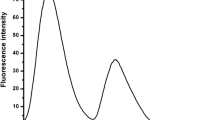
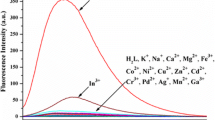
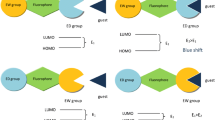
Abstract
Two BODIPY derivative sensors for metal ion recognition containing 10-(4-hydroxyphenyl) (L1) and 10-(3,4-dihydroxyphenyl) (L2) were synthesized in a one-pot reaction of benzaldehyde derivative and 2,4-dimethylpyrrole in the presence of trifluoroacetic acid as catalyst. The binding abilities between these sensors and 50 equivalents of Na+, K+, Ag+, Ca2+, Fe2+, Co2+, Ni2+, Cu2+, Zn2+, Cd2+, Pb2+, Al3+ and Cr3+ ions were studied using UV–vis and fluorescent spectroscopic methods. Of all the metal ions tested, Al3+ ion showed the greatest decrease in intensity in the spectra of the sensors, and therefore Al3+ ion forms the strongest complex. The binding abilities of BODIPY receptors with Na+, Ag+, Ca2+, Co2+, Ni2+, Cu2+, Zn2+ and Al3+ ions were also investigated using density functional theory (DFT) calculations at B3LYP/LanL2DZ theoretical level. The calculated results point to the same conclusion. DFT calculations also provided the HOMO–LUMO energy levels, which can explain the spectrum change upon complexation.

Graphical structure, fluorescent spectra, frontier orbital energy diagrams and electron-transfer paths in sensor L1, and after attachment with Al3+ ion.








Similar content being viewed by others
References
Demchenko AP (2009) Introduction to fluorescence sensing. Springer, Netherlands
Jeong Y, Yoon J (2012) Inorg Chim Acta 381:2–14
Kaur N, Kumar S (2011) Tetrahedron 67:9233–9264
Zhang JF, Zhou Y, Yoon J, Kim JS (2011) Chem Soc Rev 40:3416–3429
Zhang X, Shiraishi Y, Hirai T (2007) Tetrahedron Lett 48(31):5455–5459
Huo FJ, Zhang JJ, Yang YT, Chao JB, Yin CX, Zhang YB, Chen TG (2012) Sensors Actuators B Chem 166–167:44–49
Li CYL, Xu F, Li YF, Zhou K, Zhou Y (2012) Anal Chim Acta 717:122–126
Loudet A, Burgess K (2007) Chem Rev 107(11):4891–4932
Benstead M, Mehl GH, Boyle RW (2011) Tetrahedron 67(20):3573–3601
Suzuki S, Kozaki M, Nozaki K, Okada K (2011) J Photochem Photobiol C Photochem Rev 12(4):269–292
Boens N, Leen V, Dehaen W (2012) Chem Soc Rev 41:1130–1172
Ulrich G, Ziessel R, Harriman A (2008) Angew Chem Int Ed 47:1184–1201
Gimpl G, Gehrig-Burger K (2011) Steroids 76(3):216–231
Guzow K, Kornowska K, Wiczk W (2009) Tetrahedron Lett 50(24):2908–2910
Mankouri J, Tedbury PR, Gretton S, Hughes ME, Griffin SD, Dallas ML, Green KA, Hardie DG, Peers C, Harris M (2010) Proc Natl Acad Sci USA 107(25):11549–11554
Montejano HA, Amat-Guerri F, Costela A, García-Moreno I, Liras M, Sastre R (2006) J Photochem Photobiol A Chem 181(2–3):142–146
Badré S, Monnier V, Méallet-Renault R, Dumas-Verdes C, Schmidt EY, Mikhaleva AI, Laurent G, Levi G, Ibanez A, Trofimov BA, Pansu RB (2006) J Photochem Photobiol A Chem 183(3):238–246
Yin S, Yuan W, Huang J, Xie D, Liu B, Jiang K, Qiu H (2012) Spectrochim Acta A 96:82–88
Kubota Y, Uehara J, Funabiki K, Ebihara M, Matsui M (2010) Tetrahedron Lett 51:6195–6198
Lu H, Zhang SS, Liu HZ, Wang YW, Shen Z, Liu CG, You XZ (2009) J Phys Chem A 113(51):14081–14086
Valeura B, Leraya I (2000) Coord Chem Rev 205(1):3–40
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Hay PJ, Wadt WR (1985) J Chem Phys 82:270–283
Wadt WR, Hay PJ (1985) J Chem Phys 82:284–298
Hay PJ, Wadt WR (1985) J Chem Phys 82:299–310
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2008) Gaussian 03, Revision E01. Gaussian Inc, Wallingford, CT
Baruah M, Qin W, Basarić N, De Borggraeve WM, Boens N (2005) J Org Chem 70(10):4152–4157
Kennedy DP, Kormos CM, Burdette SC (2009) J Am Chem Soc 131:8578–8586
Gabe Y, Urano Y, Kikuchi K, Kojima H, Nagano T (2004) J Am Chem Soc 126:3357–3367
Qin W, Baruah M, Van der Auweraer M, Frans C, De Schryver FC, Boens N (2005) J Phys Chem A 109:7371–7384
Kollmannsberger M, Rurack K, Resch-Genger U, Daub JJ (1998) Phys Chem A 102:10211–10220
Ho T-L (1975) Chem Rev 75:1–20
Miessler GL, Tarr DA (2004) Inorganic Chemistry, 3rd edn. Pearson Prentice Hall, New Jersey
Chiodo S, Russo N, Sicilia E (2006) J Chem Phys 125:104107–1–104107–8
Strassner T, Taige MA (2005) J Chem Theory Comput 1:848–855
Ruangpornvisuti V (2007) Struct Chem 18:977–984
Remacle F, Grandjean F, Long GJ (2008) Inorg Chem 47:4005–4014
Weller A (1968) Pure Appl Chem 16:115–124
Acknowledgments
The authors gratefully acknowledge the faculty of science, Mahasarakham University and the Center of Excellence for Innovation in Chemistry (PERCH-CIC), Thailand for financial support of this research. Profound gratitude is also extended to the facility provided by Supramolecular Chemistry Research Unit and Department of Chemistry, Faculty of Science, Mahasarakham University, Maha Sarakham, Thailand.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 458 kb)
Rights and permissions
About this article
Cite this article
Keawwangchai, T., Morakot, N. & Wanno, B. Fluorescent sensors based on BODIPY derivatives for aluminium ion recognition: an experimental and theoretical study. J Mol Model 19, 1435–1444 (2013). https://doi.org/10.1007/s00894-012-1698-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-012-1698-3