Abstract
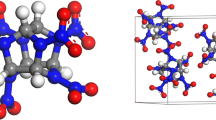
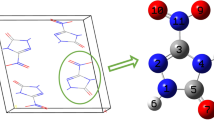
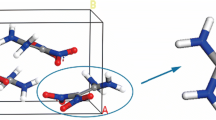
The glass transition temperature (T g) and density of poly-(phthalazinone ether sulfone ketone) (PPESK A) were estimated by molecular dynamic (MD) simulation. A novel poly-(phthalazinone ether sulfone ketone) (PPESK B) was constructed by introducing nitrol and amini energetic groups into PPESK A, and T g and density were also simulated for PPESK B. The estimated T g values of PPESK A were very close to experimental results, while for PPESK B three estimated values differed by < 5 K. The interactions between explosives and polymer binders of polymer bonded explosives (PBXs) were simulated by MD. Comparison of the cohesive energy densities (CED) and solubility parameter (δ) values of PBXs, polymer binders, and mono-explosives indicate that, upon introducing polymer binders, the CED and δ values of PBXs decreased compared with those of corresponding mono-explosives. The binding energies (E bind) imply that 2,4,6-trinitrotoluene-based PBXs are more stable than 1,3,5-triamino-2,4,6-trinitrobenzene (TATB)-based PBXs. The mechanical properties, Young’s modulus E, shear modulus G, bulk modulus K, Poisson’s ratio γ and Cauchy pressure (C 12 –C 44) of the PBXs were assessed. The rigidity of the PBXs was found to be lower than that of mono-explosives. All K/G values were positive, indicating that PBXs are flexible. Based on these mechanical properties results, PBXs using PPESK B as a binder are superior to those using PPESK A as a binder. Due to the low C 12 –C 44 values of the PBXs, the ductility of the materials of the fracture surface is poorer, especially for TATB-based PBXs.





Similar content being viewed by others
References
Shu YJ, Huo JC (2011) Theory of explosives. Chemical Industry, Beijing
Sun GX (1984) Polymer mixed explosives. National Defense Industry, Beijing
Sun YB, Hui JM, Cao XM (1995) Military mixed explosives. Weapon Industry, Beijing
Wang Z, Li G, Lao Y, Li H (2005) Study on thermal stability of PBX-HKF by accelerating rate calorimeter. Chin J Energ Mater 13(2):113–114
Elbeih A, Pachman J, Zeman S (2010) Thermal stability and detonation characteristic of pressed and elastic explosive on the basis of selected cyclic nitramines. Cent Eur J Chem 7:115–129
Singh C, Felix SP, Soni P (2005) Study on energetic compounds, Part 31. Thermolysis and kinetic of RDX andsome of its plastic bonded explosives. Theor Chim Acta 426:131–139
Singh C, Felix SP, Pandey DK, Agrawal JP, Sikder AK (2005) Study on energetic compounds, Part XXXIX. Thermal analysis of a plastic bonded explosive containing RDX and HTPB. J Therm Anal Calorim 79:631–635
Zhang JQ, Zhu H, Zhang W, Zhang JQ, Yang SJ (2006) Research progress of energetic binder system used in solid propllants. Chem Propell Polym Mater 4(3):6–9
He LM, Xiao ZL, Zhang XZ, Jing DQ (2003) The research and development on energetic binders for propellants abroad. Chin J Energ Mater 11(2):99–102
Wu YG, Wu XQ, Zhang L, Zhang CF (2008) Research progresses of energetic adhesive system for gunpowder. China Adhesives 17(8):47–50
Jian XG, Liao GX, Wang JY (2002) Research progress of poly(arylene ether ketone)s and poly(arylene ether sulfone)s containing phthalazinone moieties. China Plastic 16(4):11–15
Jan XG, Wang JY (2012) Progress on high performance engineering plastics containing phthalazinone moieties and their applications. Materials China 31(2):16–22
Agrawal JP, Hodgson RD (2007) Organic chemistry of explosives. Wiley, New York
Davis TL (1953) The chemistry of powder and explosives: complete in one volume. Wiley, New York
Cooper PW (1996) Explosives engineering. Wiley-VCH, New York
Ou YX, Meng Z, Liu JQ (2007) Review of the development of application technologies of CL-20. Chem Ind Eng Prog 26(12):1690–1694
Qiu L, Xiao HM, Zhu WH, Xiao JJ, Zhu W (2006) Ab initio and molecular dynamics studies of crystalline TNAD (trans-1,4,5,8-tetranitro-1,4,5,8-tetra azadecalin). J Phys Chem B 110(22):10651–10661
Carper WR, Davis LP, Extine MW (1982) Molecular structure of 2,4,6-trinitrotoluene. J Phys Chem 86:459
Cady HH, Larson AC (1965) The crystal structure of 1,3,5-triamino-2,4,6-trinitrobenzene. Acta Crystallogr 18:485–496
Accelrys Software. Materials Studio Accelrys Software Inc, San Diego, CA
Andersen HC (1980) Molecular dynamics simulations at constant pressure and/or temperature. J Chem Phys 72(4):2384–2393
Berendsen HJC, Postma JPM, van Gunsteren WF, DiNola A, Haak JR (1984) Molecular dynamics with coupling to an external bath. J Chem Phys 81:3684–3690
Scatchard G (1931) Equilibria in non-electrolyte solutions in relation to the vapor pressures and densities of the components. Chem Rev 8(2):321–333
Watt JP, Davies GF, O’Connell RJ (1976) The elastic properties of composite materials. Rev Geophys Space Phys 14:541–563
Qiu L, Zhu WH, Xiao JJ, Zhu W, Xiao HM, Huang H, Li JS (2007) Molecular dynamics simulations of trans-1,4,5,8-tetranitro-1,4,5,8-tetraazadecalin-based polymer-bonded explosives. J Phys Chem B 111:1559–1566
Sharma J, Garrett WL, Owens FJ, Vogel VL (1982) X-ray photoelectronic structure, ultraviolet, and isothermal decomposition of 1,3,5-triamino-2,4,6-trinitrobenzene. J Phys Chem 86(9):1657–1661
Wang XC, Huang H, Nie FD (2001) The investigation of surface modification of TATB particle. Chin J Expl Propell 1(24):33–35
Wu JL (1993) Elasticity. Tongji University Press, Shanghai
Xiao JJ, Huang H, Li JS, Zhang H, Zhu W, Xiao HM (2007) MD simulation study on the mechanical properties of HMX crystals and HMX/F2311 PBXs. Acta Chim Sinica 65(17):1746–1750.
Watt JP, Davies GF, O’Connell RJ (1976) The elastic properties of composite materials. Rev Geophys Space Phys 14:541–563
Weiner JH (1983) Statistical mechanics of elasticity. Wiley, New York
Acknowledgments
The authors appreciate financial support from the National Natural Science Foundation of China (grant. no. 51373159).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
ESM 1
(DOCX 1029 kb)
Rights and permissions
About this article
Cite this article
Shu, Y., Yi, Y., Huo, J. et al. Interactions between poly-(phthalazinone ether sulfone ketone) (PPESK) and TNT or TATB in polymer bonded explosives: a molecular dynamic simulation study. J Mol Model 23, 334 (2017). https://doi.org/10.1007/s00894-017-3492-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-017-3492-8