Abstract
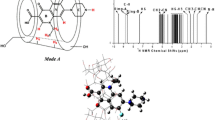
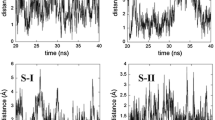
Acetic acid acts as one component of the mobile phase to influence separation of puerarin from daidzin when using β-cyclodextrin-substituted media. In this work considering an explicit acetic acid solution, host-guest complexes of β-cyclodextrin (β-CD) with puerarin and daidzin were investigated by molecular dynamics simulations. Computational results indicate different shuttle motions of puerarin and daidzin inside the cavity of β-CD. A model detailing the shuttle motion was constructed, and the relationships between shuttle depth and guest rotation angles, hydrogen bonds, and host-guest interaction energies were analyzed. The results can be used to explain the chromatographic retention mechanisms of puerarin and daidzin with β-CD, and to explore the complexity of host-guest interactions involving β-CD.







Similar content being viewed by others
References
Miro G, Rondinone A, Nappi A, Ungaro F, Quaglia F, La Rotonda MI (2009) Modulation of release rate and barrier transport of diclofenac incorporated in hydrophilic matrices: role of cyclodextrins and implications in oral drug delivery. Eur J Pharm Biopharm 72:76–82
Thi TD, Nauwelaerts K, Froeyen M, Baudemprez L, van Speybroeck M, Augustijns P, Annaert P, Martens J, van Humbeeck J, van den Mooter G (2010) Comparison of the complexation between methylprednisolone and different cyclodextrins in solution by 1H-NMR and molecular modeling studies. J Pharm Sci 99:3863–3873
Lu Z, Cheng B, Hu YL, Zhang YH, Zou GL (2009) Complexation of resveratrol with cyclodextrins: solubility and antioxidant activity. Food Chem 113:17–20
Koontz JL, Marcy JE, O’Keefe SF, Duncan SE (2009) Cyclodextrin inclusion complex formation and solid-state characterization of the natural antioxidants α-tocopherol and quercetin. J Agric Food Chem 57:1162–1171
van de Manakker F, Vermonden D, Nostrum CF, Hennink WE (2009) Cyclodextrin-based polymeric materials: synthesis, properties, and pharmaceutical/biomedical applications. Biomacromolecules 10:3157–3175
Xu MY, Wu SZ, Zeng F, Yu CM (2010) Cyclodextrin supramolecular complex as a water-soluble ratiometric sensor for ferric ion sensing. Langmuir 26:4529–4534
Si-Ahmed K, Tazerouti F, Badjah-Hadj-Ahmed AY, Aturki Z, D’Orazio G, Roccoa A, Fanali S (2010) Optical isomer separation of flavanones and flavanone glycosides by nano-liquid chromatography using a phenyl-carbamate-propyl-β-cyclodextrin chiral stationary phase. J Chromatogr A 1217:1175–1182
Crini G, Morcellet M (2002) Synthesis and applications of adsorbents containing cyclodextrins. J Sep Sci 25:789–813
Jullian C, Miranda S, Zapata-Torres G, Mendizábal F, Olea-Azar C (2007) Studies of inclusion complexes of natural and modified cyclodextrin with (+)catechin by NMR and molecular modeling. Bioorg Med Chem 15:3217–3224
Wang MY, Kan WN, He XL, Tan TW, Janson JC (2005) Preparative purification of puerarin from pueraria flavones by oligo-β-cyclodextrin-Sepharose HP matrix. J Liq Chromatogr Relat Technol 28:1509–1518
He XL, Tan TW, Xu BZ, Janson JC (2004) Separation and purification of puerarin using β-cyclodextrin-coupled agarose gel media. J Chromatogr A 1022:77–82
Li R, Zhao R, Zhang HY, Li C, Feng D, Qin PY, Tan TW (2010) A novel medium poly(vinyl acetate-triallyl isocyanurate-divinylbenzene) coupled with oligo-β-cyclodextrin for the isolation of puerarin from pueraria flavones. Chromatographia 72:47–54
Yang L, Zhu Y, Tan TW, Janson JC (2007) Coupling oligo-β-cyclodextrin on polyacrylate beads media for separation of puerarin. Process Biochem 42:1075–1083
Lipkowitz KB (1998) Applications of computational chemistry to the study of cyclodextrins. Chem Rev 98:829–1874
Sun M, Liu XH, Yan LS, Luo GA, Zhao YF (2003) Molecular recognition between 4aS/R-galanthamine diastereoisomers and α-cyclodextrin. J Mol Model 9:419–422
Dodziuk H (2006) Cyclodextrins and their complexes. Wiley-VCH, Weinheim
Sellner B, Zifferer G, Kornherr A, Krois D, Brinker UH (2008) Molecular dynamics simulations of β-cyclodextrin–ziadamantane complexes in water. J Phys Chem B 112:710–714
EI-Barghouthi MI, Jaime C, AI-sakhen NA, Issa AA, Abdoh AA, Ai Omari MM, Badwan AA, Zughul MB (2008) Molecular dynamics simulations and MM–PBSA calculations of the cyclodextrin inclusion complexes with 1-alkanols, para-substituted phenols and substituted imidazoles. J Mol Struct THEOCHEM 853:45–52
Figueiras A, Sarraguca JM, Pais AA, Carvalho RA, Veiga JF (2010) The role of l-arginine in inclusion complexes of omeprazole with cyclodextrins. AAPS PharmSciTech 11:233–240
Kokkinou A, Tsorteki F, Karpusas M, Papakyriakou A, Bethanis K, Mentzafos D (2010) Study of the inclusion of the (R)- and (S)-camphor enantiomers in α-cyclodextrin by X-ray crystallography and molecular dynamics. Carbohydr Res 345:1034–1040
Zhang HY, Feng W, Li C, Tan TW (2010) Investigation of the inclusions of puerarin and daidzin with β-cyclodextrin by molecular dynamics simulation. J Phys Chem B 114:4876–4883
van der Spoel D, Lindahl E, Hess B, Buuren AR, Apol E, Meulenhoff PJ, Tieleman DP, Sijbers AL, Feenstra KA, Drunen R, Berendsen HJ (2005) Gromacs user manual (version 3.3). http://www.gromacs.org/documentation/manual
van der Spoel D, Lindahl E, Hess B, Groenhof G, Mark AF, Berendsen HJC (2005) GROMACS: fast, flexible, and free. J Comput Chem 26:1701–1708
Schuettelkopf AW, van Aalten DMF (2004) PRODRG—a tool for high-throughput crystallography of protein–ligand complexes. Acta Crystallogr D60:1355–1363
Kollman PA, Massova I, Reyes C, Kuhn B, Huo SH, Chong L, Lee M, Lee T, Duan Y, Wang W, Donini O, Cieplak P, Srinivasan J, Case DA, Cheatham TS (2000) Calculating structures and free energies of complex molecules: combining molecular mechanics and continuum models. Acc Chem Res 33:889–897
Raffaini G, Ganazzoli F, Malpezzi L, Fuganti C, Fronza G, Panzeri W, Mele A (2009) Validating a strategy for molecular dynamics simulations of cyclodextrin inclusion complexes through single-crystal X-ray and NMR experimental data: a case study. J Phys Chem B 113:9110–9122
Acknowledgments
This research was supported by the National Natural Science Foundation of China (20876011), 863 program (2006AA02Z245, 2007AA100404), 973 program (2007CB714304), Beijing Municipal Science & Technology Commission (Z09010300840901), and special funds of cooperation project of Beijing Municipal Education Commission.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, H., Feng, W., Li, C. et al. A model for the shuttle motions of puerarin and daidzin inside the cavity of β-cyclodextrin in aqueous acetic acid: insights from molecular dynamics simulations. J Mol Model 18, 221–227 (2012). https://doi.org/10.1007/s00894-011-1036-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-011-1036-1