Abstract
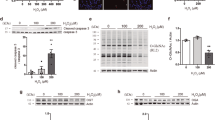
This study aimed to investigate the effects of H2O2-induced oxidative stress on cell viability and survival, as well as changes in the distribution of Golgi apparatus and in the level of Golgi reassembly and stacking protein 65 (GRASP65). Cell viability of cultured N2a cells treated with H2O2 was measured by the MTT assay. Apoptosis was measured by flow cytometry analyses. Cells labeled by indirect immunofluorescence were observed under confocal microscope to detect any Golgi morphological alterations; electron microscopy of Golgi apparatus was also done. Expression of GRASP65 and phospho-GRASP65 was examined by immunoblotting. H2O2 treatment reduced the cell viability and raised the cell mortality of N2a cells in a time-dependent manner. Notable changes were only observed in the distribution and morphology of Golgi apparatus at 6 h after H2O2 treatment. The expression of GRASP65 showed no significant changes at different time points; the phosphorylated GRASP65 level was significantly increased after H2O2 treatment, peaked at 3 h, and finally dropped at 6 h. Taken together, GRASP65 phosphorylation may have a critical role in inducing cell death at the early stage after H2O2 treatment, while its role in H2O2-induced Golgi morphological changes may be complex.





Similar content being viewed by others
References
Farquhar MG, Palade GE (1981) The Golgi apparatus (complex)-(1954–1981)-from artifact to center stage. J Cell Biol 91:77s–103s
Swaab DF, Dubelaar EJ, Scherder EJ, van Someren EJ, Verwer RW (2003) Therapeutic strategies for Alzheimer disease: focus on neuronal reactivation of metabolically impaired neurons. Alzheimer Dis Assoc Disord 17(Suppl 4):S114–S122
Stieber A, Chen Y, Wei S, Mourelatos Z, Gonatas J, Okamoto K, Gonatas NK (1998) The fragmented neuronal Golgi apparatus in amyotrophic lateral sclerosis includes the trans-Golgi-network: functional implications. Acta Neuropathol 95:245–253
Fujita Y, Ohama E, Takatama M, Al-Sarraj S, Okamoto K (2006) Fragmentation of Golgi apparatus of nigral neurons with alpha-synuclein-positive inclusions in patients with Parkinson’s disease. Acta Neuropathol 112:261–265. doi:10.1007/s00401-006-0114-4
Baloyannis SJ, Costa V, Michmizos D (2004) Mitochondrial alterations in Alzheimer’s disease. Am J Alzheimers Dis Other Demen 19:89–93
Sakurai A, Okamoto K, Yaguchi M, Fujita Y, Mizuno Y, Nakazato Y, Gonatas NK (2002) Pathology of the inferior olivary nucleus in patients with multiple system atrophy. Acta Neuropathol 103:550–554. doi:10.1007/s00401-001-0500-x
Mourelatos Z, Gonatas NK, Stieber A, Gurney ME, Dal Canto MC (1996) The Golgi apparatus of spinal cord motor neurons in transgenic mice expressing mutant Cu,Zn superoxide dismutase becomes fragmented in early, preclinical stages of the disease. Proc Natl Acad Sci USA 93:5472–5477
Karecla PI, Kreis TE (1992) Interaction of membranes of the Golgi complex with microtubules in vitro. Eur J Cell Biol 57:139–146
Strosznajder R, Gadamski R, Walski M (2005) Inhibition of poly(ADP-ribose) polymerase activity protects hippocampal cells against morphological and ultrastructural alteration evoked by ischemia-reperfusion injury. Folia Neuropathol 43:156–165
Martin LJ, Brambrink AM, Price AC, Kaiser A, Agnew DM, Ichord RN, Traystman RJ (2000) Neuronal death in newborn striatum after hypoxia-ischemia is necrosis and evolves with oxidative stress. Neurobiol Dis 7:169–191. doi:10.1006/nbdi.2000.0282
Rafols JA, Daya AM, O’Neil BJ, Krause GS, Neumar RW, White BC (1995) Global brain ischemia and reperfusion: Golgi apparatus ultrastructure in neurons selectively vulnerable to death. Acta Neuropathol 90:17–30
Ji G, Ji H, Mo X, Li T, Yu Y, Hu Z (2013) The role of GRASPs in morphological alterations of Golgi apparatus: mechanisms and effects. Rev Neurosci 24:485–497. doi:10.1515/revneuro-2013-0020
Lane JD, Lucocq J, Pryde J, Barr FA, Woodman PG, Allan VJ, Lowe M (2002) Caspase-mediated cleavage of the stacking protein GRASP65 is required for Golgi fragmentation during apoptosis. J Cell Biol 156:495–509. doi:10.1083/jcb.200110007
Wang Y, Seemann J, Pypaert M, Shorter J, Warren G (2003) A direct role for GRASP65 as a mitotically regulated Golgi stacking factor. EMBO J 22:3279–3290. doi:10.1093/emboj/cdg317
Wang Y, Satoh A, Warren G (2005) Mapping the functional domains of the Golgi stacking factor GRASP65. J Biol Chem 280:4921–4928. doi:10.1074/jbc.M412407200
Yoshimura S, Yoshioka K, Barr FA, Lowe M, Nakayama K, Ohkuma S, Nakamura N (2005) Convergence of cell cycle regulation and growth factor signals on GRASP65. J Biol Chem 280:23048–23056. doi:10.1074/jbc.M502442200
Joshi G, Chi Y, Huang Z, Wang Y (2014) Abeta-induced Golgi fragmentation in Alzheimer’s disease enhances Abeta production. Proc Natl Acad Sci USA 111:E1230–E1239. doi:10.1073/pnas.1320192111
Zhang Z, Yang W, Zhu S, Lu ZG, Zhai ZH (2001) The mechanism of neuro-2a cell death induced by H2O2. Shi Yan Sheng Wu Xue Bao 34:35–43
Gonatas JO, Mezitis SG, Stieber A, Fleischer B, Gonatas NK (1989) MG-160. A novel sialoglycoprotein of the medial cisternae of the Golgi apparatus [published erratum appears in J Biol Chem 1989 Mar 5;264(7):4264]. J Biol Chem 264:646–653
Yoshiyama Y, Zhang B, Bruce J, Trojanowski JQ, Lee VM (2003) Reduction of detyrosinated microtubules and Golgi fragmentation are linked to tau-induced degeneration in astrocytes. J Neurosci 23:10662–10671
Gonatas NK, Stieber A, Gonatas JO (2006) Fragmentation of the Golgi apparatus in neurodegenerative diseases and cell death. J Neurol Sci 246:21–30. doi:10.1016/j.jns.2006.01.019
Mourelatos Z, Hirano A, Rosenquist AC, Gonatas NK (1994) Fragmentation of the Golgi apparatus of motor neurons in amyotrophic lateral sclerosis (ALS). Clinical studies in ALS of Guam and experimental studies in deafferented neurons and in beta,beta’-iminodipropionitrile axonopathy. Am J Pathol 144:1288–1300
Hicks SW, Machamer CE (2005) Golgi structure in stress sensing and apoptosis. Biochim Biophys Acta 1744:406–414. doi:10.1016/j.bbamcr.2005.03.002
Gosavi N, Lee HJ, Lee JS, Patel S, Lee SJ (2002) Golgi fragmentation occurs in the cells with prefibrillar alpha-synuclein aggregates and precedes the formation of fibrillar inclusion. J Biol Chem 277:48984–48992. doi:10.1074/jbc.M208194200
Tang D, Yuan H, Wang Y (2010) The role of GRASP65 in Golgi cisternal stacking and cell cycle progression. Traffic 11:827–842. doi:10.1111/j.1600-0854.2010.01055.x
Warren G, Levine T, Misteli T (1995) Mitotic disassembly of the mammalian Golgi apparatus. Trends Cell Biol 5:413–416
Zhou BB, Li H, Yuan J, Kirschner MW (1998) Caspase-dependent activation of cyclin-dependent kinases during Fas-induced apoptosis in Jurkat cells. Proc Natl Acad Sci USA 95:6785–6790
Acknowledgments
We would like to thank Prof. Nobuhiro Nakamura (Kyoto Sangyo University) for his kind offering of anti-phospho-S277 GRASP65 antibody. This article was supported by the National Natural Science Foundation (Grant 81171239), China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ji, G., Zhang, W., Quan, M. et al. The role of Golgi reassembly and stacking protein 65 phosphorylation in H2O2-induced cell death and Golgi morphological changes. Med Mol Morphol 49, 217–223 (2016). https://doi.org/10.1007/s00795-016-0138-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00795-016-0138-3