Abstract
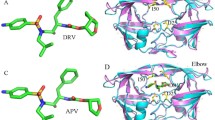
The human immunodeficiency virus type 1 protease (HIV-1 PR) is a major target for the design of anti-AIDS (acquired immune deficiency syndrome) drugs. Some C 2-symmetric inhibitors have been designed for the C 2-symmetric binding pocket of HIV-1PR. The crystallographic structures reveal that the binding modes are not C 2-symmetric for C 2- symmetric inhibitors binding to PR. In this work, four molecular dynamics (MD) simulations were performed to investigate the binding modes between four C 2-symmetric inhibitors (6 AD, 6AG, 6FD and 6FG) and PR, as well as the stabilities of these inhibitors in the binding pocket. Analysis of the hydrophobic surface of the binding pocket shows that it is necessary to add a polar group to the P1 (benzyl) and P2 (phenyl) groups of the inhibitor 6AD. Analysis of the hydrogen bonds formed between inhibitors and residues (Asp25/Asp25′, Ile50/Ile50′) indicates that the steric structures of the inhibitors are not suitable for the binding pocket. The two increased polar groups of trifluoromethyl and formamido meet the needs of the binding pocket for polar molecules. The inhibitor with both these groups (6FG) has stronger stability than the other three inhibitors, which have only one (6AG and 6FD) or none (6AD) of these groups. The ranking of binding free energies calculated by molecular mechanics-generalized born surface area (MM-GBSA) method agrees well with the experimental data. It is expected that this study will provide theoretical guidance for the design of anti-AIDS drugs targeting HIV-1PR.

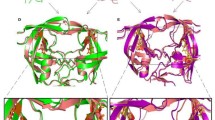
Left Inhibitor 6AD in the binding pocket of human immunodeficiency virus type 1 protease. Right Differences of free energies for trifluoromethyl and formaminde






Similar content being viewed by others
References
WHO (2013) UNAIDS report on the global AIDS epidemic 2013, http://www.unaids.org/en/resources/campaigns/globalreport2013/
Wlodawer A, Vondrasek J (1998) Annu Rev Biophys Biomol Struct 27:249–284
Nicholson LK, Yamazaki T, Torchia DA, Grzesiek S, Bax A, Stahl SJ, Kaufman JD, Wingfield PT, Lam PY, Jadhav PK et al (1995) Nat Struct Biol 2:274–280
Zhou T, Zhu J, Wu X, Moquin S, Zhang B, Acharya P, Georgiev IS, Altae-Tran HR, Chuang GY, Joyce MG, Do Kwon Y, Longo NS, Louder MK, Luongo T, McKee K, Schramm CA, Skinner J, Yang Y, Yang Z, Zhang Z, Zheng A, Bonsignori M, Haynes BF, Scheid JF, Nussenzweig MC, Simek M, Burton DR, Koff WC, Mullikin JC, Connors M, Shapiro L, Nabel GJ, Mascola JR, Kwong PD (2013) Immunity 39:245–258
Kwong PD, Mascola JR, Nabel GJ (2013) Nat Rev Immunol 13:693–701
Barbaro G, Scozzafava A, Mastrolorenzo A, Supuran CT (2005) Curr Pharm Des 11:1805–1843
Rodriguez-Barrios F, Gago F (2004) Curr Top Med Chem 4:991–1007
D'Aquila RT, Schapiro JM, Brun-Vezinet F, Clotet B, Conway B, Demeter LM, Grant RM, Johnson VA, Kuritzkes DR, Loveday C, Shafer RW, Richman DD (2002) Top HIV Med 10:21–25
Blum A, Bottcher J, Heine A, Klebe G, Diederich WE (2008) J Med Chem 51:2078–2087
Xu Y, Wang R (2006) Proteins 64:1058–1068
Wu EL, Han K, Zhang JZ (2008) Chemistry 14:8704–8714
Fu C, Shushen L, Xintian D (2013) Acta Chim Sin 71:1035–1040
Shi S, Hu G, Chen J, Zhang S-L, Zhang Q (2009) Acta Chim Sin 67:2791–2797
Ode H, Neya S, Hata M, Sugiura W, Hoshino T (2006) J Am Chem Soc 128:7887–7895
Hu G, Zhu T, Zhang S-L, Wang D, Zhang Q (2010) Eur J Med Chem 45:227–235
Leonis G, Steinbrecher T, Papadopoulos MG (2013) J Chem Inf Model 53:2141–153
Hou T, Zhang W, Wang J, Wang W (2009) Proteins 74:837–846
Stoica I, Sadiq SK, Coveney PV (2008) J Am Chem Soc 130:2639–2648
Wu EL, Wong KY, Zhang X, Han K, Gao J (2009) J Phys Chem B 113:2477–2485
Zhuang S, Zou J, Jiang Y, Mao X, Zhang B, Liu H, Yu Q (2005) J Med Chem 48:7208–7214
Chen X, Zhao X, Wang S, Wang L, Li W, Sun C (2013) Acta Chim Sin 71:199–204
Chen SF, Cao Y, Chen JJ, Chen JZ (2013) J Mol Model 19:5097–5112
Case DA, Darden TA, Cheatham TE, III, Simmerling CL, Wang J, Duke RE, Luo R, Crowley M, Walker RC, Zhang W et al (2008) AMBER 10. University of San Fransisco, San Fransisco, CA, http://ambermd.org/
Duan Y, Wu C, Chowdhury S, Lee MC, Xiong G, Zhang W, Yang R, Cieplak P, Luo R, Lee T, Caldwell J, Wang J, Kollman P (2003) J Comput Chem 24:1999–2012
Cieplak P, Cornell WD, Bayly C, Kollman PAJ (1995) J Comput Chem 16:1357
Wang J, Wolf RM, Caldwell JW, Kollman PA, Case DA (2004) J Comput Chem 25:1157–1174
Jorgensen WL, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) J Chem Phys 79:926
Darden T, York D, Pedersen L (1993) J Chem Phys 98:10089
Ryckaert JP, Ciccotti G, Berendsen HJC (1977) J Comput Phys 23:327
Still WC, Tempczyk A, Hawley RC, Hendrickson T (1990) J Am Chem Soc 112:6127–6129
Weiser J, Shenkin PS, Still WC (1999) J Comput Chem 20:217–230
Onufriev A, Bashford D, Case DA (2004) Proteins: Structure. Function Bioinform 55:383–394
Pearlman DA (2005) J Med Chem 48:7796–7807
Gohlke H, Kiel C, Case DA (2003) J Mol Biol 330:891–913
Kar P, Knecht V (2012) J Phys Chem B 116:2605–2614
Kar P, Lipowsky R, Knecht V (2011) J Phys Chem B 115:7661–7669
Hu G, Zhang Q, Chen LY (2011) J Mol Model 17:1919–1926
Hu G, Wang J (2014) Eur J Med Chem 74:726–735
Hu G, Wang D, Liu X, Zhang Q (2010) J Comput Aided Mol Des 24:687–697
Chen J, Wang J, Zhu W, Li G (2013) J Comput Aided Mol Des 27:965–974
Acknowledgments
This work was partially supported by funding from the National Natural Science Foundation of China (61271378 and 11274206), the Natural Science Foundation of Shandong Province (ZR2013AM014 and ZR2012CL09) and A Project of Shandong Province Higher Educational Science and Technology Program (J14LJ05).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shi, S., Hu, G., Zhang, X. et al. A study of the interaction between HIV-1 protease and C 2-symmetric inhibitors by computational methods. J Mol Model 20, 2369 (2014). https://doi.org/10.1007/s00894-014-2369-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-014-2369-3