Abstract
Objectives
To explore whether a computed tomography (CT) examination of the head and neck region induces biological damage and whether the damage was correlated with the radiation dose.
Materials and methods
Peripheral blood was taken from 33 individuals who received head and neck CT examinations. Blood samples were divided into three groups: the control group and the in vivo and in vitro irradiation groups. The number of DNA double-strand breaks was estimated by comparing the changes in the rates of γ-H2AX foci formation in the peripheral blood before and after CT examination. The absorbed dose and effective dose were calculated with the software VirtualDose based on the Monte Carlo method, and the absorbed doses in blood were estimated accordingly.
Results
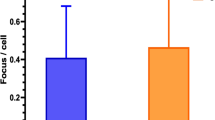
The γ-H2AX foci rates were increased in the in vivo (p < 0.001) and in vitro irradiation groups (p < 0.001) after CT examination when compared with those in the control group. The rate of γ-H2AX foci formation showed linear dose–responses for the CT dose index volume (CTDIvol), dose–length product (DLP), and blood dose after CT examination.
Conclusions
A CT examination of the head and neck region provides a high enough radiation dose to induce DNA double-strand breaks in cells in the peripheral blood. There was a linear correlation between the formation of DNA double-strand breaks and radiation doses after CT examination.
Clinical relevance
In addition to ensuring image quality, in a real clinical situation, the scanning area should be strictly administered, and repeated operations should be avoided to minimise the patient’s radiation dose.


Similar content being viewed by others
References
UNSCEAR (2018) Sources, effects and risks of ionizing radiation, Annex B. Epidemiological studies of cancer risk due to low-dose-rate radiation from environmental sources. UNSCEAR Publications. https://www.unscear.org/unscear/en/publications/2017.html. Accessed 30 March 2018
Calabrese EJ (2013) Origin of the linearity no threshold (LNT) dose-response concept. Arch Toxicol 87(9):1621–1633. https://doi.org/10.1007/s00204-013-1104-7
Shah DJ, Sachs RK, Wilson DJ (2012) Radiation-induced cancer: a modern view. Br J Radiol 85(1020):e1166–e1173. https://doi.org/10.1259/bjr/25026140
Brenner DJ (2004) Radiation risks potentially associated with low-dose CT screening of adult smokers for lung cancer. Radiology 231(2):440–445. https://doi.org/10.1148/radiol.2312030880
Brenner DJ, Elliston CD (2004) Estimated radiation risks potentially associated with full-body CT screening. Radiology 232(3):735–738. https://doi.org/10.1148/radiol.2323031095
Berrington de Gonzalez A, Darby S (2004) Risk of cancer from diagnostic X-rays: estimates for the UK and 14 other countries. Lancet 363(9406):345–351. https://doi.org/10.1016/s0140-6736(04)15433-0
Friedberg EC, Walker GC, Siede W, Wood RD, Schultz RA, Ellenberger T. (2006) DNA repair and mutagenesis, Washington
Tuteja N, Tuteja R (2001) Unraveling DNA repair in human: molecular mechanisms and consequences of repair defect. Crit Rev Biochem Mol Biol 36(3):261–290. https://doi.org/10.1080/20014091074192
Lieber MR, Ma Y, Pannicke U, Schwarz K (2004) The mechanism of vertebrate nonhomologous DNA end joining and its role in V(D)J recombination. DNA Repair (Amst) 3(8–9):817–826. https://doi.org/10.1016/j.dnarep.2004.03.015
Burma S, Chen BPC, Chen DJ (2006) Role of non-homologous end joining (NHEJ) in maintaining genomic integrity. DNA Repair (Amst) 5:1042–1048. https://doi.org/10.1016/j.dnarep.2006.05.026
Rich T, Allen RL, Wyllie AH (2000) Defying death after DNA damage. Nature 407(6805):777–783. https://doi.org/10.1038/35037717
Hoeijmakers JH (2009) DNA damage, aging, and cancer. N Engl J Med 361(15):1475–1485. https://doi.org/10.1056/NEJMra0804615
McKinnon PJ (2012) ATM and the molecular pathogenesis of ataxia telangiectasia. Annu Rev Pathol 7:303–321. https://doi.org/10.1146/annurev-pathol-011811-132509
Rothkamm K, Balroop S, Shekhdar J, Fernie P, Goh V (2007) Leukocyte DNA damage after multi-detector row CT: a quantitative biomarker of low-level radiation exposure. Radiology 242(1):244–251. https://doi.org/10.1148/radiol.2421060171
Sedelnikova OA, Rogakou EP, Panyutin IG, Bonner WM (2002) Quantitative detection of (125)IdU-induced DNA double-strand breaks with gamma-H2AX antibody. Radiat Res 158(4):486–492. https://doi.org/10.1667/0033-7587(2002)158[0486:qdoiid]2.0.co;2
Fernandez-Capetillo O, Lee A, Nussenzweig M, Nussenzweig A (2004) H2AX: the histone guardian of the genome. DNA Repair (Amst) 3(8–9):959–967. https://doi.org/10.1016/j.dnarep.2004.03.024
McCollough CH, Leng S, Yu L, Cody DD, Boone JM, McNitt-Gray MF (2011) CT dose index and patient dose: they are not the same thing. Radiology 259(2):311–316. https://doi.org/10.1148/radiol.11101800
Turner AC, Zhang D, Khatonabadi M, Zankl M, DeMarco JJ, Cagnon CH, Cody DD, Stevens DM, McCollough CH, McNitt-Gray MF (2011) The feasibility of patient size-corrected, scanner-independent organ dose estimates for abdominal CT exams. Med Phys 38(2):820–829. https://doi.org/10.1118/1.3533897
Lee C, Kim KP, Long DJ, Bolch WE (2012) Organ doses for reference pediatric and adolescent patients undergoing computed tomography estimated by Monte Carlo simulation. Med Phys 39(4):2129–2146. https://doi.org/10.1118/1.3693052
Ding A, Gao Y, Liu H, Caracappa PF, Long DJ, Bolch WE, Liu B, Xu XG (2015) VirtualDose: a software for reporting organ doses from CT for adult and pediatric patients. Phys Med Biol 60(14):5601–5625. https://doi.org/10.1088/0031-9155/60/14/5601
ICRP (2007) The 2007 Recommendations of the International Commission on Radiological Protection. ICRP publication 103. Ann ICRP 37(2–4):1–332. https://doi.org/10.1016/j.icrp.2007.10.009
ICRP (2002) Basic anatomical and physiologic data for use in radiological protection: reference values—a report of age- and genderrelated differences in the anatomical and physiological characteristics of reference individuals. ICRP publication no. 89. Ann ICRP 32:5–265. https://doi.org/10.1016/S0146-6453(03)00002-2
Shrimpton PC, Hillier MC, Lewis MA, Dunn M (2006) National survey of doses from CT in the UK: 2003. Br J Radiol 79(948):968–980. https://doi.org/10.1259/bjr/93277434
Atac GK, Parmaksiz A, Inal T, Bulur E, Bulgurlu F, Oncu T, Gundogdu S (2015) Patient doses from CT examinations in Turkey. Diagn Interv Radiol 21(5):428–434. https://doi.org/10.5152/dir.2015.14306
Chinese Society of Image Quality Control And Safety (2017) Diagnostic Reference levels for CT examinations: an expert consensus. Chin J Radiol 51(11):817–822. https://doi.org/10.3760/cma.j.issn.1005?1201.2017.11.001 (In Chinese)
Fukumoto W, Ishida M, Sakai C, Tashiro S, Ishida T, Nakano Y, Tatsugami F, Awai K (2017) DNA damage in lymphocytes induced by cardiac CT and comparison with physical exposure parameters. Eur Radiol 27(4):1660–1666. https://doi.org/10.1007/s00330-016-4519-8
Kuefner MA, Brand M, Engert C, Schwab SA, Uder M (2015) Radiation induced DNA double-strand breaks in radiology. Rofo 187(10):872–878. https://doi.org/10.1055/s-0035-1553209
Lobrich M, Rief N, Kuhne M, Heckmann M, Fleckenstein J, Rube C, Uder M (2005) In vivo formation and repair of DNA double-strand breaks after computed tomography examinations. Proc Natl Acad Sci U S A 102(25):8984–8989. https://doi.org/10.1073/pnas.0501895102
Vandevoorde C, Franck C, Bacher K, Breysem L, Smet MH, Ernst C, de Backer A, van de Moortele K, Smeets P, Thierens H (2015) gamma-H2AX foci as in vivo effect biomarker in children emphasize the importance to minimize x-ray doses in paediatric CT imaging. Eur Radiol 25(3):800–811. https://doi.org/10.1007/s00330-014-3463-8
Beels L, Bacher K, Smeets P, Verstraete K, Vral A, Thierens H (2012) Dose-length product of scanners correlates with DNA damage in patients undergoing contrast CT. Eur J Radiol 81(7):1495–1499. https://doi.org/10.1016/j.ejrad.2011.04.063
Gao Y, Quinn B, Mahmood U, Long D, Erdi Y, St. Germain J, Pandit-Taskar N, Xu XG, Bolch WE, Dauer LT (2017) A comparison of pediatric and adult CT organ dose estimation methods. BMC Med Imaging 17(1):28. https://doi.org/10.1186/s12880-017-0199-3
Popp HD, Meyer M, Brendel S, Prinzhorn W, Naumann N, Weiss C, Seifarth W, Schoenberg SO, Hofmann WK, Henzler T, Fabarius A (2016) Leukocyte DNA damage after reduced and conventional absorbed radiation doses using 3rd generation dual-source CT technology. Eur J Radiol Open 3:134–137. https://doi.org/10.1016/j.ejro.2016.06.001
Kuefner MA, Grudzenski S, Schwab SA, Wiederseiner M, Heckmann M, Bautz W, Lobrich M, Uder M (2009) DNA double-strand breaks and their repair in blood lymphocytes of patients undergoing angiographic procedures. Invest Radiol 44(8):440–446. https://doi.org/10.1097/RLI.0b013e3181a654a5
Moquet J, Barnard S, Staynova A, Lindholm C, Monteiro Gil O, Martins V, Rößler U, Vral A, Vandevoorde C, Wojewódzka M, Rothkamm K (2017) The second gamma-H2AX assay inter-comparison exercise carried out in the framework of the European biodosimetry network (RENEB). Int J Radiat Biol 93(1):58–64. https://doi.org/10.1080/09553002.2016.1207822
Funding
This study has received funding from the National Natural Science Foundation of China (No. 81671034) and National Key R&D Program of China (No. 2018YFC0807303)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was approved by the Institutional Review Board of Peking University School and Hospital of Stomatology (PKUSSIRB-201944052).
Informed consent
Informed consent was obtained from all individual participants included in this study. They received oral and written information about the design of the study and their right to withdraw at any time without negative effect on their treatment.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 124 kb)
Rights and permissions
About this article
Cite this article
Yang, P., Wang, S., Liu, D. et al. DNA double-strand breaks of human peripheral blood lymphocyte induced by CT examination of oral and maxillofacial region. Clin Oral Invest 24, 4617–4624 (2020). https://doi.org/10.1007/s00784-020-03331-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-020-03331-3