Abstract
Bisphosphonate-associated osteonecrosis of the jaw (BP-ONJ) is one of the most often seen side effects in patients treated with bisphosphonates, presenting clinically as a non-healing wound. One theory of BP-ONJ etiology describes a negative effect on soft tissues, especially on keratinocytes, which play an important role in oral wound healing and oral soft tissue regeneration. A high cell viability of keratinocytes, which can migrate to the affected location, is essential for wound healing. The aim of this in vitro study was to investigate the effect of differently potent bisphosphonates on human oral keratinocytes (HOK).
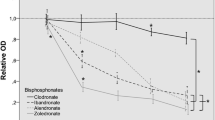
Three nitrogen-containing bisphosphonates (ibandronate, pamidronate, and zoledronate) and one non-nitrogen-containing bisphosphonate (clodronate) were compared concerning their potency on cell viability (calcein assay and MTT assay), migration ability (Boyden chamber migration assay and scratch wound proliferation assay), and apoptosis (TUNEL assay) of HOK.
The nitrogen-containing bisphosphonates, particulary highly potent pamidronate and zoledronate preparations, had a strong negative influence on cell viability, migration ability, and apoptosis of HOK. The non-nitrogen-containing clodronate even increased cell viability in higher concentrations.
This study demonstrates that bisphosphonates have a strong influence on HOK on different cellular levels like cell viability, migration ability, and apoptosis rate. The results support the theory that BP-ONJ is a multifactorially caused disease.
Furthermore, this in vitro study confirms the theory that perioperative interruption of bisphosphonate application during dental surgical procedures might be feasible to promote better tissue regeneration and wound healing.





Similar content being viewed by others
References
Diel IJ, Bergner R, Grötz KA (2007) Adverse effects of bisphosphonates: current issues. J Support Oncol 5:475–482
Marx RE (2003) Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of jaws: a growing epidemic. J Oral Maxillofac Surg 61:1115–1117
Walter C, Grötz KA, Kunkel M, Al-Nawas B (2007) Prevalence of bisphosphonate associated osteonecrosis of the jaw within the field of osteonecrosis. Support Care Cancer 15:197–202
Ruggiero SL, Dodson TB, Asseal LA, Landesberg R, Marx RE, Mehrotra B (2009) American Association of Oral and Maxillofacial Surgeons position paper on bisphosphonate-related osteonecrosis of the jaws—2009 update. J Oral Maxillofac Surg 67:2–12
Allan MR, Burr DB (2009) The pathogenesis of bisphosphonate-related osteonecrosis of the jaw: so many hypotheses, so few data. J Oral Maxillofac Surg 67:61–70
Walter C, Klein MO, Pabst A, Al-Nawas B, Duschner H, Ziebart T (2009) Influence of bisphosphonates on endothelial cells, fibroblasts, and osteogenic cells. Clin Oral Investig 14:35–41
Walter C, Pabst A, Ziebart T, Klein MO, Al-Nawas B (2010) Bisphosphonates influence migration ability and cell viability of HUVEC, fibroblasts and osteoblasts in vitro. Oral Dis (in press)
Henry D, von Moos R, Vadhan-Raj S, Hunfria V, Spencer A, Hirsh V, Wang J, Jun S, Dansey R, Yeh H (2009) A double-blind, randomized study of denosumab versus zoledronic acid for the treatment of bone metastases in patients with advanced cancer (excluding breast and prostate cancer) or multiple myeloma. Presented at the Joint ECCO 15–34th ESMO Multidisciplinary Congress, Berlin, 20–24 Sept 2009
Stopeck AT, Lipton A, Body JJ, Steger GG, Tonkin K, de Boer RH, Lichinitser M,Fujiwara Y, Yardley DA, Viniegra M, Fan M, Jiang Q, Dansey R, Jun S, Braun A (2010) Denosumab compared with zoledronic acid for the treatment of bone metastases in patients with advanced breast cancer: a randomized, double-blind study. J Clin Oncol (in press)
Neville-Webbe HL, Coleman RE (2010) Bisphosphonates and RANK ligand inhibitors for the treatment and prevention of metastatic bone diseases. Eur J Cancer 46(7):1211–1222
Taylor KH, Middlefell LS, Mizen KD (2010) Osteonecrosis of the jaws induced by anti-RANK ligand therapy. Br J Oral Maxillofac Surg 48:221–223
Santini D, Vincenzi B, Avvisati G et al (2002) Pamidronate induces modifications of circulating angiogenetic factors in cancer patients. Clin Cancer Res 8:1080–1084
Ziebart T, Pabst A, Klein MO, Kammerer P, Gauss L, Brullamnn D, Al-Nawas B, Walter C (2009) Bisphosphonates: restrictions for vasculogenesis and angiogenesis: inhibition of cell function of endothelial progenitor cells and mature endothelial cells in vitro. Clin Oral Investig (in press)
Reid IR, Bolland MJ, Grey AB (2007) Is bisphosphonate-associated osteonecrosis of the jaw caused by soft-tissue toxicity? Bone 41:318–320
Walter C, Al-Nawas B, Grötz KA, Thomas C, Thüroff JW, Zinser V, Gamm H, Beck J, Wagner W (2008) Prevalence and risk factors of bisphosphonate-associated osteonecrosis of the jaw in prostate cancer patients with advanced disease treated with zoledronate. Eur Urol 54:1066–1107
Chen T, Berenson J, Vescio R, Swift R, Gilchick A, Goodin S, LoRusso P, Ma P, Ravera C, Deckert F, Schran H, Seaman J, Skerjanec A (2002) Pharmacokinetics and pharmacodynamics of zoledronic acid in cancer patients with bone metastases. J Clin Pharmacol 42:1228–1236
Moreira MS, Katamaya E, Bombana AC, Marques MM (2005) Cytotoxicity analysis of alendronate on cultured endothelial cells and subcutaneous tissue. A pilot study. Dent Traumatol 21:329–335
Scheper MA, Badros A, Chaisuparat R, Cullen KJ, Meiller TF (2009) Effect of zoledronic acid on oral fibroblasts and epithelial cells: a potential mechanism of bisphosphonate-associated osteonecrosis. Br J Haematol 144:667–676
Scheper M, Chaisuparat R, Cullen K, Meiller T (2010) A novel-soft tissue in vitro model for bisphosphonate-associated osteonecrosis. Fibrogenesis Tissue Repair 3:6
Felix R, Guenther HL, Fleisch H (1984) The subcellular distribution of [14 C]dichloromethylenebisphosphonate and [14 C]1–hydroxyethylidene-1, 1- bisphosphonate in cultured calvaria cells. Calcif Tissue Int 36:108–113
Carano A, Teitelbaum SL, Konsek JD, Schlesinger PH, Blair HC (1990) Bisphosphonates directly inhibit the bone resorption activity of isolated avian osteoclasts in vitro. J Clin Invest 85:456–461
Fromigue O, Body JJ (2002) Bisphosphonates influence the proliferation and the maturation of normal human osteoblasts. J Endocrinol Investig 25:539–546
Xiong Y, Yang HJ, Feng J, Shi ZL, Wu LD (2009) Effects of alendronate on the proliferation and osteogenic differentiation of MG-63 cells. J Int Med Res 37:407–416
Landesberg R, Cozin M, Cremers S, Woo V, Kousteni S, Sinha S, Garrett-Sinha L, Raghavan S (2008) Inhibition of oral mucosal cell wound healing by bisphosphonates. J Oral Maxillofac Surg 66:839–847
Reszka AA, Halasy-Nagy J, Rodan GA (2001) Nitrogen bisphosphonates block retinoblastoma phosphorylation and cell growth by inhibiting the cholesterol biosynthetic pathway in a keratinocyte model for esophageal irritation. Mol Pharmacol 59:193–202
Twiss IM, de Water R, den Hartigh J, Sparidans R, Ramp-Koopmanschap W, Brill H, Wijdeveld M, Vermeij P (1994) Cytotoxic effects of pamidronate on monolayers of human intestinal epithelial (Caco-2) cells and its epithelial transport. J Pharm Sci 83:699–703
De Groen PC, Lubbe DF, Hirsch LJ, Daifotis A, Stephenson W, Freedholm D, Pryor-Tillotson S, Seleznick MJ, Pinkas H, Wang KK (1996) Esophagitis associated with the use of alendronate. N Engl J Med 335:1016–1021
Gonzalez-Moles MA, Bagan-Sebastian JV (2000) Alendronate-related oral mucosa ulcerations. J Oral Pathol Med 29:514–518
Rubegni P, Fimiani M (2006) Images in clinical medicine. Bisphosphonate-associated contact stomatitis. N Engl J Med 355:e25
Walker K, Olson MF (2005) Targeting Ras and Rho GTPases as opportunities for cancer therapeutics. Curr Opin Genet Dev 15:62–68
Suri S, Monkkonen J, Taskinen M, Peseson J, Blank MA, Phipps RJ, Rogers MJ (2001) Nitrogen-containing bisphosphoantes induce apoptosis of Caco-2 cells in vitro by inhibition the mevalonate pathway: a model of bisphosphoante-induced gastrointestinal toxicity. Bone 29:336–343
Ruggiero SL, Mehrotra B, Rosenberg TJ, Engroff SL (2004) Osteonecrosis of the jaws associated with use of bisphosphonates: a review of 63 cases. J Oral Maxillofac Surg 62:527–534
Marx RE, Cillo JE Jr, Ulloa JJ (2007) Oral bisphosphonate-induced osteonecrosis: risk factors, prediction of risk using serum CTX testing, prevention, and treatment. J Oral Maxillofac Surg 65:2397–2410
Montazeri AH, Erskine JG, McQuaker IG (2007) Oral sodium clodronate induced osteonecrosis of the jaw in a patient with myeloma. Eur J Haematol 79:69–71
Conflict of interests
No funding was received for this study. Christian Walter and Bilal Al-Nawas have received speaker fees from Roche and Christian Walter received funding from Novartis for a different study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors Pabst AM and Ziebart T contributed equally to this study.
Rights and permissions
About this article
Cite this article
Pabst, A.M., Ziebart, T., Koch, F.P. et al. The influence of bisphosphonates on viability, migration, and apoptosis of human oral keratinocytes—in vitro study. Clin Oral Invest 16, 87–93 (2012). https://doi.org/10.1007/s00784-010-0507-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-010-0507-6