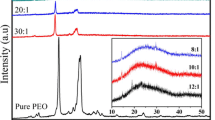
Polymer electrolyte systems were prepared for the first time by dissolution of amidomagnesium chlorides in poly(ethylene oxide), (PEO). For the preparation, solutions of (hexamethyldisilylamido)magnesium chloride, (dimethylpyrrolyl)magnesium chloride, (diisopropylamido)magnesium chloride, piperidinomagnesium chloride and morpholinomagnesium chloride were chosen. The composition of these polymer electrolyte systems corresponds to the general formula R2NMgCl·P(EO)n·THF. Most work has been done with the system (hexamethyldisilylamido)magnesium chloride in PEO, (Me3Si)2NMgCl·P(EO)n·THF, with n= 3, 4, 5, or 7. The electrolytes have a soft rubber-like consistency. At 30 °C, electrical conductivities of 10−6–10−5 S/cm were found. The conductivities were measured in the temperature range 20–60 °C. Within this temperature range a linear dependence of the logarithms of the conductivity on the inverse temperature was found and activation energies for the conducting process of 30–60 kJ/mol were calculated. Using those polymer electrolytes with a high content of the amidomagnesium compound, a reversible magnesium deposition takes place by cathodic reduction at potentials below −1.9 V vs. a Ag/AgCl reference electrode. These polymer electrolytes were found to be stable against oxidation up to about −0.3 V vs. Ag/AgCl.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Liebenow, C., Mantey, S. Preparation of solutions of amidomagnesium chlorides in poly(ethylene oxide) and their characterization by conductivity measurements. J Solid State Electrochem 7, 313–316 (2003). https://doi.org/10.1007/s10008-003-0356-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10008-003-0356-1