Abstract
Objectives
The present randomized double-blind clinical trial aimed to compare the efficacy in reducing dentin hypersensitivity of a dentifrice formulation containing nano-hydroxyapatite with a fluoride dentifrice and a placebo.
Methods and materials
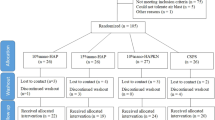
One hundred and five subjects were recruited to participate in the study. A computer-generated random table with blocking to one of the three study treatments was used in order to have 35 subjects per group: (1) nano-hydroxyapatite 2% gel toothpaste fluoride free; (2) fluoride gel toothpaste; (3) placebo. Groups 1, 2, and 3 were instructed to treat their teeth for 10 min twice a day with the provided toothpaste gel. The participant’s dentin hypersensitivity was evaluated at baseline and after 2 and 4 weeks using airblast and tactile tests. In addition, a subjective evaluation using a visual analogue scale was used.
Results
Significant lower values of cold air sensitivity and tactile sensitivity (p < 0.05) were found for the test group at 2 weeks and 4 weeks. In addition, statistically significant (p < 0.05) lower values of sensitivity were reported for group 1 compared to those for groups 2 and 3 at 2 and 4 weeks, respectively. The VAS scores were significantly lower (p < 0.05) in the test group at 2 and 4 weeks compared to those at baseline and in the control groups.
Conclusion
The application of nano-hydroxyapatite in gel toothpaste fluoride free is an effective desensitizing agent providing relief from symptoms after 2 and 4 weeks.

Similar content being viewed by others
References
Bristol AM (2005) Tooth brushing, tooth wear and dentine hypersensitivity—are they related? Int Dental J 55:261–267
Rees JS, Addy M (2002) A cross-sectional study of dentine hypersensitivity. J Clin Periodontol 29:997–1003
Brännström M, Linden LA, Johnson G (1968) Movement of dentinal and pulpal fluid caused by clinical procedures. J Dent Res 47:679–682
Chung G, Jung SJ, Oh SB (2013) Cellular and molecular mechanisms of dental nociception. J Dent Res 92:948–955. doi:10.1177/0022034513501877
Absi EG, Addy M, Adams D (1987) Dentine hypersensitivity. A study of the patency of dentinal tubules in sensitive and non-sensitive cervical dentine. J Clin Periodontol 14:280–284
Milia E, Castelli G, Bortone A, Sotgiu G, Manunta A, Pinna R, Gallina G (2013) Short-term response of three resin-based materials as desensitizing agents under oral environmental exposure. Acta Odontol Scand 71:599–609
Chidchuangchai W, Vongsavan N, Matthews B (2007) Sensory transduction mechanisms responsible for pain caused by cold stimulation of dentine in man. Arch Oral Biol 52:154–160
Han SY, Jung HI, Kwon HK, Kim BI (2013) Combined effects of Er:YAG laser and nano-carbonate apatite dentifrice on dentinal tubule occlusion: in vitro study. Photomed Laser Surg 31:342–348. doi:10.1089/pho.2012.3449
Markowitz K, Pashley DH (2007) Personal reflections on a sensitive subject. J Dent Res 86:292–295
Lin PY, Cheng YW, Chu CY, Chien KL, Lin CP, Tu YK (2013) In-office treatment for dentin hypersensitivity: a systematic review and network meta-analysis. J Clin Periodontol 40:53–64. doi:10.1111/jcpe.12011
Pinna R, Bortone A, Sotgiu G, Dore S, Usai P, Milia E (2015) Clinical evaluation of the efficacy of one self-adhesive composite in dental hypersensitivity. Clin Oral Investig 19:1663–1672. doi:10.1007/s00784-014-1390-3
Vano M, Derchi G, Barone A, Covani U (2014) Effectiveness of nano-hydroxyapatite toothpaste in reducing dentin hypersensitivity: a double-blind randomized controlled trial. Quintessence Int 45:703–711. doi:10.3290/j.qi.a32240
Vano M, Derchi G, Barone A, Genovesi A, Covani U (2015) Tooth bleaching with hydrogen peroxide and nano-hydroxyapatite: a 9-month follow-up randomized clinical trial. Int J Dent Hyg 13:301–307. doi:10.1111/idh.12123
Najibfard K, Ramalingam K, Chedjieu I, Amaechi BT (2011) Remineralization of early caries by a nano-hydroxyapatite dentifrice. J Clin Dent 22:139–143
Huang SB, Gao S, Cheng L, Yu H (2010) Combined effects of nano-hydroxyapatite and Galla chinensis on remineralisation of initial enamel lesion in vitro. J Dent 38:811–819. doi:10.1016/j.jdent.2010.06.013
Huang S, Gao S, Yu H (2011) Remineralization potential of nano-hydroxyapatite on initial enamel lesions: an in vitro study. Caries Res 45:460–468. doi:10.1159/000331207
Lee S, Kwon H, Kim B (2008) Effect of dentinal tubule occlusion by dentifrice containing nano-carbonate apatite. J Oral Rehabil 35:847–853. doi:10.1111/j.1365-2842.2008.01876.x
Roveri N, Battistella E, Bianchi CL, Foltran I, Foresti E, Iafasco M, Lelli M, Naldoni A, Palazzo B, Rimondini L (2009) Surface enamel remineralization: biomimetic apatite nanocrystals and fluoride ions different effects. J Nanomater 1:1–9. doi:10.1155/2009/746383
Roveri N, Battistella E, Foltran I, Foresti E, Iafasco M, Lelli M, Palazzo B, Rimondini L (2008) Synthetic biomimetic carbonate-hydroxyapatite nanocrystals for enamel remineralization. Adv Mater Res 47-50:821–824. doi:10.4028/www.scientific.net/AMR.47-50.821
Roveri, N, Foresti E, Lelli M, Lesci, IG. (2009) Recent advancements in preventing teeth health hazard: the daily use of hydroxyapatite instead of fluoride. Recent Patents on Biomedical Engineering 197–215. doi: 10.2174/1874764710902030197
Rimondini L, Palazzo B, Iafisco M, Canegallo L, Demarosi F, Merlo M, Roveri N (2007) The remineralizing effect of carbonate-hydroxyapatite nanocrystals on dentine. Mater Sci Forum 1:539–543. doi:10.4028/www.scientific.net/MSF.539-543.602
Yuan P, Shen X, Liu J, Hou Y, Zhu M, Huang J, Xu P (2012) Effects of dentifrice containing hydroxyapatite on dentinal tubule occlusion and aqueous hexavalent chromium cations sorption: a preliminary study. PLoS One 7:e45283. doi:10.1371/journal.pone.0045283
Browning WD, Cho SD, Deschepper EJ (2012) Effect of a nano-hydroxyapatite paste on bleaching-related tooth sensitivity. J Esthet Restor Dent 24:268–276. doi:10.1111/j.1708-8240.2011.00437.x
Holland GR, Narhi MN, Addy M, Gangarosa L, Orchardson R (1997) Guidelines for the design and conduct of clinical trials on dentine hypersensitivity. J Clin Periodontol 24:808–813
Schulz KF, Altman DG, Moher D (2010) CONSORT Group. CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. Trials 11:32
Plagmann HC, König J, Bernimoulin JP, Rudhart AC, Deschner J (1997) A clinical study comparing two high-fluoride dentifrices for the treatment of dentinal hypersensitivity. Quintessence Int 28:403–408
Schiff T, Dotson M, Cohen S, De Vizio W, McCool J, Volpe A (1994) Efficacy of a dentifrice containing potassium nitrate, soluble pyrophosphate, PVM/MA copolymer, and sodium fluoride on dentinal hypersensitivity: a twelve-week clinical study. J Clin Dent 5:87–92
Sowinski J, Ayad F, Petrone M, DeVizio W, Volpe A, Ellwood R, Davies R (2001) Comparative investigations of the efficacy of a new toothpaste containing potassium nitrate and triclosan 57 desensitising efficacy of a new dentifrice. J Clin Periodontol 28:1032–1036
Pereira R, Chava V (2001) Efficacy of a 3% potassium nitrate desensitizing mouthwash in the treatment of dentinal hypersensitivity. J Periodontol 72:1720–1725
Singal P, Gupta R, Pandit N (2005) 2% sodium fluoride-iontophoresis compared to a commercially available desensitizing agent. J Periodontol 76:351–357
Orsini G, Procaccini M, Manzoli L, Giuliodori F, Lorenzini A, Putignano A (2010) A double-blind randomized-controlled trial comparing the desensitizing efficacy of a new dentifrice containing carbonate/hydroxyapatite nanocrystals and a sodium fluoride/potassium nitrate dentifrice. J Clin Periodontol 37:510–517. doi:10.1111/j.1600-051X.2010.01558.x
Earl J, Leary R, Muller K, Langford R, Greenspan D (2011) Physical and chemical characterization of dentin surface following treatment with NovaMin technology. J Clin Dent 22:62–67
Mason S, Hughes N, Sufi F, Bannon L, Maggio B, North M, Holt J (2010) A comparative clinical study investigating the efficacy of a dentifrice containing 8% strontium acetate and 1040 ppm fluoride in a silica base and a control dentifrice containing 1450 ppm fluoride in a silica base to provide immediate relief of dentine hypersensitivity. J Clin Dent 21:42–48
Hughes N, Mason S, Jeffery P, Welton H, Tobin M, O’Shea C, Browne M (2010) A comparative clinical study investigating the efficacy of a test dentifrice containing 8% strontium acetate and 1040 ppm sodium fluoride versus a marketed control dentifrice containing 8% arginine, calcium carbonate, and 1450 ppm sodium monofluorophosphate in reducing dentinal hypersensitivity. J Clin Dent 21:49–55
Seong J, Macdonald E, Newcombe RG, Davies M, Jones SB, Johnson S, West NX (2013) In situ randomised trial to investigate the occluding properties of two desensitising toothpastes on dentine after subsequent acid challenge. Clin Oral Investig 17:195–203
Peacock JM, Orchardson R (1995) Effects of potassium ions on action potential conduction in A- and C-fibers of rat spinal nerves. J Dent Res 74:634–641
Waraaswapati N, Krongnawakul D, Jiraviboon D, Adulyanon S, Karimbux N, Pitiphat W (2005) The effect of a new toothpaste containing potassium nitrate and triclosan on gingival health, plaque formation and dentine hypersensitivity. J Clin Periodontol 32:53–58
Kim SY, Kim EJ, Kim DS, Lee IB (2013) The evaluation of dentinal tubule occlusion by desensitizing agents: a real-time measurement of dentinal fluid flow rate and scanning electron microscopy. Oper Dent 38:419–428. doi:10.2341/11-504-L
Frechoso C, Menendez M, Guisasola C, Arregui I, Tejerina J, Sieilsa A (2003) Evaluation of the efficacy of two potassium nitrate bioadhesive gels (5% and 10%) in the treatment of dentinal hypersensitivity: a randomized clinical trail. J Clin Periodontol 30:315–320
Tschoppe P, Zandim DL, Martus P, Kielbassa AM (2011) Enamel and dentine remineralization by nano-hydroxyapatite toothpastes. J Dent 39:430–437. doi:10.1016/j.jdent.2011.03.008
Sgolastra F, Petrucci A, Gatto R, Monaco A (2011) Effectiveness of laser in dentinal hypersensitivity treatment: a systematic review. J Endod 37:297–303. doi:10.1016/j.joen.2010.11.034
Sgolastra F, Petrucci A, Severino M, Gatto R, Monaco A (2013) Lasers for the treatment of dentin hypersensitivity: a meta-analysis. J Dent Res 92:492–499
Gillam D, Newman H, Davies E, Bulman J (1992) Clinical efficacy of a low abrasive dentifrice for the relief of cervical dentinal hypersensitivity. J Clin Periodontol 19:197–201
Walsh T, Worthington HV, Glenny AM, Appelbe P, Marinho VC, Shi X (2010) Fluoride toothpastes of different concentrations for preventing dental caries in children and adolescents. Cochrane Database Syst Rev 20:CD007868. doi:10.1002/14651858.CD007868.pub2
Sharma D, McGuire JA, Amini P (2013) Randomized trial of the clinical efficacy of a potassium oxalate-containing mouthrinse in rapid relief of dentin sensitivity. J Clin Dent 24:62–67
Colloca L, Benedetti F (2005) Placebos and painkillers: is mind as real as matter? Nat Rev Neurosci 6:545–552
Di Blasi Z, Harkness E, Ernst E, Georgiu A, Kleijnen J (2001) Influence of context effect on health outcomes. A systematic review. Lancet 375:757–762
Barrett B, Muller D, Rakel D, Rabago D, Marchand L, Scheder JC (2006) Placebo, meaning, and health. Perspect Biol Med 49:178–198
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
The authors declare that no financial relationships exist regarding any of the products involved in this study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
The protocol and informed consent forms were approved by the ethics committee of the University of Sassari (n° DH 2362CE).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Clinical Relevance
Based on the clinical study results, a daily application of a toothpaste containing nano-hydroxyapatite can effectively reduce dentin hypersensitivity.
Rights and permissions
About this article
Cite this article
Vano, M., Derchi, G., Barone, A. et al. Reducing dentine hypersensitivity with nano-hydroxyapatite toothpaste: a double-blind randomized controlled trial. Clin Oral Invest 22, 313–320 (2018). https://doi.org/10.1007/s00784-017-2113-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-017-2113-3