Abstract
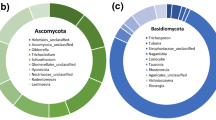
Fungal communities represent an indispensable part of the geothermal spring ecosystem; however, studies on fungal community within hot springs are still scant. Here, we used Illumina HiSeq 2500 sequencing to detect fungal community diversity in extremely acidic hot springs (pH < 4) and neutral and alkaline springs (pH > 6) of Tengchong—indicated by the presence of over 0.75 million valid reads. These sequences were phylogenetically assigned to 5 fungal phyla, 67 order, and 375 genera, indicating unexpected fungal diversity in the hot springs. The genera such as Penicillium, Entyloma, and Cladosporium dominated the fungal community in the acidic geothermal springs, while the groups such as Penicillium, Engyodontium, and Schizophyllum controlled the fungal assemblages in the alkaline hot springs. The alpha-diversity indices and the abundant fungal taxa were significantly correlated with physicochemical factors of the hot springs particularly pH, temperature, and concentrations of Fe2+, NH4+, NO −,2 and S2−, suggesting that the diversity and distribution of fungal assemblages can be influenced by the complex environmental factors of hot springs.





Similar content being viewed by others
References
Ahn SJ, Costa J, Emanuel JR (1996) PicoGreen quantitation of DNA: effective evaluation of samples pre- or post-PCR. Nucleic Acids Res 24:2623–2625
Amaral-Zettler LA (2013) Eukaryotic diversity at pH extremes. Front Microbiol 3:441
Angeles A (2013) Eukaryotic organisms in extreme acidic environments, the Rio Tinto Case. Life (Basel) 3:363–374
Appoloni S, Lekberg Y, Tercek MT, Zabinski CA, Redecker D (2008) Molecular community analysis of arbuscular mycorrhizal fungi in roots of geothermal soils in Yellowstone National Park (USA). Microb Ecol 56:649–659
Badhai J, Ghosh TS, Das SK (2015) Taxonomic and functional characteristics of microbial communities and their correlation with physicochemical properties of four geothermal springs in Odisha, India. Front Microbiol 6:1166
Briggs BR, Brodie EL, Tom LM, Dong H, Jiang H, Huang Q, Wang S, Hou W, Wu G, Huang L, Hedlund BP, Zhang C, Dijkstra P, Hungate BA (2014) Seasonal patterns in microbial communities inhabiting the hot springs of Tengchong, Yunnan Province, China. Environ Microbiol 16:1579–1591
Canganella F, Wiegel J (2014) Anaerobic thermophiles. Life (Basel) 4:77–104
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Peña AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Cole JK, Peacock JP, Dodsworth JA, Williams AJ, Thompson DB, Dong H, Wu G, Hedlund BP (2013) Sediment microbial communities in great boiling spring are controlled by temperature and distinct from water communities. ISME J 7:718–729
Colman DR, Feyhl-Buska J, Robinson KJ, Fecteau KM, Xu H, Shock EL, Boyd ES (2016) Ecological differentiation in planktonic and sediment-associated chemotrophic microbial populations in Yellowstone hot springs. FEMS Microbiol Ecol 92:fiw168
de Oliveira TB, Gomes E, Rodrigues A (2015) Thermophilic fungi in the new age of fungal taxonomy. Extremophiles 19:31–37
Delgado-Serrano L, López G, Bohorquez LC, Bustos JR, Rubiano C, Osorio-Forero C, Junca H, Baena S, Zambrano MM (2014) Neotropical Andes hot springs harbor diverse and distinct planktonic microbial communities. FEMS Microbiol Ecol 89:56–66
Du J, Liu C, Fu B, Ninomiya Y, Zhang YL, Wang CY, Wang HL, Sun ZG (2005) Variations of geothermometry and chemical-isotopic compositions of hot spring fluids in the Rehai geothermal field, Southwestern China. J Volcanol Geotherm Res 142:243–261
Duarte S, Pascoal C, Garabétian F, Cássio F, Charcosset JY (2009) Microbial decomposer communities are mainly structured by trophic status in circumneutral and alkaline streams. Appl Environ Microbiol 75:6211–6221
Fujimura R, Kim SW, Sato Y, Oshima K, Hattori M, Kamijo T, Ohta H (2016) Unique pioneer microbial communities exposed to volcanic sulfur dioxide. Sci Rep 6:19687
Guo Q (2012) Hydrogeochemistry of high-temperature geothermal systems in China: a review. Appl Geochem 27:1887–1898
Guo Q, Wang Y (2012) Geochemistry of hot springs in the Tengchong hydrothermal areas, Southwestern China. J Volcanol Geotherm Res 215–216:61–73
Hedlund BP, Murugapiran SK, Alba TW, Levy A, Dodsworth JA, Goertz GB, Ivanova N, Woyke T (2015) Uncultivated thermophiles: current status and spotlight on ‘Aigarchaeota’. Curr Opin Microbiol 25:136–145
Hou W, Wang S, Dong H, Jiang H, Briggs BR, Peacock JP, Huang Q, Huang L, Wu G, Zhi X, Li W, Dodsworth JA, Hedlund BP, Zhang C, Hartnett HE, Dijkstra P, Hungate BA (2013) A comprehensive census of microbial diversity in hot springs of Tengchong, Yunnan Province China using 16S rRNA gene pyrosequencing. PLoS One 8:e53350
Inskeep WP, Rusch DB, Jay ZJ, Herrgard MJ, Kozubal MA, Richardson TH, Macur RE, Hamamura N, Jennings Rd, Fouke BW, Reysenbach AL, Roberto F, Young M, Schwartz A, Boyd ES, Badger JH, Mathur EJ, Ortmann AC, Bateson M, Geesey G, Frazier M (2010) Metagenomes from high-temperature chemotrophic systems reveal geochemical controls on microbial community structure and function. PLoS One 5:e9773
Jiang X, Takacs-Vesbach CD (2017) Microbial community analysis of pH 4 thermal springs in Yellowstone National Park. Extremophiles 21(1):135–152
Jiang Z, Li P, Jiang D, Dai X, Zhang R, Wang Y, Wang Y (2016a) Microbial community structure and arsenic biogeochemistry in an acid vapor-Formed Spring in Tengchong Geothermal Area, China. PLoS One 11:e0146331
Jiang Z, Li P, Van Nostrand JD, Zhang P, Zhou J, Wang Y, Dai X, Zhang R, Jiang D, Wang Y (2016b) Microbial communities and arsenic biogeochemistry at the outflow of an alkaline sulfide-rich hot spring. Sci Rep 6:25262
Jiménez DJ, Andreote FD, Chaves D, Montaña JS, Osorio-Forero C, Junca H, Zambrano MM, Baena S (2012) Structural and functional insights from the metagenome of an acidic hot spring microbial planktonic community in the Colombian Andes. PLoS One 7:e52069
Kambura AK, Mwirichia RK, Kasili RW, Karanja EN, Makonde HM, Boga HI (2016a) Diversity of fungi in sediments and water sampled from the hot springs of Lake Magadi and Little Magadi in Kenya. Afr J Microbiol Res 10:330–338
Kambura AK, Mwirichia RK, Kasili RW, Karanja EN, Makonde HM, Boga HI (2016b) Bacteria and archaea diversity within the hot springs of Lake Magadi and Little Magadi in Kenya. BMC Microbiol 16:136
Kozubal MA, Macur RE, Jay ZJ, Beam JP, Malfatti SA, Tringe SG, Kocar BD, Borch T, Inskeep WP (2012) Microbial iron cycling in acidic geothermal springs of Yellowstone National Park: integrating molecular surveys, geochemical processes, and isolation of novel Fe-active microorganisms. Front Microbiol 3:109
Li SL, Lin Q, Li XR, Xu H, Yang YX, Qiao DR, Cao Y (2012) Biodiversity of the oleaginous microorganisms in Tibetan Plateau. Braz J Microbiol 43:627–634
Ma X, Baron JL, Vikram A, Stout JE, Bibby K (2015) Fungal diversity and presence of potentially pathogenic fungi in a hospital hot water system treated with on-site monochloramine. Water Res 71:197–206
Mathur J, Bizzoco RW, Ellis DG, Lipson DA, Poole AW, Levine R, Kelley ST (2007) Effects of abiotic factors on the phylogenetic diversity of bacterial communities in acidic thermal springs. Appl Environ Microbiol 73:2612–2623
Merkel AY, Pimenov NV, Rusanov II, Slobodkin AI, Slobodkina GB, Tarnovetckii IY, Frolov EN, Dubin AV, Perevalova AA, Bonch-Osmolovskaya EA (2017) Microbial diversity and autotrophic activity in Kamchatka hot springs. Extremophiles 21:307–317
Meyer-Dombard DR, Shock EL, Amend JP (2005) Archaeal and bacterial communities in geochemically diverse hot springs of Yellowstone National Park, USA. Geobiology 3:211–227
Morgenstern I, Powlowski J, Ishmael N, Darmond C, Marqueteau S, Moisan MC, Quenneville G, Tsang A (2012) A molecular phylogeny of thermophilic fungi. Fungal Biol 116:489–502
Mouchacca J (1997) Thermophilic fungi: biodiversity and taxonomic status. Cryptogam Mycol 18:19–69
Pagaling E, Wang H, Venables M, Wallace A, Grant WD, Cowan DA, Jones BE, Ma Y, Ventosa A, Heaphy S (2009) Microbial biogeography of six salt lakes in Inner Mongolia, China, and a salt lake in Argentina. Appl Environ Microbiol 75:5750–5760
Pan WZ, Huang XW, Wei KB, Zhang CM, Yang DM, Ding JM, Zhang KQ (2010) Diversity of thermophilic fungi in Tengchong Rehai National Park revealed by ITS nucleotide sequence analyses. J Microbiol 48:146–152
Peralta AL, Matthews JW, Kent AD (2014) Habitat specialization along a wetland moisture gradient differs between ammonia-oxidizing and denitrifying microorganisms. Microb Ecol 68:339–350
Powell AJ, Parchert KJ, Bustamante JM, Ricken JB, Hutchinson MI, Natvig DO (2012) Thermophilic fungi in an aridland ecosystem. Mycologia 104:813–825
Purcell D, Sompong U, Yim LC, Barraclough TG, Peerapornpisal Y, Pointing SB (2007) The effects of temperature, pH and sulphide on the community structure of hyperthermophilic streamers in hot springs of northern Thailand. FEMS Microbiol Ecol 60:456–466
Rousk J, Bååth E, Brookes PC, Lauber CL, Lozupone C, Caporaso JG, Knight R, Fierer N (2010) Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J 4:1340–1351
Salano OA, Makonde HM, Kasili RW, Wangai LN, Nawiri MP, Boga HI (2017) Diversity an distribution of fungal communities within the hot springs of soda lakes in the Kenyan rift valley. Afr J Microbiol Res 19:764–775
Sharp CE, Brady AL, Sharp GH, Grasby SE, Stott MB, Dunfield PF (2014) Humboldt’s spa: microbial diversity is controlled by temperature in geothermal environments. ISME J 8:1166–1174
Song ZQ, Chen JQ, Jiang HC, Zhou EM, Tang SK, Zhi XY, Zhang LX, Zhang CL, Li WJ (2010) Diversity of Crenarchaeota in terrestrial hot springs in Tengchong, China. Extremophiles 14:287–296
Song ZQ, Wang FP, Zhi XY, Chen JQ, Zhou EM, Liang F, Xiao X, Tang SK, Jiang HC, Zhang CL, Dong H, Li WJ (2013) Bacterial and archaeal diversities in Yunnan and Tibetan hot springs, China. Environ Microbiol 15:1160–1175
Stielow JB, Lévesque CA, Seifert KA, Meyer W, Iriny L, Smits D, Renfurm R, Verkley GJ, Groenewald M, Chaduli D, Lomascolo A, Welti S, Lesage-Meessen L, Favel A, Al-Hatmi AM, Damm U, Yilmaz N, Houbraken J, Lombard L, Quaedvlieg W, Binder M, Vaas LA, Vu D, Yurkov A, Begerow D, Roehl O, Guerreiro M, Fonseca A, Samerpitak K, van Diepeningen AD, Dolatabadi S, Moreno LF, Casaregola S, Mallet S, Jacques N, Roscini L, Egidi E, Bizet C, Garcia-Hermoso D, Martín MP, Deng S, Groenewald JZ, Boekhout T, de Beer ZW, Barnes I, Duong TA, Wingfield MJ, de Hoog GS, Crous PW, Lewis CT, Hambleton S, Moussa TA, Al-Zahrani HS, Almaghrabi OA, Louis-Seize G, Assabgui R, McCormick W, Omer G, Dukik K, Cardinali G, Eberhardt U, de Vries M, Robert V (2015) One fungus, which genes? Development and assessment of universal primers for potential secondary fungal DNA barcodes. Persoonia 35:242–263
Sun H, Santalahti M, Pumpanen J, Köster K, Berninger F, Raffaello T, Asiegbu FO, Heinonsalo J (2016) Bacterial community structure and function shift across a northern boreal forest fire chronosequence. Sci Rep 6:32411
Tripathy S, Padhi SK, Mohanty S, Samanta M, Maiti NK (2016) Analysis of the metatranscriptome of microbial communities of an alkaline hot sulfur spring revealed different gene encoding pathway enzymes associated with energy metabolism. Extremophiles 20:525–536
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267
Wang S, Dong H, Hou W, Jiang H, Huang Q, Briggs BR, Huang L (2014) Greater temporal changes of sediment microbial community than its waterborne counterpart in Tengchong hot springs, Yunnan Province, China. Sci Rep 4:7479
Wang JT, Zheng YM, Hu HW, Zhang LM, Li J, He JZ (2015) Soil pH determines the alpha diversity but not beta diversity of soil fungal community along altitude in a typical Tibetan forest ecosystem. J Soils Sediments 15:1224
Zhang XM, Liu W, Zhang GM, Jiang L, Han XG (2015) Mechanisms of soil acidification reducing bacterial diversity. Soil Biol Biochem 81:275–281
Zhang T, Wang NF, Liu HY, Zhang YQ, Yu LY (2016) Soil pH is a key determinant of soil fungal community composition in the Ny-Ålesund Region, Svalbard (High Arctic). Front Microbiol 7:227
Acknowledgements
We are especially grateful to the editor and anonymous reviewers for their valuable comments on the manuscript. The work was financed by Key Project of International Cooperation of Ministry of Science and Technology (MOST) (Nos. 2013DFA31980, 2015FY110100), National Natural Science Foundation Program of China (No. 31100017), Shaanxi University of Technology (Nos. SLGQD16-06, SLGQD16-07), China Postdoctoral Science Foundation (2016M602566) and Visiting Scholar Grant of State Key Laboratory of Biocontrol, Sun Yat-Sen University (No. SKLBC14F02). W-J Li is also supported by projected from Guangdong Province Higher Vocational Colleges & Schools Pearl River Scholar Funded Scheme (2014). The manuscript has been linguistically edited by Natalie Kaplan, Johns Hopkins University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Communicated by A. Oren.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, KH., Ding, XW., Salam, N. et al. Unexpected fungal communities in the Rehai thermal springs of Tengchong influenced by abiotic factors. Extremophiles 22, 525–535 (2018). https://doi.org/10.1007/s00792-018-1014-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-018-1014-y