Abstract
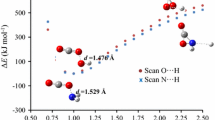
A computational study of metal difluorides (MF2; M = Ca to Zn) and their interactions with carbon dioxide and water molecules was performed. The structural parameter values obtained and the results of AIM analysis and energy decomposition analysis indicated that the Ca–F bond is weaker and less ionic than the bonds in the transition metal difluorides. A deformation density plot revealed the stablizing influence of the Jahn–Teller effect in nonlinear MF2 molecules (e.g., where M= Sc, Ti, Cr). An anaysis of the metal K-edge peaks of the difluorides showed that shifts in the edge energy were due to the combined effects of the ionicity, effective nuclear charge, and the spin state of the metal. The interactions of CO2 with ScF2 (Scc3 geometry) and TiF2 (Tic2 geometry) caused CO2 to shift from its usual linear geometry to a bent geometry (η2(C=O) binding mode), while it retained its linear geometry (η1(O) binding mode) when it interacted with the other metal difluorides. Energy decomposition analysis showed that, among the various geometries considered, the Scc3 and Tic2 geometries possessed the highest interaction energies and orbital interaction energies. Heavier transition metal difluorides showed stronger affinities for H2O, whereas the lighter transition metal (Sc and Ti) difluorides preferred CO2. Overall, the results of this study suggest that fluorides of lighter transition metals with partially filled d orbitals (e.g., Sc and Ti) could be used for CO2 capture under moist conditions.

Interaction of metal difluorides with carbon dioxide and water












Similar content being viewed by others
References
Sumida K, Rogow DL, Mason JA, McDonald TM, Bloch ED, Herm ZR, Bae T-H, Long JR (2012) Carbon dioxide capture in metal–organic frameworks. Chem Rev 112(2):724–781. https://doi.org/10.1021/cr2003272
Sanna A, Uibu M, Caramanna G, Kuusik R, Maroto-Valer MM (2014) A review of mineral carbonation technologies to sequester CO2. Chem Soc Rev 43(23):8049–8080. https://doi.org/10.1039/c4cs00035h
Spigarelli BP, Kawatra SK (2013) Opportunities and challenges in carbon dioxide capture. J CO2 Util 1:69–87. https://doi.org/10.1016/j.jcou.2013.03.002
Sreenivasulu B, Sreedhar I, Suresh P, Raghavan KV (2015) Development trends in porous adsorbents for carbon capture. Environ Sci Technol 49(21):12641–12661. https://doi.org/10.1021/acs.est.5b03149
Zhang B, Duan Y, Johnson K (2012) Density functional theory study of CO2 capture with transition metal oxides and hydroxides. J Chem Phys 136(6):064516. https://doi.org/10.1063/1.3684901
Dey C, Kundu T, Biswal BP, Mallick A, Banerjee R (2014) Crystalline metal-organic frameworks (MOFs): synthesis, structure and function. Acta Crystallogr B 70(1):3–10. https://doi.org/10.1107/S2052520613029557
Wang J, Huang L, Yang R, Zhang Z, Wu J, Gao Y, Wang Q, O’Hare D, Zhong Z (2014) Recent advances in solid sorbents for CO2 capture and new development trends. Energy Environ Sci 7(11):3478–3518. https://doi.org/10.1039/c4ee01647e
Mitoraj MP (2011) Bonding in ammonia borane: an analysis based on the natural orbitals for chemical valence and the extended transition state method (ETS-NOCV). J Phys Chem A 115(51):14708–14716. https://doi.org/10.1021/jp209712s
Mitoraj MP, Parafiniuk M, Srebro M, Handzlik M, Buczek A, Michalak A (2011) Applications of the ETS-NOCV method in descriptions of chemical reactions. J Mol Model 17(9):2337. https://doi.org/10.1007/s00894-011-1023-6
Su P, Li H (2009) Energy decomposition analysis of covalent bonds and intermolecular interactions. J Chem Phys 131(1):014102. https://doi.org/10.1063/1.3159673
Mitoraj M, Michalak A (2007) Natural orbitals for chemical valence as descriptors of chemical bonding in transition metal complexes. J Mol Model 13(2):347–355. https://doi.org/10.1007/s00894-006-0149-4
Mitoraj MP, Michalak A (2013) Theoretical description of halogen bonding—an insight based on the natural orbitals for chemical valence combined with the extended-transition-state method (ETS-NOCV). J Mol Model 19(11):4681–4688. https://doi.org/10.1007/s00894-012-1474-4
Frenking G (2001) Understanding the nature of the bonding in transition metal complexes: from Dewar’s molecular orbital model to an energy partitioning analysis of the metal–ligand bond. J Organomet Chem 635(1):9–23. https://doi.org/10.1016/S0022-328X(01)01154-8
Bhatt PM, Belmabkhout Y, Cadiau A, Adil K, Shekhah O, Shkurenko A, Barbour LJ, Eddaoudi M (2016) A fine-tuned fluorinated MOF addresses the needs for trace CO2 removal and air capture using physisorption. J Am Chem Soc 138(29):9301–9307. https://doi.org/10.1021/jacs.6b05345
Wilmer CE, Farha OK, Bae Y-S, Hupp JT, Snurr RQ (2012) Structure–property relationships of porous materials for carbon dioxide separation and capture. Energy Environ Sci 5(12):9849–9856. https://doi.org/10.1039/c2ee23201d
Zhang D-S, Chang Z, Li Y-F, Jiang Z-Y, Xuan Z-H, Zhang Y-H, Li J-R, Chen Q, Hu T-L, Bu X-H (2013) Fluorous metal–organic frameworks with enhanced stability and high H2/CO2 storage capacities. 3:3312. doi:https://doi.org/10.1038/srep03312. https://www.nature.com/articles/srep03312#supplementary-information
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery Jr JA, Peralta JE, Ogliaro F, Bearpark MJ, Heyd J, Brothers EN, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell AP, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09. Gaussian, Inc., Wallingford
Bader RFW (1991) A quantum theory of molecular structure and its applications. Chem Rev 91(5):893–928. https://doi.org/10.1021/cr00005a013
Popelier PLA (1994) An analytical expression for interatomic surfaces in the theory of atoms in molecules. Theor Chim Acta 87(6):465–476. https://doi.org/10.1007/bf01127809
Popelier PLA (1996) MORPHY, a program for an automated “atoms in molecules” analysis. Comput Phys Commun 93(2):212–240
Popelier PLA (1996) On the differential geometry of interatomic surfaces. Can J Chem 74(6):829–838. https://doi.org/10.1139/v96-092
Popelier PLA (1996) Integration of atoms in molecules: a critical examination. Mol Phys 87(5):1169–1187. https://doi.org/10.1080/00268979600100781
Popelier PLA (1998) A method to integrate an atom in a molecule without explicit representation of the interatomic surface. Comput Phys Commun 108(2):180–190
te Velde G, Bickelhaupt FM, Baerends EJ, et al. (2001) Chemistry with ADF. J Comput Chem 22:931--967. doi:https://doi.org/10.1002/jcc.1056
Baerends EJ, Ziegler T, Atkins AJ, et al. (2017) ADF2017. SCM, Amsterdam. https://www.scm.com
Zhao Y, Truhlar DG (2008) The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor Chem Accounts 120(1):215–241. https://doi.org/10.1007/s00214-007-0310-x
Atkins AJ, Bauer M, Jacob CR (2015) High-resolution X-ray absorption spectroscopy of iron carbonyl complexes. Phys Chem Chem Phys 17(21):13937–13948. https://doi.org/10.1039/c5cp01045d
van Leeuwen R, Baerends EJ (1994) Exchange-correlation potential with correct asymptotic behavior. Phys Rev A 49(4):2421–2431
Van Lenthe E, Baerends EJ (2003) Optimized Slater-type basis sets for the elements 1–118. J Comput Chem 24(9):1142–1156. doi:https://doi.org/10.1002/jcc.10255
Vogt N (2001) Equilibrium bond lengths, force constants and vibrational frequencies of MnF2, FeF2, CoF2, NiF2, and ZnF2 from least-squares analysis of gas-phase electron diffraction data. J Mol Struct 570(1):189–195. https://doi.org/10.1016/S0022-2860(01)00510-5
Wang SG, Schwarz WHE (1998) Density functional study of first row transition metal dihalides. J Chem Phys 109(17):7252–7262. https://doi.org/10.1063/1.477359
Vest B, Schwerdtfeger P, Kolonits M, Hargittai M (2009) Chromium difluoride: probing the limits of structure determination. Chem Phys Lett 468(4):143–147. https://doi.org/10.1016/j.cplett.2008.12.008
Pandey RK, Waters K, Nigam S, He H, Pingale SS, Pandey AC, Pandey R (2014) A theoretical study of structural and electronic properties of alkaline-earth fluoride clusters. Comput Theor Chem 1043:24–30. https://doi.org/10.1016/j.comptc.2014.05.007
Freund HJ, Roberts MW (1996) Surface chemistry of carbon dioxide. Surf Sci Rep 25(8):225–273. https://doi.org/10.1016/S0167-5729(96)00007-6
Stoneburner SJ, Livermore V, McGreal ME, Yu D, Vogiatzis KD, Snurr RQ, Gagliardi L (2017) Catechol-ligated transition metals: a quantum chemical study on a promising system for gas separation. J Phys Chem C 121(19):10463–10469. https://doi.org/10.1021/acs.jpcc.7b02685
Ev L, Baerends EJ, Snijders JG (1993) Relativistic regular two-component Hamiltonians. J Chem Phys 99(6):4597–4610. https://doi.org/10.1063/1.466059
Ev L, Baerends EJ, Snijders JG (1994) Relativistic total energy using regular approximations. J Chem Phys 101(11):9783–9792. https://doi.org/10.1063/1.467943
Park J, Kim H, Han SS, Jung Y (2012) Tuning metal–organic frameworks with open-metal sites and its origin for enhancing CO2 affinity by metal substitution. J Phys Chem Lett 3(7):826–829. https://doi.org/10.1021/jz300047n
Lü K, Zhou J, Zhou L, Chen XS, Chan SH, Sun Q (2012) Pre-combustion CO2 capture by transition metal ions embedded in phthalocyanine sheets. J Chem Phys 136(23):234703. https://doi.org/10.1063/1.4729471
Kunkel C, Vines F, Illas F (2016) Transition metal carbides as novel materials for CO2 capture, storage, and activation. Energy Environ Sci 9(1):141–144. https://doi.org/10.1039/c5ee03649f
Acknowledgements
This work was supported by the INSPIRE program under the Department of Science and Technology, India [DST/INSPIRE fellowship/2015/IF0672].
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOC 2132 kb)
Rights and permissions
About this article
Cite this article
Arokiyanathan, A.L., Lakshmipathi, S. Molecular properties of metal difluorides and their interactions with CO2 and H2O molecules: a DFT investigation. J Mol Model 23, 345 (2017). https://doi.org/10.1007/s00894-017-3511-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-017-3511-9