Abstract
Objectives
The aim of the present study was to compare the barrier function during bacterial invasion and wound healing properties of 3 commonly used membranes including horizontal platelet-rich fibrin (H-PRF) against two commercially available resorbable collagen membranes.
Materials and methods
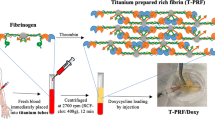
H-PRF membranes were prepared by collecting venous blood from 3 healthy volunteers using a 700 g for 8-min centrifugation protocol followed by compression into membranes. To evaluate their barrier function, 3 groups (H-PRF membrane, collagen membrane A (Bio-Gide, Geistlich), collagen membrane B (Megreen, Shanxi Ruisheng Biotechnology Co) were placed between an inner chamber and outer chamber and inoculated with S. aureus. At 2 h, 24 h, and 48 h post-inoculation, cultures from the inner and outer chambers were assessed for bacterial CFUs. Then, scanning electron microscope (SEM) was utilized to visualized the morphological destruction by bacteria of the inner and outer surfaces of the membranes. To assess the wound healing properties of each membrane, leachates from each group were applied to human gingival fibroblasts (HGF) and a scratch assay was performed at 24 h and 48 h.
Results
S. aureus showed a minimal bacterial attachment or invasion rate through either collagen membranes at 2 h post-inoculation, yet over time demonstrated rapid degradation, especially on the rougher surface. While PRF demonstrated higher number of CFUs after 2 h, no significant penetration/degradation of the H-PRF membranes was observed at 24 h and 48 h in the H-PRF group. Both collagen membranes demonstrated significant morphological changes 48 h post-bacterial innoculation, while minimal obvious morphological changes were observed in the H-PRF group. The wound healing assay also demonstrated significantly better wound closure rates in the H-PRF group.
Conclusion
H-PRF membranes exhibited better barrier function towards S. aureus over 2 days of innoculation and better wound healing ability when compared to two commercially available collagen membranes.
Clinical relevance
This study provides further evidence for the application of H-PRF membranes during guided bone regeneration by minimizing bacterial invasion. Furthermore, H-PRF membranes have significantly better ability to promote wound healing.






Similar content being viewed by others
Data availability
All data related to this paper may be requested from the authors.
References
Jung RE et al (2013) Long-term outcome of implants placed with guided bone regeneration (GBR) using resorbable and non-resorbable membranes after 12–14 years. Clin Oral Implant Res 24(10):1065–1073
Lim G et al (2018) Wound healing complications following guided bone regeneration for ridge augmentation: a systematic review and meta-analysis. Int J Oral Maxillofac Implants 33(1):41–50
Burkhardt R, Lang NP (2010) Role of flap tension in primary wound closure of mucoperiosteal flaps: a prospective cohort study. Clin Oral Implants Res 21(1):50–54
Burkhardt R et al (2008) Influence of suture tension to the tearing characteristics of the soft tissues: an in vitro experiment. Clin Oral Implants Res 19(3):314–319
Sanz-Sánchez I et al (2022) Complications in bone-grafting procedures: classification and management. Periodontol 2000 88(1):86–102
Garcia J et al (2018) Effect of membrane exposure on guided bone regeneration: a systematic review and meta-analysis. Clin Oral Implants Res 29(3):328–338
Duarte AS, Correia A, Esteves AC (2016) Bacterial collagenases - a review. Crit Rev Microbiol 42(1):106–126
Tal H et al (2008) Cross-linked and non-cross-linked collagen barrier membranes disintegrate following surgical exposure to the oral environment: a histological study in the cat. Clin Oral Implants Res 19(8):760–766
Carreño Carreño J et al (2016) Bacterial influence on consolidation of bone grafts in maxillary sinus elevation. Clin Oral Implants Res 27(11):1431–1438
Filho HN et al (2014) Autogenous bone grafts contamination after exposure to the oral cavity. J Craniofac Surg 25(2):412–414
Zhang Y (2021) Past and present of plasmatrix. Zhonghua Kou Qiang Yi Xue Za Zhi 56(8):740–746
Masuki H et al (2016) Growth factor and pro-inflammatory cytokine contents in platelet-rich plasma (PRP), plasma rich in growth factors (PRGF), advanced platelet-rich fibrin (A-PRF), and concentrated growth factors (CGF). Int J Implant Dent 2(1):1–6
Bai M-Y et al (2018) Relationships of age and sex with cytokine content and distribution in human platelet fibrin gels. Sci Rep 8(1):1–9
Crisci A, Manfred S, Crisci M (2019) The L-PRF membrane (fibrin rich in platelets and leukocytes) and its derivatives useful as a source of stem cells in wound surgery. J Stem Cell Rep 1:1–11
Choukroun J, Ghanaati S (2018) Reduction of relative centrifugation force within injectable platelet-rich-fibrin (PRF) concentrates advances patients’ own inflammatory cells, platelets and growth factors: the first introduction to the low speed centrifugation concept. Eur J Trauma Emerg Surg 44(1):87–95
Feng M et al (2020) Antibacterial effects of platelet-rich fibrin produced by horizontal centrifugation. Int J Oral Sci 12(1):1–8
Miron RJ (2021) Understanding platelet rich fibrin.: Quintessence
Castro AB et al (2017) Regenerative potential of leucocyte- and platelet-rich fibrin. Part B: sinus floor elevation, alveolar ridge preservation and implant therapy. A systematic review. J Clin Periodontol 44(2):225–234
Zhu J et al (2021) Effect of platelet-rich fibrin on the control of alveolar osteitis, pain, trismus, soft tissue healing, and swelling following mandibular third molar surgery: an updated systematic review and meta-analysis. Int J Oral Maxillofac Surg 50(3):398–406
Miron RJ et al (2019) Standardization of relative centrifugal forces in studies related to platelet‐rich fibrin. Wiley Online Library. p. 817–820
Miron R et al (2018) Controversies related to scientific report describing g-forces from studies on platelet-rich fibrin: necessity for standardization of relative centrifugal force values. J Periodontol 1(3):80
Lee J et al (2018) Flap management in alveolar ridge preservation: a systematic review and meta-analysis. Int J Oral Maxillofac Implants 33(3):613–621
Engler-Hamm D et al (2011) Ridge preservation using a composite bone graft and a bioabsorbable membrane with and without primary wound closure: a comparative clinical trial. J Periodontol 82(3):377–387
Baker JL et al (2017) Ecology of the oral microbiome: beyond bacteria. Trends Microbiol 25(5):362–374
McCormack MG et al (2015) Staphylococcus aureus and the oral cavity: an overlooked source of carriage and infection? Am J Infect Control 43(1):35–37
Quirynen M, De Soete M, Van Steenberghe D (2002) Infectious risks for oral implants: a review of the literature. Clin Oral Implant Res 13(1):1–19
Preeja C, Arun S (2014) Platelet-rich fibrin: its role in periodontal regeneration. Saudi J Dent Res 5(2):117–122
Jiménez-Aristazábal RF, Carmona JU, Prades M (2019) Changes on the structural architecture and growth factor release, and degradation in equine platelet-rich fibrin clots cultured over time. J Equine Vet 82:102789
Tang YQ, Yeaman MR, Selsted ME (2002) Antimicrobial peptides from human platelets. Infect Immun 70(12):6524–6533
Wei Y et al (2022) The effect of resting and compression time post-centrifugation on the characteristics of platelet rich fibrin (PRF) membranes. Clin Oral Investig 26(8):5281–5288
Toledano M et al (2020) Differential biodegradation kinetics of collagen membranes for bone regeneration. Polymers (Basel) 12(6):1290
Degen JL, Bugge TH, Goguen JD (2007) Fibrin and fibrinolysis in infection and host defense. J Thromb Haemost 5(s1):24–31
Collen D (1998) Staphylokinase: a potent, uniquely fibrin-selective thrombolytic agent. Nat Med 4(3):279–284
Haraji A et al (2012) Effect of plasma rich in growth factors on alveolar osteitis. Natl J Maxillofac Surg 3(1):38–41
Etulain J (2018) Platelets in wound healing and regenerative medicine. Platelets 29(6):556–568
Rodrigues M et al (2019) Wound healing: a cellular perspective. Physiol Rev 99(1):665–706
Feng MG et al (2022) Preliminary study on centrifugation time of liquid plasmatrix for soft tissue regeneration. Zhonghua Kou Qiang Yi Xue Za Zhi 57(4):366–374
Arteagoitia I, Rodriguez Andrés C, Ramos E (2018) Does chlorhexidine reduce bacteremia following tooth extraction? A systematic review and meta-analysis. Plos One 13(4):e0195592
Yu S et al (2021) Structure, barrier function, and bioactivity of platelet-rich fibrin following thermal processing. Tissue Eng Part C Methods 27(11):605–615
Funding
This work is funded by National Natural Science Foundation of China (82101019).
Author information
Authors and Affiliations
Contributions
YQ and YW conceived and designed the research. YQ, SB, and HW performed the experiments and analyzed the data. YZ and SB resiponsible for validation and supervision. YQ and YW wrote the original draft. RJM reviewed and edited the draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study was approved by the Ethics Committee of the School and Hospital of Stomatology, Wuhan University (B52/2020).
Consent to participate
Before blood drawing, the informed consent of the volunteers was obtained and signed.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qiu, Y., Bao, S., Wei, H. et al. Bacterial exclusion and wound healing potential of horizontal platelet-rich fibrin (H-PRF) membranes when compared to 2 commercially available collagen membranes. Clin Oral Invest 27, 4795–4802 (2023). https://doi.org/10.1007/s00784-023-05108-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-023-05108-w