Abstract
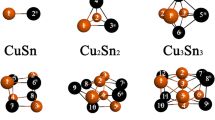
The ground-state structural transition in small lithium clusters Lin (n = 4 − 6) is analyzed based on the many-body expansion of the interaction energy using the total energy calculated by the fixed-node diffusion Monte Carlo (FN-DMC) simulations. The results show that the transition from 2D to 3D structure occurs through an intricate competition of attractive and repulsive interaction energies. As the structure dimensionality increases from 2D to 3D, the electron-correlation contribution to the interaction energy in the isomer of the ground-state structure is always the largest.


Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
The calculations have been carried out using Gaussian03 and Casino code.
References
Bragg A., Verlet J., Kammrath A., Cheshnovsky O., Neumark D. (2004) Hydrated electron dynamics: From clusters to bulk. Science 306(5696):669
Gatteschi D., Caneschi A., Pardi L., Sessoli R. (1994) Large clusters of metal ions: the transition from molecular to bulk magnets. Science 265(5175):1054
Kawai R., Sung M., Weare J. (1992) Structural transition from small clusters to bulk-like structures. In: Physics and chemistry of finite systems: From clusters to crystals. Springer, pp 441–446
Ludwig R. (2001) Water: from clusters to the bulk. Angewandte Chemie International Edition 40(10):1808
Gafner S., Goloven’ko Z. V., Gafner Y. Y., Samsonov V., Kharechkin S., et al. (2009) Structural transitions in small nickel clusters. JETP Letters 89(7):364
Sakurai M., Souto-Casares J., Chelikowsky J.R. (2016) Size dependence of structural stability and magnetization of nickel clusters from real-space pseudopotentials. Physical Review B 94(2):024437
Ishizaki T., Yatsugi K., Akedo K. (2016) Effect of particle size on the magnetic properties of Ni nanoparticles synthesized with trioctylphosphine as the capping agent. Nanomaterials 6(9):172
Daudey J., Novaro O., Kolos W., Berrondo M. (1979) Convergence of multibody expansions for alkaline earth metals. Contrast between magnesium and beryllium clusters. The Journal of Chemical Physics 71(11):4297
Novaro O., Kolos W. (1977) Nonadditive effects in small beryllium clusters. The Journal of Chemical Physics 67(11):5066
Kaplan I., Santamaria R., Novaro O. (1995) Nonadditive interactions and the relative stability of neutral and anionic silver clusters. International Journal of Quantum Chemistry 55(3):237
Hartke B. (1993) Global geometry optimization of clusters using genetic algorithms. The Journal of Physical Chemistry 97(39):9973
Deaven D. M., Ho K. M. (1995) Molecular geometry optimization with a genetic algorithm. Physical Review Letters 75(2):288
Xiao Y., Williams D. E. (1993) Genetic algorithm: a new approach to the prediction of the structure of molecular clusters. Chemical Physics Letters 215(1-3):17
Alexandrova A. N., Boldyrev A. I., Fu Y. J., Yang X., Wang X. B., Wang L. S. (2004) Structure of the Nax Clx+ 1− (x = 1–4) clusters via ab initio genetic algorithm and photoelectron spectroscopy. The Journal of Chemical Physics 121(12):5709
Heiles S., Johnston R. L. (2013) Global optimization of clusters using electronic structure methods. International Journal of Quantum Chemistry 113(18):2091
García-Prieto J., Feng W., Novaro O. (1984) Study of collective effects in the stability and chemisorption properties of finite lithium clusters. Surface Science 147(2-3):555
Kaplan I. G., Hernández-Cobos J., Ortega-Blake I., Novaro O. (1996) Many-body forces and electron correlation in small metal clusters. Physical Review A 53(4):2493
Brito B., Hai G.Q., Cândido L. (2019) Quantum Monte Carlo simulation for the many-body decomposition of the interaction energy and electron correlation of small superalkali lithium clusters. The Journal of Chemical Physics 151(1):014303
Frisch M.J., Trucks G.W., Schlegel H.B., Scuseria G.E., Robb M.A., Cheeseman J.R., Montgomery J.A., Vreven T. Jr., Kudin K.N., Burant J.C., Millam J.M., Iyengar S.S., Tomasi J., Barone V., Mennucci B., Cossi M., Scalmani G., Rega N., Petersson G.A., Nakatsuji H., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Klene M., Li X., Knox J.E., Hratchian H.P., Cross J.B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R.E., Yazyev O., Austin A.J., Cammi R., Pomelli C., Ochterski J.W., Ayala P.Y., Morokuma K., Voth G.A., Salvador P., Dannenberg J.J., Zakrzewski V.G., Dapprich S., Daniels A.D., Strain M.C., Farkas O., Malick D.K., Rabuck A.D., Raghavachari K., Foresman J.B., Ortiz J.V., Cui Q., Baboul A.G., Clifford S., Cioslowski J., Stefanov B.B., Liu G., Liashenko A., Piskorz P., Komaromi I., Martin R.L., Fox D.J., Keith T., Al-Laham M.A., Peng C.Y., Nanayakkara A., Challacombe M., Gill P.M.W., Johnson B., Chen W., Wong M.W., Gonzalez C., Pople J.A. (2004) Gaussian 03, revision c.02. Gaussian inc., Wallingford
Brito B., Cândido L., Rabelo J. T., Hai G. Q. (2014) Binding energies of small lithium clusters: a comparison of different theoretical calculations. Chemical Physical Letters 616: 212
Kolos W., Nieves F., Novaro O. (1976) The importance of non-additive terms in the interaction energy of tetrahedral Be4 clusters. Chemical Physics Letters 41(3):431
Needs R., Towler M., Drummond N., Lopez Rios P., Trail J. (2020) Variational and diffusion quantum Monte Carlo calculations with the CASINO code. The Journal of Chemical Physics 152(15):154106
Foulkes W., Mitas L., Needs R., Rajagopal G. (2001) Quantum Monte Carlo simulations of solids. Reviews of Modern Physics 73(1):33
Reynolds P. J., Ceperley D. M., Alder B. J., Lester W.A. Jr. (1982) Fixed-node quantum Monte Carlo for molecules. The Journal of Chemical Physics 77(11):5593
Feller D. (1993) The use of systematic sequences of wave functions for estimating the complete basis set, full configuration interaction limit in water. The Journal of Chemical Physics 98(9):7059
Bréchignac C., Busch H., Cahuzac P., Leygnier J. (1994) Dissociation pathways and binding energies of lithium clusters from evaporation experiments. The Journal of Chemical Physics 101(8):6992
Wu C. (1983) Experimental investigation of a stable lithium cluster. The thermochemical study of the molecule Li4. The Journal of Physical Chemistry 87(9):1534
Wu C. (1989) Thermochemical properties of the lithium cluster Li5. The Journal of Chemical Physics 91(1):546
Acknowledgements
The authors acknowledge computational resources from LaMCAD/UFG, and the National Laboratory for Scientific Computing (LNCC/MCTI, Brazil) for providing HPC resources of the SDumont supercomputer.
Funding
This research receive founds from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa do Estado de Goiás (FAPEG).
Author information
Authors and Affiliations
Contributions
B. G. A. Brito and E. L. Verde: formal analysis, investigation, methodology, writing original draft, visualization. G.-Q. Hai and L. Cândido: conceptualization, methodology, validation, writing, review and editing, project administration, and funding acquisition.
Corresponding author
Ethics declarations
Ethics approval
The manuscript is prepared in compliance with the Ethics in Publishing Policy as described in the Guide for Authors
Consent to participate
The manuscript is approved by all authors for publication.
Consent for publication
The consent for publication was obtained from all participants.
Conflict of interest
The authors declare no competing interests
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This paper belongs to the Topical Collection VIII Symposium on Electronic Structure and Molecular Dynamics – VIII SeedMol
Rights and permissions
About this article
Cite this article
Brito, B.G.A., Verde, E.L., Hai, GQ. et al. Probing the ground-state structural transition in small lithium clusters by quantum Monte Carlo simulations. J Mol Model 27, 207 (2021). https://doi.org/10.1007/s00894-021-04810-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-021-04810-4