Abstract
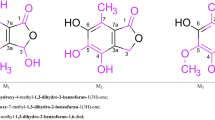
Ab initio calculations at the MP2/6-31G* level have shown that variously substituted di- and trifluorobenzenes form non-covalent complexes with benzene that adopt either aromatic–aromatic or H---F binding, the choice being determined by the pattern of fluorination. The binding energies of these structures are from 3.4 to 4.5 kcal mol−1. This range is large enough to account for observed variations in the binding affinity of a library of fluoroaromatic inhibitors of carbonic anhydrase. This enzyme has an aromatic amino acid at a central position in the active site. The diverse modes of binding of the dimers also suggest that aggregates of fluorobenzenes might adopt specified 3-dimensional shapes in the solid state.

Observed structure for 1,2-difluorobenzene


Similar content being viewed by others
References
Baldwin JJ, Ponticello GC, Anderson PS, Christy ME, Murko MA, Randall WC, Schwam H, Sugrue MF, Springer JP, Gautheron P, Grove J, Mallorga P, Viader MP, McKeever BM, Navia MA (1989) J Med Chem 32:2510–2513
(a) Doyon JB, Jain A (1999) Org Lett 1:183–185; (b) Doyon JB, Hansen EAM, Kim CY, Chang JS, Christianson DW, Madder RD, Voet JG, Baird Jr TA, Fierke CA, Jain A (2000) Org Lett 2:1189–1192
Kim CY, Chang JS, Doyon JB, Baird Jr TA, Fierke CA, Jain A, Christianson DW (2000) J Am Chem Soc 122:12125–12134
Madder RD, Kim CY, Chandra PP, Doyon JB, Baird Jr TA, Fierke CA, Christianson DW, Voet JG, Jain A (2002) J Org Chem 67:582–584
DerHovanessian A, Doyon JB, Jain A, Rablen PR, Sapse AM (1999) Org Lett 1:1359–1362
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JA, Stratman RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Baboul AG, Stefanov BB, Liu C, Liashenko A, Piskorz P, Komaromi, I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Gonzalez C, Challacombe M, Gill PMW, Johnson BG, Chen W, Wong MW, Andres JL, Gonzales C, Head-Gordon M, Replogle ES, Pople JA (1998) Gaussian 98, Revision A7. Gaussian, Pittsburgh, Pa.
West AP, Mecozzi S, Dougherty DA (1997) J Phys Org Chem 10:347–350
DerHovanessian A, Rablen PR, Jain A (2000) J Phys Chem A 104:6056–6061
(a) Schwenke DW, Truhlar DG (1985) J Chem Phys 82:2418–2426; (b) Szalewicz K, Cole SJ, Kolos W, Bartlett RJ (1988) J Chem Phys 89:3662–3672
Whitesides GM, Mathias JP, Seto CT (1991) Science 254:1312–1319
Thalladi VR, Weiss H-C, Blaser D, Boese R, Nangia A, Desiraju GR (1998) J Am Chem Soc 120:8702–8710
Acknowledgments
AJ thanks Swarthmore College, the Petroleum Research Fund, DuPont (Young Professor Award) and the Howard Hughes Medical Institute for support. AMS thanks the City University of New York for a PSC-CUNY award.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chandra, P.P., Jain, A. & Sapse, AM. An ab initio study of di- and trifluorobenzene–benzene complexes as relevant to carbonic anhydrase II–drug interactions. J Mol Model 10, 1–5 (2004). https://doi.org/10.1007/s00894-003-0146-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-003-0146-9