Higuchi M, Katayama K, Azuma Y, Yukawa M, Suhara M (2003) J Power Sources 119:258
Croce F, Epitanio AD, Hassoun J, Deptula A, Olczac T, Scrosati B (2002) Electrochem Solid State Lett 5:A47
Yang JS, Xu JJ (2004) Electrochem Solid State Lett 7:A515
Fu LJ, Liu H, Wu YP, Rahm E, Holze R, Wu HQ (2005) Prog Mater Sci 50:881
Bewlay SL, Konstantinov K, Wang GX, Dou SX, Liu HK (2004) Nat Mater Lett 58:1788
Cho TH, Chung HT (2004) J Power Sources 133:272
Park HS, Son JT, Chung HT, Kim SJ, Lee CH, Him HG (2003) Electrochem Commun 5:839
Prosini PP, Carewska M, Scaccia S, Wisniewski P, Pasquali M (2003) Electrochim Acta 48:4205
Song SW, Reade RP, Kostecki R, Striebel KA (2005) J Electrochem Soc 153:A12
Sides CR, Croce F, Young VY, Martin CR, Scrosati B (2005) Electrochem Solid State Lett 8:A484
Huang H, Yin SC, Nazar LF (2001) Electrochem Solid State Lett 4:A170
Ravet N, Choccinard Y, Magman JF, Besner S, Gauthier M, Armand M (2001) J Power Sources 97:503
Zaghib K, Shim J, Guerfi A, Charest P, Striebed KA (2005) Electrochem Solid State Lett 8:A207
Myang ST, Komaba S, Hirosak N, Yashiro H, Kumakai N (2004) Electrochim Acta 49:4213
Spong AD, Vitins G, Owen JR (2005) J Electrochem Soc 152:A2376
Streitsove VA, Belokoneva EL, Tsirelson VG, Hansen N (1993) Acta Crystallogr B Struct Sci 49:147
Liu H, Cao Q, Fu LJ, Li C, Wu YP, Wu HQ (2006) Electrochem Commun 8:1553
Prosini PP, Zane D, Pasquali M (2001) Electrochem Acta 46:3517
Lee J, Teja AS (2005) J Supercrit Fluids 35:83
JCPDS card No 40-1499
Liu H, Cao Q, Fu LJ, Li C, Wu YP, Wu HQ (2006) Electrochem Commun 8:1553
Nakamura T, Sakumoto K, Okamoto M, Seki S, Kobayashi Y, Takeuchi T, Tabuchi M, Yamada Y (2007) J Power Sources 174:435
Nakamura T, Mima Y, Tabuchi M, Yamada Y (2006) J Electrochem Soc 153:A1108
Burba CM, Frech R (2004) J Electrochem Soc 151:A1032
Paques-Ledent MT, Tarte P (1974) Spectrochim Acta Part A 30:673
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arumugam, D., Paruthimal Kalaignan, G. & Manisankar, P. Synthesis and electrochemical characterizations of nano-crystalline LiFePO4 and Mg-doped LiFePO4 cathode materials for rechargeable lithium-ion batteries. J Solid State Electrochem 13, 301–307 (2009). https://doi.org/10.1007/s10008-008-0533-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-008-0533-3