Abstract
Molecular recognition between 4aS/R-galanthamine diastereoisomers (1: 4aS-galanthamine; 2: 4aR-galanthamine) and α-cyclodextrin (α-CD) were studied by use of docking and molecular dynamics (MD) simulation approaches. The binding energy of constructed 2···α-CD complexes is ~17 kcal mol−1 lower than that of 1···α-CD, implying a stronger binding ability of 2 with α-CD than that of 1. The theoretical modeling result is consistent with our previous CZE result, which demonstrated that α-CD is an efficient chiral additive for separating 1 and 2. The modeling result also indicates that both hydrophobic interaction and H-bond force may work as major factors for molecular recognition between the galanthamine diastereoisomers and α-CD.
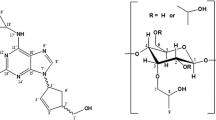
Figure Chemical structures of 4aS-galanthamine (left) and 4aR-galanthamine (right)




Similar content being viewed by others
Abbreviations
- Galanthamine:
-
4aS,6R,8aS-4a,5,9,10,11,12-Hexahydroxy-3-methoxy-11-methyl-6H-benzofuro[3a,3,2-e,f]benzazepin-6-ol
References
Lyseng-Williamson KA, Plosker GL (2002) Pharmacoeconomics 20:919-942
Harvey AL (1995) Pharmacol Ther 68:113–128
Raskind MA, Peskind ER, Wessel T, Yuan W (2000) Neurology 54:2261–2268
Tariot PN, Solomon PR, Morris JC, Kershaw P, Lilienfeld S, Ding C (2000) Neurology 54:2269–2276
Kuenburg B, Czollner L, Frohlic J, Jordis U (1999) Org Process Res Dev 3:425–431
Guillou C, Beunard JL, Gras E, Thal C (2001) Angew Chem Int Ed 40:4745–4746
Trost BM, Tang W (2002) Angew Chem Int Ed 41:2795–2797
Node M, Kodama S, Hamashima Y, Baba T, Hamamichi N, Nishide K (2001) Angew Chem Int Ed 40:3060–3061
Ingkaninan K, de Best CM, van der Heijden R, Hofte AJ, Karabatak B, Irth H, Tjaden UR, van der Greef J, Verpoorte (2000) J Chromatogr A 872:61–73
Yan LS (2003) PhD Thesis, Tsinghua University
Molecular Simulations Inc(MSI) (1997) InsightII 97, San Diego, Calif.
Molecular Simulations Inc(MSI) (1997) Discover_3 version 2.98, San Diego, Calif.
Dauber-Osguthorpe P, Roberts VA, Osguthorpe DJ, Wolff J, Genest M, Hagler AT (1988) Proteins Struct Funct Genet 4:31–47
Peeters OM, Blaton NM, De Ranter CJ (1997) Acta Crystallogr C 53:1284–1286
Bartolucci C, Perola E, Pilger C, Fels G, Lamba D (2001) Proteins Struct Funct Genet 42:182–191
Lindner K, Saenger W (1982) Acta Crystallogr B 38:203–205
Szejtli J (1998) Chem Rev 98:1743–1754
Molecular Simulations Inc(MSI) (1998) Affinity, San Diego, Calif.
Acknowledgements
This work was supported by the Chinese National Natural Science Foundation (No. 20132020 and No. 20175026), The Ministry of Science and Technology, The Chinese Ministry of Education and Tsinghua University, and The Tianjin Science Foundation (No. 023606511).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, M., Liu, X., Yan, L. et al. Molecular recognition between 4aS/R-galanthamine diastereoisomers and α-cyclodextrin. J Mol Model 9, 419–422 (2003). https://doi.org/10.1007/s00894-003-0162-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-003-0162-9