Abstract
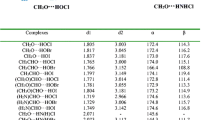
The effect of alkyl substitution (CH3, C2H5, n-C3H7, i-C3H7, and t-C4H9) on the hydrogen bond strengths (H-bond) of substituted amide-alcohol complexes has been systematically explored. B3LYP/aug-cc-pVDZ method was applied to a total of 215 alkyl substituted amide-alcohol complexes to delineate the effect of substitution on the H-bond strength; formamide-water complex is taken as reference point. Complexes are classified into five types depending on the hydrogen donor, acceptor and the site of alkyl substitution (Type-IA, Type-IIA, Type-IB, Type-IIB and Type-III). The strength of H-bond was correlated with geometrical parameters such as proton-acceptor (H∙∙∙∙Y) distance, the length of proton donating bond (X–H). In all the complexes N–H and O–H stretching frequencies are red-shifted. The effect of alkyl substitution on N–H and O–H stretching frequencies were analyzed. Topological parameters like electron density at H∙∙∙∙Y and X–H bond critical points as derived from atom in molecules (AIM) theory was also evaluated. When C = O group is participating in H-bond, the strength of H-bond decreases with increasing size of alcohols except for methanol (Type-IA, Type-III and Type-IB complexes). But it increases with increasing size of alkyl groups on amide and decreases with bulky groups. In the case of N–H group as H-bond donor, the strength of H-bond increases with increasing size of alcohols (Type-IIA and Type-IIB complexes) whereas decreases with increasing size of alkyl groups on amide. Type-IA, IIA, IB and IIB complexes exhibit good correlations among IE, H-bond distance and electron density at bcp. In Type-III complexes, average H-bond distance and sum of electron densities shows better correlation with IEs than the corresponding individuals. The correlation of IE less with electron density at RCP compared to sum of electron densities.

Effect of alkyl substitution on H-bond strengths of substituted amide-alcohol complexes









Similar content being viewed by others
References
Jeffrey GA, Saenger W (1991) Hydrogen Bonding in Biological Structures. Springer, New York
Deechongkit S, Dawson PE, Kelly JW (2004) Toward Assessing the Position-Dependent Contributions of Backbone Hydrogen Bonding to β-Sheet Folding Thermodynamics Employing Amide-to-Ester Perturbations. J Am Chem Soc 126:16762–16771
Noveron JC, Lah MS, Del Sesto RE, Arif AM, Miller JS, Stang PJ (2002) Engineering the Structure and Magnetic Properties of Crystalline Solids via the Metal-Directed Self-Assembly of a Versatile Molecular Building Unit. J Am Chem Soc 124:6613–6625
Stockton WB, Rubner MF (1997) Molecular-Level Processing of Conjugated Polymers. 4. Layer-by-Layer Manipulation of Polyaniline via Hydrogen-Bonding Interactions. Macromolecules 30:2717–2725
Vijay D, Zipse H, Sastry GN (2008) On the Cooperativity of Cation−π and Hydrogen Bonding Interactions. J Phys Chem B 112:8863–8867
Sunita SS, Rohini NK, Kulkarni MG, Nagaraju M, Sastry GN (2007) Hydrogen Bonding in Trivinyl Monomers: Implications for Inclusion Complexation and Polymerization. Macromolecules 40:1824–1830
Greenberg A, Breneman CM, Liebman JF (2002) The amide linkage: Structural significance in chemistry, biochemistry and materials science. Wiley, New York
Boyd DB (1993) Application of the hypersurface iterative projection method to bicyclic pyrazolidinone antibacterial agents. J Med Chem 36:1443–1449
Jungheim LN, Boyd DB, Indelicato JM, Pasini CE, Preston DE, Alborn WE (1991) Synthesis, hydrolysis rates, supercomputer modeling, and antibacterial activity of bicyclic tetrahydropyridazinones. J Med Chem 34:1732–1739
Michael MN (2005) The Ionic Hydrogen Bond. Chem Rev 105:213–284
Custelcean R, Jackson JE (2001) Dihydrogen Bonding: Structures, Energetics, and Dynamics. Chem Rev 101:1963–1980
Hobza P, Havlas Z (2000) Blue-Shifting Hydrogen Bonds. Chem Rev 100:4253–4264
Alkorta I, Rozas I, Elguero J (1998) Non-conventional hydrogen bonds. Chem Soc Rev 27:163–170
Bryantsev VS, Hay BP (2005) Are C−H Groups Significant Hydrogen Bonding Sites in Anion Receptors? Benzene Complexes with Cl-, NO -3 , and ClO -4 . J Am Chem Soc 127:8282–8283
Wilkens SJ, Westler WM, Weinhold F, Markley JL (2002) Trans-Hydrogen-Bond h2 J NN and h1 J NH Couplings in the DNA A−T Base Pair: Natural Bond Orbital Analysis. J Am Chem Soc 124:1190–1191
Del Bene JE, Elguero J (2004) One-Bond Spin−Spin Coupling Constants of X−1H Proton Donors in Complexes with X−H−Y Hydrogen Bonds, for X = 13C, 15N, 17O, and 19F: Predictions, Comparisons, and Relationships among 1 J X-H, 1 K X-H, and X−H Distances. J Am Chem Soc 126:15624–15631
Alonso JL, Antolinez S, Blanco S, Lesarri A, Lopez JC, Caminati W (2004) Weak C−H···O and C−H···F−C Hydrogen Bonds in the Oxirane−Trifluoromethane Dimer. J Am Chem Soc 126:3244–3249
Thureau P, Ancian B, Viel S, Thevand A (2006) NMR diffusion and nuclear Overhauser investigation of the hydration properties of thymine: influence of the methyl group. Chem Commun 1884-1886
Dai Q, Xu CY, Sato Y, Yoshimoto K, Nishizawa S, Teramae N (2006) Enhancement of the binding ability of a ligand for nucleobase recognition by introducing a methyl group. Anal Sci 22:201–203
Chenoweth K, Dykstra CE (2005) Selecting weak bonding sites: effect of a methyl group in the acetylene–polyyne interaction. Chem Phys Lett 402:329–334
Choi KH, Lee HJ, Karpfen A, Yoon CJ, Park J, Choi YS (2001) Hydrogen-bonding interaction of methyl-substituted pyridines with thioacetamide: steric hindrance of methyl group. Chem Phys Lett 345:338–344
Ramos MN, Lopes KC, Silva WLV, Tavares AM, Castriani FA, do Monte SA, Ventura E, Araujo RCMU (2006) An ab initio study of the C2H2−HF, C2H(CH3)−HF and C2(CH3)2−HF hydrogen-bonded complexes. Spectrochim Acta A 63:383–390
Papai I, Jancso G (2000) Hydrogen Bonding in Methyl-Substituted Pyridine−Water Complexes: A Theoretical Study. J Phys Chem A 104:2132–2137
Qingzhong Li, Guoshi Wu, Zhiwu Yu (2006) The Role of Methyl Groups in the Formation of Hydrogen Bond in DMSO−Methanol Mixtures. J Am Chem Soc 128:1438–1439
Qingzhong Li, Xiulin An, Luan F, Wenzuo Li, Gong B, Cheng J (2008) Regulating Function of Methyl Group in Strength of CH···O Hydrogen Bond: A High-Level Ab Initio Study. J Phys Chem A 112:3985–3990
Lovas FJ, Suenram RD, Fraser GT (1988) The microwave spectrum of formamide–water and formamide–methanol complexes. J Chem Phys 88:722–729
Jasien PG, Stevens WJ (1986) Ab initio study of the hydrogen bonding interactions of formamide with water and methanol. J Chem Phys 84:3271–3277
Coitino EL, Irving K (1990) Theoretical studies of hydrogen-bonded complexes using semiempirical methods. J Mol Struct THEOCHEM 210:405–426
Engdahl A, Nelander B (1993) Complex formation between water and formamide. J Chem Phys 99:4894–4907
Sim F, Amant StA, Papai I, Salahub DR (1992) Gaussian density functional calculations on hydrogen-bonded systems. J Am Chem Soc 114:4391–4400
Besley NA, Hirst JD (1999) Ab Initio Study of the Electronic Spectrum of Formamide with Explicit Solvent. J Am Chem Soc 121:8559–8566
Dannenberg JJ (2006) Enthalpies of hydration of N-Methylacetamide by one, two, and three waters and the effect upon the C = O stretching frequency. An Ab initio DFT study. J Phys Chem A 110:5798–5802
Pacios LF (2006) Computational study of the process of hydrogen bond breaking: The case of the formamide–formic acid complex. J Comput Chem 27:1641–1649
Moisen S, Dannenberg JJ (2003) Molecular Orbital Calculations on the Protonation of Hydrogen-Bonded Formamide Chains. Implications for Peptides. J Phys Chem B 107:12842–12846
Fu AP, Du DM, Zhou ZY (2003) Density functional theory study of the hydrogen bonding interaction of 1:1 complexes of formamide with methanol. Chem Phys Lett 377:537–543
Fu AP, Du DM, Zhou Z (2003) Density functional theory study of the hydrogen bonding interaction of 1:1 complexes of formamide with water. J Mol Struct THEOCHEM 623:315–325
Bende A, Suhai S (2005) BSSE-corrected geometry and harmonic and anharmonic vibrational frequencies of formamide–water and formamide–formamide dimers. Int J Quantum Chem 103:841–853
Parreira RLT, Valdes H, Galembeck SE (2006) Computational study of formamide–water complexes using the SAPT and AIM methods. Chem Phys 331:96–110
Rubicelia V, Jorge G, Richard AF, Harry S, Benjamin PH, David AD (2001) Strength of the N−H···O = C and C−H···O = C Bonds in Formamide and N-Methylacetamide Dimers. J Phys Chem A 105:4963–4968
Lu JF, Zhou ZY, Zhao G, Wu QY (2005) Density functional theory study of the hydrogen bonding interaction in formamide dimer. J Mol Struct THEOCHEM 724:107–114
Nagaraju M, Sastry GN (2010) Comparative study on formamide–water complex. Int J Quantum Chem 110:1994–2003
Frisch MJ et al (2003) Gaussian 03, Revision B.04. Gaussian Inc, Wallingford, CT
Boys SF, Bernardi F (1970) The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol Phys 19:553–566
Bader RFW (1990) Atoms in Molecules - A Quantum Theory. Oxford University Presss, Oxford, UK
Popelier P (2000) Atoms in Molecules: An Intriduction. Printice Hall, New York
Urban JJ, Tillman GB, Cronin AW (2006) Fluoroolefins as Peptide Mimetics: A Computational Study of Structure, Charge Distribution, Hydration, and Hydrogen Bonding. J Phys Chem A 110:11120–11129
Langley CH, Allinger NL (2003) Molecular Mechanics (MM4) and ab Initio Study of Amide−Amide and Amide−Water Dimers. J Phys Chem A 107:5208–5216
Vijay D, Sastry GN (2008) Exploring the size dependence of cyclic and acyclic π –systems on cation-π binding. Phys Chem Chem Phys 582-590
Reddy AS, Vijay D, Sastry GM, Sastry GN (2006) From subtle to substantial: Role of metal ions on π-π interactions. J Phys Chem B 110:2479–2481
Rao JS, Dinadayalane TC, Sastry GN, Leszczynski J (2008) A comprehensive study on the solvation of mono and di valent metal ions: Li+,Na+,K+,Be2+,Mg2+,and Ca2+. J Phys Chem A 112:12944–12953
Rao JS, Zipse H, Sastry GN (2009) Explicit solvent effect on Cation-π interactions: A first principle investigation. J Phys Chem 113:7225–7236
Reddy AS, Zipse H, Sastry GN (2007) Cation-π interactions of bare and coordinately saturated metal ions: Contrasting structural and energetic characteristics. J Phys Chem B 111:1546–11553
Alkorta I, Elguero J, Foces-Foces C (1996) Dihydrogen bonds (A–H∙∙∙∙∙H–B). Chem Commun 1633-1634
González L, Mó O, Yáňez M (1997) High-Level ab Initio Calculations on the Intramolecular Hydrogen Bond in Thiomalonaldehyde. J Phys Chem A 101:9710–9719
Espinosa E, Souhassou M, Lachekar H, Lecomte C (1999) Topological analysis of the electron density in hydrogen bonds. Acta Crystallogr B 55:563–572
Grabowski SJ (2000) High-Level Ab Initio Calculations of Dihydrogen-Bonded Complexes. J Phys Chem A 104:5551–5557
Quiňonero D, Frontera A, Ballester P, Garau C, Costa A, Deyá PM (2001) A topological analysis of charge density in complexes between derivatives of squaric acid and ammonium cation. Chem Phys Lett 339:369–374
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagaraju, M., Narahari Sastry, G. Effect of alkyl substitution on H-bond strength of substituted amide-alcohol complexes. J Mol Model 17, 1801–1816 (2011). https://doi.org/10.1007/s00894-010-0886-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-010-0886-2