Abstract
Objectives
The aim of this study was to investigate the plaque inhibitory effect of a new 0.03 % chlorhexidine digluconate (CHX) and 0.05 % cetylpyridinium chloride (CPC) mouthrinse formulation and to explore patients’ experience and side effects after its use.
Materials and methods
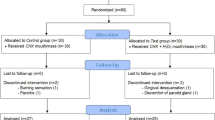
This short-term, randomized, double blind, parallel, clinical trial enrolled 150 periodontally healthy patients. These volunteers were randomly allocated to one of following mouthrinse groups (n = 50/group): 0.12 % CHX + 0.05 % CPC (Perio-Aid® Treatment alcohol-free), 0.03 % CHX + 0.05 % CPC new test formulation or to the placebo group. Clinical parameters (plaque, gingival, and stain indexes) and microbiological samples were taken at baseline, before supragingival cleaning, and after 4 days of undisturbed plaque growth, rinsing twice/day with one of the mouthrinses.
Results
Plaque reduction was similar for the 0.12 % CHX (−0.52 ± 0.55) and 0.03 % CHX (−0.47 ± 0.49) groups. Both showed significant reductions in plaque accumulation compared to the placebo (p < 0.001). The new formulation had less of a negative impact on taste perception when compared to the 0.12 % CHX solution. The new CHX mouthrinse was also able to control bacterial loads and reduce some periodontopathogens.
Conclusions
This study indicated that the new 0.03 % CHX + 0.05 % CPC formulation exerted clinical efficacy similar to that achieved by an already-marketed 0.12 % CHX + 0.05 % CPC mouthrinse, but with slightly fewer side effects.
Clinical relevance
Lower CHX mouthrinse formulations could be effective in the inhibition of plaque regrowth with reduced unpleasant subjective side effects.


Similar content being viewed by others
References
Van der Weijden F, Slot DE (2011) Oral hygiene in the prevention of periodontal diseases: the evidence. Periodontal 2000 55:104--123
Socransky SS, Haffajee AD (2002) Dental biofilms: difficult therapeutic targets. Periodontol 2000 28:12–55
Axelsson P, Lindhe J (1981) The significance of maintenance care in the treatment of periodontal disease. J Clin Periodontol 8:281–294
Lindhe J, Westfelt E, Nyman S, Socransky SS, Haffajee AD (1984) Long-term effect of surgical/non-surgical treatment of periodontal disease. J Clin Periodontol 11:448–458
Jones CG (1997) Chlorhexidine: is it still the gold standard? Periodontol 2000 15:55–62
Gjermo P, Bonesvoll P, Rölla G (1974) Relationship between plaque-inhibiting effect and retention of chlorhexidine in the human oral cavity. Arch Oral Biol 19:1031–1034
Bonesvoll P, Lökken P, Rölla G, Pous P (1974) Retention of chlorhexidine in the human oral cavity after mouth-rinses. Arch Oral Biol 19:1025–1029
Kozlovsky A, Artzi Z, Moses O, Kamin-Belsky N, Greenstein RB (2006) Interaction of chlorhexidine with smooth and rough types of titanium surfaces. J Periodontol 77:1194–1200
Hugo WB, Longworth AR (1964) Some aspects of the mode of action of chlorhexidine. J Pharm Pharmacol 16:655–662
Jenkins S, Addy M, Wade W (1988) The mechanisms of action of chlorhexidine. A study of plaque growth on enamel inserts in vivo. J Clin Periodontol 15:415–424
Schiott C, Löe H, Jensen SB, Killian M, Davies RM, Glanvind K (1970) The effect of chlorhexidine mouthrinses on the human oral flora. J Periodontal Res 5:84–89
Van Strydonck DAC, Slot DE, van der Velden U, van der Weijden F (2012) Effect of a chlorhexidine mouthrinse on plaque, gingival inflammation and staining in gingivitis patients: a systematic review. J Clin Periodontol 39:1042–1055
Cumming BR, Löe H (1973) Optimal dosage and method of delivering chlorhexidine solutions for the inhibition of dental plaque. J Periodontal Res 8:57–62
Flötra L, Gjermo P, Rölla G, Waerhaug J (1971) Side effects of chlorhexidine mouth washes. Scand J Dent Res 79:119–125
van Strydonck DAC, Timmerman MF, van der Velden U, van der Weijden GA (2005) Plaque inhibition of two commercially available chlorhexidine mouthrinses. J Clin Periodontol 32:305–309
Pizzo G, Guiglia R, Imburgia M, Pizzo I, D’Angelo M, Giuliana G (2006) The effects of antimicrobial sprays and mouthrinses on supragingival plaque regrowth: a comparative study. J Periodontol 77:248–256
McCoy LC, Wehler CJ, Rich SE, Garcia RI, Miller DR, Jones JA (2008) Adverse events associated with chlorhexidine use: results from the Department of Veterans Affairs Dental Diabetes Study. J Am Dent Assoc 139:178–283
Duss C, Lang NP, Cosyn J, Persson GR (2010) A randomized, controlled clinical trial on the clinical, microbiological, and staining effects of a novel 0.05 % chlorhexidine/herbal extract and a 0.1 % chlorhexidine mouthrinse adjunct to periodontal surgery. J Clin Periodontol 37:988–997
Hofer D, Meier A, Sener B, Guggenheim B, Attin T, Schmidlin PR (2011) Biofilm reduction and staining potential of a 0.05 % chlorhexidine rinse containing essential oils. Int J Dent Hyg 9:60–67
Bonesvoll P, Germo P (1978) A comparison between chlorhexidine and some quaternary ammonium compounds with regard to retention, salivary concentration and plaque inhibiting effect in the human mouth after mouthrinses. Arch Oral Biol 23:289–294
Segreto VA, Collins EM, Beiswanger BB (1986) A comparison of mouthrinses containing two concentrations of chlorhexidine. J Periodontol Res 21:23–32
Mandel ID (1988) Chemotherapeutic agents for controlling plaque and gingivitis. J Clin Periodontol 15:488–498
Costa X, Serrano J, Laguna E, Herrera D, Serrano J, Alonso B, Sanz M (2013) Efficacy of a new mouth rinse formulation based on 0.07 % cetylpyridinium chloride in the control of plaque and gingivitis: a 6-month randomized clinical trial. J Clin Periodontol 40:1007–1015
Herrera D, Roldán S, Santacruz I, Santos S, Masdevall M, Sanz M (2003) Differences in antimicrobial activity of four commercial 0.12 % chlorhexidine mouthrinse formulations: an in vitro contact test and salivary bacterial counts study. J Clin Periodontol 30:307–314
Addy M, Jenkins S, Newcombe R (1991) The effect of some chlorhexidine-containing mouthrinses on salivary bacterial counts. J Clin Periodontol 18:90–93
Harper PR, Milsom S, Wade W, Addy M, Moran J, Newcombe RG (1995) An approach to efficacy screening of mouthrinses: studies on a group of French products (II). Inhibition of salivary bacteria and plaque in vivo. J Clin Periodontol 22:723–727
Claydon N, Smith S, Stiller S, Newcombe RG, Addy MA (2002) Comparison of the plaque-inhibitory properties of stannous fluoride and low-concentration chlorhexidine mouthrinses. J Clin Periodontol 29:1072–1077
Mendieta C, Vallcorba N, Binney A, Addy M (1994) Comparison of 2 chlorhexidine mouthwashes on plaque regrowth in vivo and dietary staining in vitro. J Clin Periodontol 21:296–300
Quirynen M, Avontroodt P, Peeters W, Pawels M, Coucke W, Van Steenberghe D (2001) Effect of different chlorhexidine formulations in mouthrinses on de novo plaque formation. J Clin Periodontol 28:1127–1136
Barnett ML (2003) The role of therapeutic antimicrobial mouthrinses in clinical practice: control of supragingival plaque and gingivitis. J Am Dent Assoc 134:669–704
Van der Weijden GA, Timmerman MF, Niiboer A, Reijerse E, Van der Velden U (1994) Comparison of different approaches to assess bleeding on probing as indicators of gingivitis. J Clin Periodontol 21:589–594
Löe H, Silness J (1963) Periodontal disease in pregnancy. I. Prevalence and severity. Acta Odontol Scand 21:533–551
Brecx M, Macdonald LL, Legary K, Cheang M, Forgay MG (1993) Long-term effects of meridol and chlorhexidine mouthrinses on plaque, gingivitis, staining and bacterial vitality. J Dent Res 72:1194–1197
Syed SA, Loesche WJ (1972) Survival of human dental plaque flora in various transport media. Appl Microbiol 24:638–644
Turesky S, Gilmore ND, Glickman I (1970) Reduced plaque formation by the chloromethyl analogue of vitamin C. J Periodontol 41:41–43
Ashimoto A, Chen C, Bakker I, Slots J (1996) Polymerase chain reaction detection of 8 putative periodontal pathogens in subgingival plaque of gingivitis and advanced periodontitis lesions. Oral Microbiol Immunol 11:266–273
Nadkarni MA, Martin FE, Jacques NA, Hunter N (2002) Determination of bacterial load by real-time PCR using a broad-range (universal) probe and primers set. Microbiology 148:257–266
Loozen G, Boon N, Pauwels M, Quirynen M, Teughels W (2011) Live/dead real-time polymerase chain reaction to assess new therapies against dental plaque-related pathologies. Mol Oral Microbiol 26:253–261
Àlvarez G, González M, Isabal S, Blanc V, León R (2013) Method to quantify live and dead cells in multi-species oral biofilm by real-time PCR with propidium monoazide. AMB Express 3:1
Boutaga K, van Winkelhoff AJ, Vandenbroucke-Grauls CM, Savelkoul PH (2003) Comparison of real-time PCR and culture for detection of Porphyromonas gingivalis in subgingival plaque samples. J Clin Microbiol 41:4950–4954
Seow WK, Lam JHC, Tsang AKL, Holcombe T, Bird PS (2009) Oral Streptococcus species in pre-term and full-term children—a longitudinal study. Int J Paediatr Dent 19:406–411
Taskin B, Gozen AG, Duran M (2011) Selective quantification of viable Escherichia coli in biosolids by quantitative PCR with propidium monoazide modification. Appl Environ Microbiol 77:4329–4335
Addy M, Willis L, Moran J (1983) Effect of toothpaste rinses compared with chlorhexidine on plaque formation during 4-day period. J Clin Periodontol 10:89–99
Hoffmann T, Bruhn G, Richter S, Netuschil L, Brecx M (2001) Clinical controlled study on plaque and gingivitis reduction under long-term use of low-dose chlorhexidine solutions in a population exhibiting good oral hygiene. Clin Oral Investig 5:89–95
Santos S, Herrera D, Lopez E, O’Connor A, González I, Sanz M (2004) A randomized clinical trial on the short-term clinical and microbiological effects of the adjunctive use of a 0.05 % chlorhexidine mouthrinse for patients in supportive periodontal care. J Clin Periodontol 31:45–51
Quirynen M, Soers C, Desnyder M, Dekeyser C, Pauwels M, van Steenberghe D (2005) A 0.05 % cetyl pyridinium chloride/0.05 % chlorhexidine mouth rinse during maintenance phase after initial periodontal therapy. J Clin Periodontol 32:390–400
Escribano M, Herrera D, Morante S, Teughels W, Quirynen M, Sanz M (2010) Efficacy of low-concentration chlorhexidine mouth rinse in non-compliant periodontitis patients attending a supportive periodontal care programme: a randomized clinical trial. J Clin Periodontol 37:266–275
Bagis B, Baltacioglu E, Özcan M, Ustaomer S (2011) Evaluation of chlorhexidine gluconate mouthrinse-induced staining using a digital colorimeter: an in vitro study. Quintessence Int 42:213–223
Addy M, Mahdavi SA, Loyn T (1995) Dietary staining in vitro by mouth-rinses as a comparative measure of antiseptic activity and predictor of staining in vivo. J Dent 23:95–99
Boyd T, Vazquez J, Williams M (2010) Reduction of VSC and salivary bacteria by a multibenefit mouthrinse. J Breath Res 2:017013
Abiko Y, Sato T, Mayanagi G, Takahashi N (2010) Profiling of subgingival plaque biofilm microflora from periodontally healthy subjects and from subjects with periodontitis using quantitative real-time PCR. J Periodontal Res 45:389–395
Maurin M (2012) Real-time PCR as a diagnostic tool for bacterial diseases. Expert Rev Mol Diagn 12:731–754
Gunsolley JC (2006) A meta-analysis of six-month studies of antiplaque and antigingivitis agents. J Am Dent Assoc. 137:1649–1657
Acknowledgments
The authors wish to thank Vanessa Blanc, Rubén León, and Ann Bangle for their help during the study design and their suggestions during manuscript preparation.
Disclosure form
This study was funded by the Universitat Internacional de Catalunya and partially supported by a grant from Dentaid (Cerdanyola del Valles, Barcelona, Spain). Dentaid provided us with the RTF vials and conducted microbiological analysis in the Dentaid Research Center. The authors report no conflicts of interest related to this study. There is no financial relationship between any author and the commercial firm.
Author information
Authors and Affiliations
Corresponding author
Additional information
ClinicalTrials.gov Identifier: NCT02194023
Electronic supplementary material
ESM 1
(DOC 83.5 kb)
Rights and permissions
About this article
Cite this article
Mor-Reinoso, C., Pascual, A., Nart, J. et al. Inhibition of de novo plaque growth by a new 0.03 % chlorhexidine mouth rinse formulation applying a non-brushing model: a randomized, double blind clinical trial. Clin Oral Invest 20, 1459–1467 (2016). https://doi.org/10.1007/s00784-015-1625-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-015-1625-y