Abstract
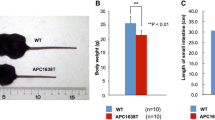
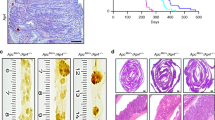
Adenomatous polyposis coli (APC), a well-known anti-oncogene, is considered to have multiple functions through its several binding domains. We have continuingly studied APC1638T/1638T mice (APC1638T mice) to elucidate the functions of APC other than tumor suppression. A distinctive feature of the APC1638T mice is they are tumor free and live as long as APC+/+ mice (WT mice). Previously, we found the length of crypt–villus axis in the jejunum was significantly elongated in APC1638T mice compared with that of WT mice. The populations of goblet cells, Paneth cells, and enteroendocrine cells were also disordered in APC1638T mice. Here, we further analyzed the intestinal dyshomeostasis in APC1638T mice, focusing on the proliferation and differentiation of intestinal stem cell (ISC) lineages, and apoptotic cell shedding at the villus tips. We found that the proliferation of ISC lineages was normally controlled; however, the shedding process of apoptosis cells was significantly delayed in the APC1638T mouse jejunum. Furthermore, the number of microfold cells (M cells) was significantly increased in the APC1638T mouse jejunum. Our data suggested both differentiation process of ISCs and turnover process of intestinal epithelia were disturbed in APC1638T mice, and that contributed to the villus elongation in the APC1638T mouse jejunum.








Similar content being viewed by others
References
Groden J, Thliveris A, Samowitz W, Carlson M, Gelbert L, Albertsen H, Joslyn G, Stevens J, Spirio L, Robertson M et al (1991) Identification and characterization of the familial adenomatous polyposis coli gene. Cell 66(3):589–600
Fodde R (2002) The APC gene in colorectal cancer. Eur J Cancer 38(7):867–871
Smith KJ, Johnson KA, Bryan TM, Hill DE, Markowitz S, Willson JK, Paraskeva C, Petersen GM, Hamilton SR, Vogelstein B et al (1993) The APC gene product in normal and tumor cells. Proc Natl Acad Sci USA 90(7):2846–2850
Senda T, Iizuka-Kogo A, Onouchi T, Shimomura A (2007) Adenomatous polyposis coli (APC) plays multiple roles in the intestinal and colorectal epithelia. Med Mol Morphol 40(2):68–81
Smits R, Kielman MF, Breukel C, Zurcher C, Neufeld K, Jagmohan-Changur S, Hofland N, van Dijk J, White R, Edelmann W, Kucherlapati R, Khan PM, Fodde R (1999) Apc1638T: a mouse model delineating critical domains of the adenomatous polyposis coli protein involved in tumorigenesis and development. Genes Dev 13(10):1309–1321
Yokoyama A, Nomura R, Kurosumi M, Shimomura A, Onouchi T, Iizuka-Kogo A, Smits R, Fodde R, Itoh M, Senda T (2012) Some fine-structural findings on the thyroid gland in Apc1638T/1638T mice that express a C-terminus lacking truncated Apc. Med Mol Morphol 45(3):161–167
Li C, Onouchi T, Hirayama M, Sakai K, Matsuda S, Yamada NO, Senda T (2020) Morphological and functional abnormalities of hippocampus in APC(1638T/1638T) mice. Med Mol Morphol (in press)
Onouchi T, Kobayashi K, Sakai K, Shimomura A, Smits R, Sumi-Ichinose C, Kurosumi M, Takao K, Nomura R, Iizuka-Kogo A, Suzuki H, Kondo K, Akiyama T, Miyakawa T, Fodde R, Senda T (2014) Targeted deletion of the C-terminus of the mouse adenomatous polyposis coli tumor suppressor results in neurologic phenotypes related to schizophrenia. Mol Brain 7:21
Yamada NO, Wenduerma MS, Senda T (2018) Validation and application of a novel APC antibody in western blotting, immunoprecipitation, and immunohistochemistry. Med Mol Morphol 51(4):227–236
Wang T, Onouchi T, Yamada NO, Matsuda S, Senda T (2017) A disturbance of intestinal epithelial cell population and kinetics in APC1638T mice. Med Mol Morphol 50(2):94–102
Marshman E, Booth C, Potten CS (2002) The intestinal epithelial stem cell. BioEssays 24(1):91–98
Gehart H, Clevers H (2019) Tales from the crypt: new insights into intestinal stem cells. Nat Rev Gastroenterol Hepatol 16(1):19–34
Vanuytsel T, Senger S, Fasano A (1830) Shea-Donohue T (2013) Major signaling pathways in intestinal stem cells. Biochim Biophys Acta 2:2410–2426
Barker N, van Es JH, Kuipers J, Kujala P, van den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H, Peters PJ, Clevers H (2007) Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449(7165):1003–1007
Barker N, van de Wetering M, Clevers H (2008) The intestinal stem cell. Genes Dev 22(14):1856–1864
Tian H, Biehs B, Warming S, Leong KG, Rangell L, Klein OD, de Sauvage FJ (2011) A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature 478(7368):255–259
Snippert HJ, van Es JH, van den Born M, Begthel H, Stange DE, Barker N, Clevers H (2009) Prominin-1/CD133 marks stem cells and early progenitors in mouse small intestine. Gastroenterology 136(7):2187-2194e2181
Fevr T, Robine S, Louvard D, Huelsken J (2007) Wnt/beta-catenin is essential for intestinal homeostasis and maintenance of intestinal stem cells. Mol Cell Biol 27(21):7551–7559
Kanaya T, Ohno H (2014) The mechanisms of M-cell differentiation. Biosci Microbiota Food Health 33(3):91–97
Mabbott NA, Donaldson DS, Ohno H, Williams IR, Mahajan A (2013) Microfold (M) cells: important immunosurveillance posts in the intestinal epithelium. Mucosal Immunol 6(4):666–677
van der Flier LG, Clevers H (2009) Stem cells, self-renewal, and differentiation in the intestinal epithelium. Annu Rev Physiol 71:241–260
Gebert A, Rothkötter HJ, Pabst R (1996) M cells in Peyer’s patches of the intestine. Int Rev Cytol 167:91–159
Jang MH, Kweon MN, Iwatani K, Yamamoto M, Terahara K, Sasakawa C, Suzuki T, Nochi T, Yokota Y, Rennert PD, Hiroi T, Tamagawa H, Iijima H, Kunisawa J, Yuki Y, Kiyono H (2004) Intestinal villous M cells: an antigen entry site in the mucosal epithelium. Proc Natl Acad Sci USA 101(16):6110–6115
Berlin P, Reiner J, Wobar J, Bannert K, Glass Ä, Walter M, Bastian M, Willenberg HS, Vollmar B, Klar E, Seidler U, Lamprecht G, Witte M (2019) Villus growth, increased intestinal epithelial sodium selectivity, and hyperaldosteronism are mechanisms of adaptation in a murine model of short bowel syndrome. Dig Dis Sci 64(5):1158–1170
Knoop KA, Kumar N, Butler BR, Sakthivel SK, Taylor RT, Nochi T, Akiba H, Yagita H, Kiyono H, Williams IR (2009) RANKL is necessary and sufficient to initiate development of antigen-sampling M cells in the intestinal epithelium. J Immunol 183(9):5738–5747
Kanaya T, Hase K, Takahashi D, Fukuda S, Hoshino K, Sasaki I, Hemmi H, Knoop KA, Kumar N, Sato M, Katsuno T, Yokosuka O, Toyooka K, Nakai K, Sakamoto A, Kitahara Y, Jinnohara T, McSorley SJ, Kaisho T, Williams IR, Ohno H (2012) The Ets transcription factor Spi-B is essential for the differentiation of intestinal microfold cells. Nat Immunol 13(8):729–736
de Lau W, Kujala P, Schneeberger K, Middendorp S, Li VS, Barker N, Martens A, Hofhuis F, DeKoter RP, Peters PJ, Nieuwenhuis E, Clevers H (2012) Peyer’s patch M cells derived from Lgr5(+) stem cells require SpiB and are induced by RankL in cultured “miniguts.” Mol Cell Biol 32(18):3639–3647
Sato S, Kaneto S, Shibata N, Takahashi Y, Okura H, Yuki Y, Kunisawa J, Kiyono H (2013) Transcription factor Spi-B-dependent and -independent pathways for the development of Peyer’s patch M cells. Mucosal Immunol 6(4):838–846
Fouquet S, Lugo-Martínez VH, Faussat AM, Renaud F, Cardot P, Chambaz J, Pinçon-Raymond M, Thenet S (2004) Early loss of E-cadherin from cell-cell contacts is involved in the onset of anoikis in enterocytes. J Biol Chem 279(41):43061–43069
Miyashiro I, Senda T, Matsumine A, Baeg GH, Kuroda T, Shimano T, Miura S, Noda T, Kobayashi S, Monden M et al (1995) Subcellular localization of the APC protein: immunoelectron microscopic study of the association of the APC protein with catenin. Oncogene 11(1):89–96
Funding
This work was supported by JSPS KAKENHI, Grant Number JP18K06827 to Senda T.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
All of the experiments using animals were conducted according to the regulations set by the Committee for Ethics in Animal Experimentation at Gifu University, which are in accordance with the “principles of laboratory animal care” (NIH publication no. 86-23, revised 1985).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wenduerma, Yamada, N.O., Wang, T. et al. A further study on a disturbance of intestinal epithelial cell population and kinetics in APC1638T mice. Med Mol Morphol 54, 203–215 (2021). https://doi.org/10.1007/s00795-020-00279-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00795-020-00279-x