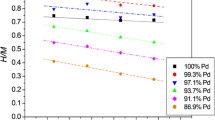
The effect of temperature on hydrogen and deuterium electrosorption into a palladium LVE (limited-volume electrode) has been investigated. A decrease in hydrogen capacity (H/Pd ratio) with increasing temperature has been observed. Temperature strongly influences the plots of measured H(D)/Pd values vs. potential scan rate. In addition, hydrogen absorption was found to be dependent on the composition of the surrounding electrolyte solution. These results have confirmed the hypothesis that two different mechanisms of hydrogen desorption from the palladium electrode take place, namely electrochemical oxidation and non-electrochemical recombination. Further, the ratio between the rate constants for these two processes has been found to change with temperature.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Czerwiński, A., Kiersztyn, I. & Grdeń, M. Temperature influence on hydrogen sorption in palladium limited-volume electrodes (Pd-LVE). J Solid State Electrochem 7, 321–326 (2003). https://doi.org/10.1007/s10008-003-0352-5
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10008-003-0352-5