Abstract
Objectives
To prevent oral candidiasis, it is crucial to inactivate Candida-based biofilms on dentures. Common denture cleansing solutions cannot sufficiently inactivate Candida albicans. Therefore, we investigated the anticandidal efficacy of a physical plasma against C. albicans biofilms in vitro.
Materials and methods
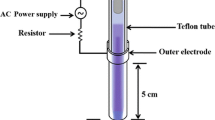
Argon or argon plasma with 1 % oxygen admixture was applied on C. albicans biofilms grown for 2, 7, or 16 days on polymethylmethacrylate discs; 0.1 % chlorhexidine digluconate (CHX) and 0.6 % sodium hypochlorite (NaOCl) solutions served as positive treatment controls. In addition, these two solutions were applied in combination with plasma for 30 min to assess potential synergistic effects. The anticandidal efficacy was determined by the number of colony forming units (CFU) in log10 and expressed as reduction factor (RF, the difference between control and treated specimen).
Results
On 2-day-biofilms, plasma treatment alone or combined with 30 min CHX treatment led to significant differences of means of CFU (RF = 4.2 and RF = 4.3), clearly superior to CHX treatment alone (RF = 0.6). Plasma treatment of 7-day-or 16-day-old biofilms revealed no significant CFU reduction. The treatment of 7-day-old (RF = 1.7) and 16-day-old (RF = 1.3) biofilms was slightly more effective with NaOCl alone than with the combined treatment of NaOCl and plasma (RF = 1.6/RF = 1.9). The combination of CHX and plasma increased the RF immaterially.
Conclusion
The use of plasma alone and in combination with antiseptics is promising anticandidal regimens for daily use on dentures when biofilms are not older than 2 days.
Clinical relevance
Plasma could help to reduce denture-associated candidiasis.

Similar content being viewed by others
References
Skupien JA, Valentini F, Boscato N, Pereira-Cenci T (2013) Prevention and treatment of Candida colonization on denture liners: a systematic review. J Prosthet Dent 110(5):356–362. doi:10.1016/j.prosdent.2013.07.003
Radford DR, Challacombe S, Walter JD (1999) Denture plaque and adherence of Candida albicans to denture-base materials in vivo and in vitro. Crit Rev Oral Biol Med 10(1):99–116. doi:10.1177/10454411990100010501
Perezous LF, Stevenson GC, Flaitz CM, Goldschmidt ME, Engelmeier RL, Nichols CM (2006) The effect of complete dentures with a metal palate on candida species growth in HIV-infected patients. J Prosthodont 15(5):306–315. doi:10.1111/j.1532-849X.2006.00127.x
Davenport JC (1970) The oral distribution of candida in denture stomatitis. Br Dent J 129(4):151–156
Jainkittivong A, Aneksuk V, Langlais RP (2010) Oral mucosal lesions in denture wearers. Gerodontology 27(1):26–32. doi:10.1111/j.1741-2358.2009.00289.x
Hawser SP, Baillie GS, Douglas LJ (1998) Production of extracellular matrix by Candida albicans biofilms. J Med Microbiol 47(3):253–256
Hawser SP, Douglas LJ (1995) Resistance of Candida albicans biofilms to antifungal agents in vitro. Antimicrob Agents Chemother 39(9):2128–2131. doi:10.1128/AAC.39.9.2128
Pietrokovski J, Azuelos J, Tau S, Mostavoy R (1995) Oral findings in elderly nursing home residents in selected countries: oral hygiene conditions and plaque accumulation on denture surfaces. J Prosthet Dent 73(2):136–141
Hong G, Murata H, Li Y, Sadamori S, Hamada T (2009) Influence of denture cleansers on the color stability of three types of denture base acrylic resin. J Prosthet Dent 101(3):205–213. doi:10.1016/S0022-3913(09)60032-9
Durkan R, Ayaz EA, Bagis B, Gurbuz A, Ozturk N, Korkmaz FM (2013) Comparative effects of denture cleansers on physical properties of polyamide and polymethyl methacrylate base polymers. Dent Mater J 32(3):367–375. doi:10.4012/dmj. 2012-110
Glass RT, Goodson LB, Bullard JW, Conrad RS (2001) Comparison of the effectiveness of several denture sanitizing systems: a clinical study. Compend Contin Educ Dent 22(12):1093–1096, 1098, 1100-2 passim; quiz 1108
Lucena-Ferreira, de Silvia Carneiro, Cavalcanti, Indira Moraes Gomes, Cury, Altair Antoninha Del Bel (2013) Efficacy of denture cleansers in reducing microbial counts from removable partial dentures: a short-term clinical evaluation. Braz Dent J 24(4):353–356. doi:10.1590/0103-6440201302183
Kramer A, Lademann J, Bender C, Sckell A, Hartmann B, Münch S, Hinz P, Ekkernkamp A, Matthes R, Koban I, Partecke L, Heidecke C, Masur K, Reuter S, Weltmann K, Koch S, Assadian O (2013) Suitability of tissue tolerable plasmas (TTP) for the management of chronic wounds. Clin Plasma Med 1(1):11–18. doi:10.1016/j.cpme.2013.03.002
von Woedtke T, Reuter S, Masur K, Weltmann K (2013) Plasmas for medicine. Phys Rep 530(4):291–320. doi:10.1016/j.physrep.2013.05.005
Koban I, Matthes R, Hübner N, Welk A, Meisel P, Holtfreter B, Sietmann R, Kindel E, Weltmann K, Kramer A, Kocher T (2010) Treatment of Candida albicans biofilms with low-temperature plasma induced by dielectric barrier discharge and atmospheric pressure plasma jet. New J Phys 12(7):073039. doi:10.1088/1367-2630/12/7/073039
He M, Du M, Fan M, Bian Z (2007) In vitro activity of eugenol against Candida albicans biofilms. Mycopathologia 163(3):137–143. doi:10.1007/s11046-007-0097-2
Sánchez MC, Llama-Palacios A, Blanc V, León R, Herrera D, Sanz M (2011) Structure, viability and bacterial kinetics of an in vitro biofilm model using six bacteria from the subgingival microbiota. J Periodont Res 46(2):252–260. doi:10.1111/j.1600-0765.2010.01341.x
European Committee for Standardisation (CEN) (2006) German version EN 1040:2005: chemical disinfectants and antiseptics—quantitative suspension test for the evaluation of basic bactericidal activity of chemical disinfectants and antiseptics—test method and requirements (phase 1). Beuth Verlag GmbH, Berlin
Ramage G, Tomsett K, Wickes BL, Lopez-Ribot JL, Redding SW (2004) Denture stomatitis: a role for Candida biofilms. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 98(1):53–59. doi:10.1016/j.tripleo.2003.04.002
Izumida FE, Ribeiro RC, Giampaolo ET, Machado AL, Pavarina AC, Vergani CE (2011) Effect of microwave disinfection on the surface roughness of three denture base resins after tooth brushing. Gerodontology 28(4):277–282. doi:10.1111/j.1741-2358.2010.00393.x
Paranhos, de Freitas Oliveira H, Salles, Sparca AE, Macedo, de Dorigan L, Silva-Lovato, da Claudia Helena, Pagnano VO, Watanabe E (2013) Complete denture biofilm after brushing with specific denture paste, neutral soap and artificial saliva. Braz Dent J 24(1):47–52. doi:10.1590/0103-6440201301946
Pisani MX, Bruhn JP, Paranhos, Oliveira HF, Silva-Lovato CH, de Souza, Freitas R, Panzeri H (2010) Evaluation of the abrasiveness of dentifrices for complete dentures. J Prosthodont 19(5):369–373. doi:10.1111/j.1532-849X.2010.00592.x
Paranhos, de Freitas Oliveira H, Peracini A, Pisani MX, Oliveira, de Cássia V, de Souza, Freitas R, Silva-Lovato CH (2013) Color stability, surface roughness and flexural strength of an acrylic resin submitted to simulated overnight immersion in denture cleansers. Braz Dent J 24(2):152–156. doi:10.1590/0103-6440201302151
Matthews D (2011) No difference between 0.12 and 0.2% chlorhexidine mouthrinse on reduction of gingivitis. Evid Based Dent 12(1):8–9. doi:10.1038/sj.ebd.6400771
de Andrade, Machado I, Cruz PC, Silva-Lovato CH, de Souza, Raphael F, Souza-Gugelmin, Monteiro MC, Paranhos, de Freitas Oliveira H (2012) Effect of chlorhexidine on denture biofilm accumulation. J Prosthodont 21(1):2–6. doi:10.1111/j.1532-849X.2011.00774.x
Felipucci, Borges DN, Davi LR, Paranhos, Oliveira HF, Bezzon OL, Silva RF, Pagnano VO (2011) Effect of different cleansers on the surface of removable partial denture. Braz Dent J 22(5):392–7
Barnabe W, de Mendonca Neto T, Pimenta FC, Pegoraro LF, Scolaro JM (2004) Efficacy of sodium hypochlorite and coconut soap used as disinfecting agents in the reduction of denture stomatitis, Streptococcus mutans and Candida albicans. J Oral Rehabil 31(5):453–459. doi:10.1111/j.1365-2842.2004.01254.x
Brożek R, Koczorowski R, Rogalewicz R, Voelkel A, Czarnecka B, Nicholson JW (2011) Effect of denture cleansers on chemical and mechanical behavior of selected soft lining materials. Dent Mater 27(3):281–290. doi:10.1016/j.dental.2010.11.003
de Sousa P, Rodrigues S, de Lucena-Ferreira, Carneiro S, da Silva, Wander J, Del Bel Cury, Altair Antoninha (2013) Evaluation of sodium hypochlorite as a denture cleanser: a clinical study. Gerodontology. doi:10.1111/ger.12104
Piskin B, Sipahi C, Akin H (2014) Effect of different chemical disinfectants on color stability of acrylic denture teeth. J Prosthodont 23(6):476–483. doi:10.1111/jopr.12131
European Committee for Standardisation (CEN) (2005) European standard EN 1275: Chemical disinfectants and antiseptics - Quantitative suspension test for the evaluation of basic fungicidal or basic yeasticidal activity of chemical disinfectants and antiseptics - Test method and requirements (phase 1). Brussels, Belgium
Lim JP, Uhm HS, Li SZ (2007) Influence of oxygen in atmospheric-pressure argon plasma jet on sterilization of Bacillus atrophaeous spores. Phys Plasma 14(9):093504. doi:10.1063/1.2773705
McKay K, Liu DX, Rong MZ, Iza F, Kong MG (2012) Generation and loss of reactive oxygen species in low-temperature atmospheric-pressure RF He + O 2 + H 2 O plasmas. J Phys D Appl Phys 45(17):172001. doi:10.1088/0022-3727/45/17/172001
Fricke K, Koban I, Tresp H, Jablonowski L, Schroder K, Kramer A, Weltmann K, von Woedtke T, Kocher T (2012) Atmospheric pressure plasma: a high-performance tool for the efficient removal of biofilms. PLoS ONE 7(8):e42539. doi:10.1371/journal.pone.0042539
Koban I, Geisel MH, Holtfreter B, Jablonowski L, Hübner N, Matthes R, Masur K, Weltmann K, Kramer A, Kocher T (2013) Synergistic effects of nonthermal plasma and disinfecting agents against dental biofilms in vitro. ISRN Dent 2013(2):1–10. doi:10.1155/2013/573262
Wagatsuma K, Hirokawa K (1995) Effect of oxygen addition to an argon glow-discharge plasma source in atomic-emission spectrometry. Anal Chim Acta 306(2–3):193–200. doi:10.1016/0003-2670(95)00007-M
Moravej M, Yang X, Hicks RF, Penelon J, Babayan SE (2006) A radio-frequency nonequilibrium atmospheric pressure plasma operating with argon and oxygen. J Appl Phys 99(9):093305
Spencer HR, Ike V, Brennan PA (2007) Review: the use of sodium hypochlorite in endodontics—potential complications and their management. Br Dent J 202(9):555–559. doi:10.1038/bdj.2007.374
Bürgers R, Witecy C, Hahnel S, Gosau M (2012) The effect of various topical peri-implantitis antiseptics on Staphylococcus epidermidis, Candida albicans, and Streptococcus sanguinis. Arch Oral Biol 57(7):940–947. doi:10.1016/j.archoralbio.2012.01.015
Al-Fattani MA, Douglas LJ (2006) Biofilm matrix of Candida albicans and Candida tropicalis: chemical composition and role in drug resistance. J Med Microbiol 55(Pt 8):999–1008. doi:10.1099/jmm. 0.46569-0
Matthes R, Bender C, Schlüter R, Koban I, Bussiahn R, Reuter S, Lademann J, Weltmann K, Kramer A (2013) Antimicrobial efficacy of two surface barrier discharges with air plasma against in vitro biofilms. PLoS ONE 8(7):e70462. doi:10.1371/journal.pone.0070462
Maisch T, Shimizu T, Isbary G, Heinlin J, Karrer S, Klampfl TG, Li Y, Morfill G, Zimmermann JL (2012) Contact-free inactivation of Candida albicans biofilms by cold atmospheric air plasma. Appl Environ Microbiol 78(12):4242–4247. doi:10.1128/Aem. 07235-11
Estrela C, Estrela CRA, Decurcio DA, Silva JA, Bammann LL (2006) Antimicrobial potential of ozone in an ultrasonic cleaning system against Staphylococcus aureus. Braz Dent J 17(2):134–138. doi:10.1590/S0103-64402006000200010
Pavlovich MJ, Chang HW, Sakiyama Y, Clark DS, Graves DB (2013) Ozone correlates with antibacterial effects from indirect air dielectric barrier discharge treatment of water. J Phys D Appl Phys 46(14):145202. doi:10.1088/0022-3727/46/14/145202
Zamperini CA, Machado AL, Vergani CE, Pavarina AC, Giampaolo ET, da Cruz, Cristino N (2010) Adherence in vitro of Candida albicans to plasma treated acrylic resin. Effect of plasma parameters, surface roughness and salivary pellicle. Arch Oral Biol 55(10):763–770. doi:10.1016/j.archoralbio.2010.06.015
Zamperini CA, Carneiro, de Lima H, Rangel EC, Cruz NC, Vergani CE, Machado AL (2013) In vitro adhesion of Candida glabrata to denture base acrylic resin modified by glow-discharge plasma treatment. Mycoses 56(2):134–144. doi:10.1111/j.1439-0507.2012.02223.x
Acknowledgments
This work was realized within the framework of the multidisciplinary research cooperation “Campus PlasmaMed,” particularly within the project “PlasmaDent.” This work was supported by a grant from the German Ministry of Education and Research (BMBF, Grant no. 13N9779), and by the Ministry of Science and Culture of the State of Mecklenburg-Western Pomerania, and the European Union, European Social Fund within the project “Plasmamedical Research – New pharmaceutical and medical fields of application” (Grant no. AU 11 038; ESF/IV-BM-B35-0010/13). The authors thank Claudia Lehnert for her excellent technical assistance and Rüdiger Titze for his skillful support in operating the plasma equipment.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matthes, R., Jablonowski, L., Koban, I. et al. In vitro treatment of Candida albicans biofilms on denture base material with volume dielectric barrier discharge plasma (VDBD) compared with common chemical antiseptics. Clin Oral Invest 19, 2319–2326 (2015). https://doi.org/10.1007/s00784-015-1463-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-015-1463-y