Abstract
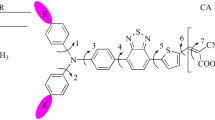
The geometries have been optimized by using density functional theory. The highest occupied molecular orbitals are delocalized on triphenylamine moiety while lowest unoccupied molecular orbital are localized on anchoring group. Intramolecular charge transfer has been observed from highest occupied molecular orbitals to lowest unoccupied molecular orbital. By replacing the vinyl hydrogens with methoxy as well as one benzene ring as bridge leads to a raised energy gap while extending the bridge decreases the energy gap compared to parent molecule. The HOMO energies bump up by extending the bridge. The LUMO energies of all the investigated dyes are above the conduction band of TiO2 and HOMOs are below the redox couple except 3c. The distortion between anchoring group and triphenylamine can hamper the recombination reaction.

Triphenylamine based dyes



Similar content being viewed by others
References
Regan BO, Grätzel M (1991) A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 353:737–740
Nazeeruddin MK et al (1993) Conversion of light to electricity by cis-X2bis(2,2′-bipyridyl-4,4′-dicarboxylate)ruthenium(II) chargetransfer sensitizers (X = Cl–, Br–, I–, CN– and SCN–) on nanocrystalline titanium dioxide electrodes. J Am Chem Soc 115:6382–6390
Chen J, Too CO, Burrel AK, Collis GE, Officer DL, Wallace GG (2003) Photovoltaic devices based on poly(bisterthiophenes) and substituted poly(bisterthiophene). Synth Met 137:1373–1374
Smestad G, Bignozzi C, Argazzi R (1994) Testing of dye sensitized TiO2 solar cells I: Experimental photocurrent output and conversion efficiencies. Sol Energy Mater Sol Cells 32:259–272
Hara K, Sayama K, Ohga Y, Shinpo A, Suga S, Arakawa H (2001) A coumarin-derivative dye sensitized nanocrystalline TiO2 solar cell having a high solar-energy conversion efficiency up to 5.6%. Chem Commun 6:569–570
Gazotti WA, Girotto EM, Nogueira AF, Paoli MA (2001) Solid-state photoelectrochemical cell using a polythiophene derivative as photoactive electrode. Sol Energy Mater Sol Cells 69:315–323
Nazeeruddin MK, Pechy P, Renouard T, Zakeeruddin SM, Humphry-Baker R, Comte P, Liska P, Cevey L, Costa E, Shklover V, Spiccia L, Deacon GB, Bignozzi CA, Grätzel M (2001) Engineering of efficient panchromatic sensitizers for nanocrystalline TiO2-based solar cells. J Am Chem Soc 123:1613–1624
Cao F, Oskam G, Searson PC (1995) A solid state, dye sensitized photoelectrochemical cell. J Phys Chem 99:17071–17073
Matsumoto M, Miyazaki H, Matsuhiro K, Kumashiro Y, Takaoka Y (1996) A dye sensitized TiO2 photoelectrochemical cell constructed with polymer solid electrolyte. Solid State Ionics 89:263–267
Coakely KM, McGehee MD (2003) Photovoltaic cells made from conjugated polymers infiltrated into mesoporous titania. Appl Phys Lett 83:3380–3382
Hara K, Kurashige M, Ito S, Shinpo A, Suga S, Sayama K, Arakawa H (2003) Novel polyene dyes for highly efficient dye-sensitized solar cells. Chem Commun 2:252–253
Liu D, Fessenden RW, Hug GL, Kamat PV (1997) Dye capped semiconductor nanoclusters. Role of back electron transfer in the photosensitization of SnO2 nanocrystallites with cresyl violet aggregates. J Phys Chem B 101:2583–2590
Burfeindt B, Hannappel T, Storck W, Willig F (1996) Measurement of temperature-independent femtosecond interfacial electron transfer from an anchored molecular electron donor to a semiconductor as acceptor. J Phys Chem 100:16463–16465
Sayama K, Tsukagochi S, Hara K, Ohga Y, Shinpou A, Abe Y, Suga S, Arakawa H (2002) Photoelectrochemical properties of J aggregates of benzothiazole merocyanine dyes on a nanostructured TiO2 film. J Phys Chem B 106:1363–1371
Hagfeldt A, Gratzel M (2000) Molecular photovoltaics. Acc Chem Res 33:269–277
Ning Z, Zhang Q, Wu W, Pei H, Liu B, Tian H (2008) Starburst triarylamine based dyes for efficient dye-sensitized solar cells. J Org Chem 73:3791–3797
Xu W, Peng B, Chen J, Liang M, Cai F (2008) New triphenylamine-based dyes for dye-sensitized solar cells. J Phys Chem C 112:874–880
Irfan A, Al-Sehemi AG, Jin RF (2012) Effect of the donor group on the electronic properties of triphenylamine sensitizers. Submitted
Frisch MJ, Trucks GW, Schlegel HB et al (2009) Gaussian 09, Revision A.1. Gaussian Inc, Wallingford, CT
Politzer P, Abu-Awwad F (1998) Some approximate Kohn—Sham molecular energy formulas. Mol Phys 95:681–688
Abu-Awwad F, Politzer P (2000) Variation of parameters in Becke-3 hybrid exchange-correlation functional. J Comput Chem 21:227–238
Preat J, Jacquemi D, Perpete EA (2010) Design of new triphenylamine-sensitized solar cells: a theoretical approach. Environ Sci Technol 44:5666–5671
Preat J, Michaux C, Jacquemin D, Perpète EA (2009) Enhanced efficiency of organic dye-sensitized solar cells: Triphenylamine derivatives. J Phys Chem C 113:16821–16833
Becke AD (1993) A new mixing of Hartree–Fock and local density–functional theories. J Chem Phys 98:1372–1377
Becke AD (1988) Density-functional exchange-energy approximation with correct asymptotic behavior. Phys Rev A 38:3098–3100
Lee C, Yang W, Parr RG (1988) Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. J Chem Phys 72:650–655
Irfan A, Al-Sehemi AG (2012) Quantum chemical investigations of electron injection in triphenylamine based sensitizers for dye sensitized solar cells. Submitted
De Oliveira MA, Duarte HA, Pernaut J-M, De Almeida WB (2000) Energy gaps of α, α‘-substituted oligothiophenes from semiempirical, ab initio, and density functional methods. J Phys Chem A 104:8256–8262
Aihara J-i (1999) Reduced HOMO − LUMO gap as an index of kinetic stability for polycyclic aromatic hydrocarbons. J Phys Chem A 103:7487–7495
Kaur I, Jia W, Kopreski RP, Selvarasah S, Dokmeci MR, Pramanik C, McGruer NE, Miller GP (2008) Substituent effects in pentacenes: Gaining control over HOMO − LUMO gaps and photooxidative resistances. J Am Chem Soc 130:16274–16286
do Couto PC, Guedes RC, Cabral BJC (2004) The density of states and band gap of liquid water by sequential Monte Carlo/Quantum mechanics calculations. Braz J Phys 34:42–47
Irfan A, Cui RH, Zhang JP, Hao LZ (2009) Push–pull effect on the charge transfer, and tuning of emitting color for disubstituted derivatives of mer-Alq3. Chem Phys 364:39–45
Balanay MP, Kim DH (2008) DFT/TD-DFT molecular design of porphyrin analogues for use in dye-sensitized solar cells. Phys Chem Chem Phys 10:5121–5127
De Angelis F, Fantacci S, Selloni A (2008) Alignment of the dye's molecular levels with the TiO2 band edges in dye-sensitized solar cells: a DFT–TDDFT study. Nanotechnology 19:424002–424008
Ito S, Zakeeruddin SM, Humphry-Baker R, Liska P, Charvet R, Comte P, MdK N, Pechy P, Takata M, Miura H, Uchida S, Gratzel M (2006) High-efficiency organic-dye-sensitized solar cells controlled by nanocrystalline-TiO2 electrode thickness. Adv Mater 18:1202–1205
Acknowledgments
Authors are thankful to the King Khalid University support and their facilities to carry out the computational work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Irfan, A., Al-Sehemi, A.G. & Asiri, A.M. Donor-enhanced bridge effect on the electronic properties of triphenylamine based dyes: density functional theory investigations. J Mol Model 18, 3609–3615 (2012). https://doi.org/10.1007/s00894-012-1372-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-012-1372-9