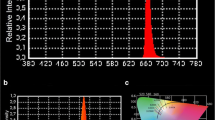
Abstract
Objectives
The aim of this study was to demonstrate that the periodontal pathogen Aggregatibacter actinomycetemcomitans (AA) can be killed by irradiation with blue light derived from a LED light-curing unit due to its endogenous photosensitizers.
Materials and methods
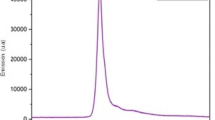
Planktonic cultures of AA and Escherichia coli were irradiated with blue light from a bluephase® C8 light-curing unit with an emission peak at 460 nm, which is usually applied for polymerization of dental resins. A CFU-assay was performed for the analysis of viable bacteria after treatment. Moreover, bacterial cells were lysed and the lysed AA and E. coli were investigated for generation of singlet oxygen. Spectroscopic measurements of lysed AA and E. coli were performed and analyzed for characteristic absorption and emission peaks.
Results
A light dose of 150 J/cm2 induced a reduction of ≥5 log10 steps of viable AA, whereas no effect of blue light was found against E. coli. Spectrally resolved measurements of singlet oxygen luminescence showed clearly that a singlet oxygen signal is generated from lysed AA upon excitation at 460 nm. Spectroscopic measurements of lysed AA exhibited characteristic absorption and emission peaks similar to those of known porphyrins and flavins.
Conclusions
AA can be inactivated by irradiation with blue light only, without application of an exogenous photosensitizer.
Clinical relevance
These results encourage further studies on the potential use of these blue light-mediated auto-photosensitization processes in the treatment of periodontitis for the successful inactivation of Aggregatibacter actinomycetemcomitans.





Similar content being viewed by others
References
Rossolini GM, Mantengoli E (2008) Antimicrobial resistance in Europe and its potential impact on empirical therapy. Clin Microbiol Infect 14(Suppl 6):2–8
Yazdankhah SP, Scheie AA, Høiby EA et al (2006) Triclosan and antimicrobial resistance in bacteria: an overview. Microb Drug Resist 12:83–90
Yamamoto T, Tamura Y, Yokota T (1988) Antiseptic and antibiotic resistance plasmid in Staphylococcus aureus that possesses ability to confer chlorhexidine and acrinol resistance. Antimicrob Agents Chemother 32:932–935
Dai T, Huang Y-Y, Hamblin MR (2009) Photodynamic therapy for localized infections—state of the art. Photodiagnosis Photodyn Ther 6:170–188
St Denis TG, Dai T, Izikson L et al (2011) All you need is light: antimicrobial photoinactivation as an evolving and emerging discovery strategy against infectious disease. Virulence 2:509–520
Wainwright M (1998) Photodynamic antimicrobial chemotherapy (PACT). J Antimicrob Chemother 42:13–28
Maisch T, Wagner J, Papastamou V et al (2009) Combination of 10 % EDTA, photosan, and a blue light hand-held photopolymerizer to inactivate leading oral bacteria in dentistry in vitro. J Appl Microbiol 107:1569–1578
Papastamou V, Nietzsch T, Staudte H et al (2011) Photoinactivation of F. nucleatum and P. gingivalis using the ruthenium-based RD3 sensitizer and a conventional halogen lamp. Arch Oral Biol 56:264–268
Al-Ahmad A, Tennert C, Karygianni L et al (2013) Antimicrobial photodynamic therapy using visible light plus water-filtered infrared-A (wIRA). J Med Microbiol 62:467–473
Ashkenazi H, Malik Z, Harth Y, Nitzan Y (2003) Eradication of Propionibacterium acnes by its endogenic porphyrins after illumination with high intensity blue light. FEMS Immunol Med Microbiol 35:17–24
Lipovsky A, Nitzan Y, Friedmann H, Lubart R (2009) Sensitivity of Staphylococcus aureus strains to broadband visible light. Photochem Photobiol 85:255–260
Hamblin MR, Viveiros J, Yang C et al (2005) Helicobacter pylori accumulates photoactive porphyrins and is killed by visible light. Antimicrob Agents Chemother 49:2822–2827
Soukos NS, Som S, Abernethy AD et al (2005) Phototargeting oral black-pigmented bacteria. Antimicrob Agents Chemother 49:1391–1396
Dai T, Gupta A, Murray CK et al (2012) Blue light for infectious diseases: Propionibacterium acnes, Helicobacter pylori, and beyond? Drug Resist Updat 15:223–236
Klinger R (1912) Untersuchungen über menschliche Aktinomykose. Zentralbl Bakteriol 62:191–200
Pulverer G, Schütt-Gerowitt H (1998) Actinobacillus actinomycetemcomitans in the human oral microflora. Zentralbl Bakteriol 288:87–92
Fine DH, Kaplan JB, Kachlany SC (2000) Schreiner HC (2006) How we got attached to Actinobacillus actinomycetemcomitans: a model for infectious diseases. Periodontol 42:114–157
Das M, Badley AD, Cockerill FR et al (1997) Infective endocarditis caused by HACEK microorganisms. Annu Rev Med 48:25–33
Henderson B, Nair SP, Ward JM, Wilson M (2003) Molecular pathogenicity of the oral opportunistic pathogen Actinobacillus actinomycetemcomitans. Annu Rev Microbiol 57:29–55
Kachlany SC (2010) Aggregatibacter actinomycetemcomitans leukotoxin: from threat to therapy. J Dent Res 89:561–570
Bjurshammar N, Johannsen A, Buhlin K et al (2012) On the red fluorescence emission of Aggregatibacter actinomycetemcomitans. OJST 2:299–306
Song H-H, Lee J-K, Um H-S et al (2013) Phototoxic effect of blue light on the planktonic and biofilm state of anaerobic periodontal pathogens. J Periodontal Implant Sci 43:72–78
Miles AA, Misra SS, Irwin JO (1938) The estimation of the bactericidal power of the blood. J Hyg (Lond) 38:732–749
Baier J, Maisch T, Maier M et al (2006) Singlet oxygen generation by UVA light exposure of endogenous photosensitizers. Biophys J 91:1452–1459
Baier J, Fuss T, Pöllmann C et al (2007) Theoretical and experimental analysis of the luminescence signal of singlet oxygen for different photosensitizers. J Photochem Photobiol B Biol 87:163–173
Boyce JM, Pittet D (2002) Guideline for hand hygiene in health-care settings: recommendations of the healthcare infection control practices advisory committee and the HICPAC/SHEA/APIC/IDSA hand hygiene task force. Infect Control Hosp Epidemiol 23:3–40
Regensburger J, Maisch T, Felgenträger A et al (2010) A helpful technology—the luminescence detection of singlet oxygen to investigate photodynamic inactivation of bacteria (PDIB). J Biophotonics 3:319–327
Eley M, Lee J, Lhoste JM et al (1970) Bacterial bioluminescence. Comparisons of bioluminescence emission spectra, the fluorescence of luciferase reaction mixtures, and the fluorescence of flavin cations. Biochemistry 9:2902–2908
Kotaki A, Yagi K (1970) Fluorescence properties of flavins in various solvents. J Biochem 68:509–516
Rimington C (1960) Spectral-absorption coefficients of some porphyrins in the Soret-band region. Biochem J 75:620–623
Gouterman M (1961) Spectra of porphyrins. J Mol Spectrosc 6:138–163
Maisch T, Baier J, Franz B et al (2007) The role of singlet oxygen and oxygen concentration in photodynamic inactivation of bacteria. Proc Natl Acad Sci U S A 104:7223–7228
Liebmann J, Born M, Kolb-Bachofen V (2010) Blue-light irradiation regulates proliferation and differentiation in human skin cells. J Invest Dermatol 130:259–269
Spranley TJ, Winkler M, Dagate J et al (2012) Curing light burns. Gen Dent 60:e210–e214
Feuerstein O, Persman N, Weiss EI (2004) Phototoxic effect of visible light on Porphyromonas gingivalis and Fusobacterium nucleatum: an in vitro study. Photochem Photobiol 80:412–415
Feuerstein O, Ginsburg I, Dayan E et al (2005) Mechanism of visible light phototoxicity on Porphyromonas gingivalis and Fusobacterium nucleatum. Photochem Photobiol 81:1186–1189
Kawada A, Aragane Y, Kameyama H et al (2002) Acne phototherapy with a high-intensity, enhanced, narrow-band, blue light source: an open study and in vitro investigation. J Dermatol Sci 30:129–135
Ganz RA, Viveiros J, Ahmad A et al (2005) Helicobacter pylori in patients can be killed by visible light. Lasers Surg Med 36:260–265
Acknowledgments
Judith Heider is gratefully acknowledged for her help with modifying the Laemmli buffer for bacterial cell lysis. Johannes Regensburger and Anita Gollmer are funded by grants of the German Research Foundation (DFG-RE-3323/2-1 and DFG-GO-2340/1-1, respectively).
Conflict of interest
All authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Tim Maisch and Gottfried Schmalz share senior authorship.
Rights and permissions
About this article
Cite this article
Cieplik, F., Späth, A., Leibl, C. et al. Blue light kills Aggregatibacter actinomycetemcomitans due to its endogenous photosensitizers. Clin Oral Invest 18, 1763–1769 (2014). https://doi.org/10.1007/s00784-013-1151-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-013-1151-8