Abstract
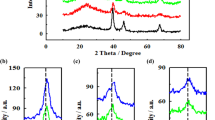
Binary carbon-supported platinum (Pt) nanoparticles were prepared by a chemical reduction method of Pt precursor on two types of carbon materials such as carbon blacks (CBs) and graphite nanofibers (GNFs). Average sizes and loading levels of Pt metal particles were dependent on a mixing ratio of two carbon materials. The highest electroactivity for methanol oxidation was obtained by preparing the binary carbon supports consisting of GNFs and CBs with a weight ratio of 30:70. Furthermore, with an increase of GNFs content from 0% to 30%, a charge-transfer resistance changed from 19 Ohm cm2 to 11 Ohm cm2. The change of electroactivity or the resistance of catalyst electrodes was attributed to the changes of specific surface area and morphological changes of carbon-supported catalyst electrodes by controlling the mixing ratio of GNFs and CBs.








Similar content being viewed by others
References
McNicol BD, Rand DAJ, Williams KR (1999) J Power Sources 83:15
Wasmus S, Kuver A (1999) J Electroanal Chem 461:14
Arico AS, Srinivasan S, Antonucci V (2001) Fuel Cells 1:133
Allen RG, Lim C, Yang LX, Scott K, Roy S (2005) J Power Sources 143:142
Scott K, Taama WM, Argyropoulos P (1999) J Power Sources 79:43
Joo SH, Choi SJ, Oh I, Kwak J, Liu Z, Terasaki O, Ryoo R (2001) Nature 412:169
Kuk ST, Wieckowski A (2005) J Power Sources 141:1
Rajesh B, Thampi KR, Bonard JM, Mathieu HJ, Xanthopoulos N, Viswanathan B (2005) J Power Sources 141:35
Bessel CA, Laubernds K, Rodriguez NM, Baker RTK (2001) J Phys Chem B 105:1115
Steigerwalt ES, Deluga GA, Cliffel DE, Lukehart CM (2001) J Phys Chem B 105:8097
Park SJ, Jung HJ, Nah CW (2003) Polymer (Korea) 27:46
Sakaguchi M, Uematsu K, Sakata A, Sato Y, Sato M (1989) Electrochim Acta 34:625
Watanabe M, Makita K, Usami H, Motoo S (1986) J Electroanal Chem 197:195
Liu YC, Qiu XP, Huang YQ, Zhu WT (2002) J Power Sources 111:160
Pozio A, Francesco MD, Cemmi A, Cardellini F, Giorgi L (2002) J Power Sources 105:13
Cherstiouk OV, Simonov PA, Savinova ER (2003) Electrochim Acta 48:3851
Arico AS, Baglio V, Modica E, Di Blasi A, Antonucci V (2004) Electrochem Commun 6:164
Bonnemann H, Brinkmann R, Brijoux W, Dinjus E, Joussen T, Korall B (1991) Angew Chem Int Ed Engl 30:1312
Gotz M, Wendt H (1998) Electrochim Acta 43:3637
Hamnett A (1997) Catal Today 38:445
Tang H, Chen JH, Huang ZP, Wang DZ, Ren ZF, Nie LH, Kuang YF, Yao SZ (2004) Carbon 42:191
Liu Z, Lin X, Lee JY, Zhang W, Han M, Gan LM (2002) Langmuir 18:4054
Liu Z, Lee JY, Chen W, Han M, Gan LM (2004) Langmuir 20:181
Jusys Z, Kaiser J, Behm RJ (2003) Langmuir 19:6759
Deivaraj TC, Lee JY (2005) J Power Sources 142:43
Li W, Liang C, Qiu J, Zhou W, Han H, Wei Z, Sun G, Xin Q (2002) Carbon 40:791
Li W, Liang C, Qiu J, Zhou W, Qiu J, Zhou Z, Sun G, Xin Q (2003) J Phys Chem B 107:6292
Rajesh B, Ravindranathan TK, Bonard JM, Viswanathan B (2000) J Mater Chem 10:1757
Schmidt TJ, Noeske M, Gasteiger HA, Behm RJ (1998) J Electrochem Soc 145:925
Watanabe M, Igarashi H, Yosioka K (1995) Electrochim Acta 40:329
Schmidt TJ, Gasteiger HA, Stab GD, Urban PM, Kolb DM, Behm RJ (1998) J Electrochem Soc 145:2354
Kinoshita K (1988) Carbon: electrochemical and physicochemical properties. Wiley, New York, p 31
Radmilovic V, Gasteiger HA, Ross PN (1995) J Catal 154:98
Park K, Choi J, Kwon B, Lee S, Sung Y, Ha H, Hong S, Kim H, Wieckowski A (2002) J Phys Chem B 106:1869
Liu Z, Ling XY, Su X, Lee JY (2004) J Phys Chem B 108:8234
Gao L, Huang H, Korzeniewski C (2004) Electrochim Acta 49:1281
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, S., Park, SJ. Preparation and electrochemical behaviors of platinum nanoparticles impregnated on binary carbon supports as catalyst electrodes of direct methanol fuel cells. J Solid State Electrochem 11, 821–828 (2007). https://doi.org/10.1007/s10008-006-0228-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-006-0228-6