Abstract
Background
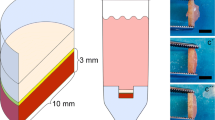
The combination of a β-tricalcium phosphate (βTCP) block with a scaffold-free chondrocyte sheet formed by the centrifugation of chondrocytes in a well was investigated with the aim of constructing an osteochondral-like structure.
Methods
Human and porcine articular cartilage chondrocytes were respectively centrifuged in a 96-well plate or cell culture insert (0.32 cm2) that was set in a 24-well plate, cultivated in the respective vessel for 3 weeks, and the cell sheets were harvested. In some cases, a cylindrical βTCP block (diameter 5 mm, height 3 mm) was placed on the sheet on days 1–7. The sheet size, cell number, and sulfated glycosaminoglycan accumulation were determined.
Results
The use of a 96-well plate for not suspension but adhesion culture and the initial centrifugation of a well containing cells were crucial to obtaining a uniformly thick cell sheet. The glycosaminoglycan density of the harvested cell sheet was comparable to that of the pellet culture. An inoculum cell number of more than 31 × 105 cells tended to result in a curved cell sheet. Culture involving 18.6 × 105 cells and the 96-well plate for adhesion culture showed no curving of the cell sheet (thickness of 0.85 mm), and these were found to be the best of the culture conditions tested. The timing of the addition of a βTCP block to the cell sheet (1–7 days) markedly affected the balance between the thickness of cell sheet parts on and in the βTCP block.
Conclusion
Centrifugation and subsequent cultivation of chondrocytes (18.6 × 105 cells) in a 96-well plate for adhesion culture led to the production of a scaffold-free cartilage-like cell sheet with a thickness of 0.85 mm. A combined osteochondral-like structure was produced by putting a βTCP block on the cell sheet. The thickness of the cell sheet on the βTCP block and the binding strength between the cell sheet and the βTCP block could be optimized by adjusting the inoculum cell number and timing of βTCP block addition.





Similar content being viewed by others
References
Daniels AU, Andriano KP, Smutz WP, Chang MK, Heller J. Evaluation of absorbable poly(ortho esters) for use in surgical implants. J Appl Biomater. 1994;5:51–64.
van der Elst M, Klein CP, de Blieck-Hogervorst JM, Patka P, Haarman HJ. Bone tissue response to biodegradable polymers used for intramedullary fracture fixation: a long-term in vivo study in sheep femora. Biomaterials. 1999;20:121–8.
Yang C, Hillas PJ, Baez JA, Nokelainen M, Balan J, Tang J, Spiro R, Polarek JW. The application of recombinant human collagen in tissue engineering. BioDrugs. 2004;18:103–19.
Martin MJ, Muotri A, Gage F, Varki A. Human embryonic stem cells express an immunogenic nonhuman sialic acid. Nat Med. 2005;11:228–32.
Takagi M, Fukui Y, Wakitani S, Yoshida T. Effect of PLGA mesh on a three-dimensional culture of chondrocytes. J Biosci Bioeng. 2004;98:477–81.
Warren L, Grayson P-H, Grace C, Darja M, David LK, Gordana V-N. Engineering custom-designed osteochondral tissue grafts. Trends Biotechnol. 2008;26:181–9.
Mano JF, Reis RL. Osteochondral defects: present situation and tissue engineering approaches. J Tissue Eng Regen Med. 2007;1:261–73.
Ivan M, Sylvie M, Andrea B, Marcel J, David W. Osteochondral tissue engineering. J Biomech. 2007;40:750–65.
Schaefer D, Martin I, Shastri P, Padera RF, Langer R, Freed LE. In vitro generation of osteochondral composites. Biomaterials. 2000;1:2599–606.
Guo X, Wang C, Duan C, Descamps M, Zhao Q, Dong L, Lü S, Anselme K, Lu J, Song YQ. Repair of osteochondral defects with autologous chondrocytes seeded onto bioceramic scaffold in sheep. Tissue Eng. 2004;10:1830–40.
van Susante JLC, Buma P, Homminga GN, van den Berg WB, Veth RPH. Chondrocyte-seeded hydroxyapatite for repair of large articular cartilage defects. A pilot study in the goat. Biomaterials. 1998;19:2367–74.
Tanaka T, Komaki H, Chazono M, Fujii K. Use of a biphasic graft constructed with chondrocytes overlying a beta-tricalcium phosphate block in the treatment of rabbit osteochondral defects. Tissue Eng. 2005;11:331–9.
Mizuno S, Tateishi T, Ushida T, Glowacki J. Hydrostatic fluid pressure enhances matrix synthesis and accumulation by bovine chondrocytes in three-dimensional culture. J Cell Physiol. 2002;193:319–27.
Freed LE, Hollander AP, Martin I, Barry JR, Langer R, Vunjak-Novakovic G. Chondrogenesis in a cell-polymer-bioreactor system. Exp Cell Res. 1998;240:58–65.
Nagai T, Furukawa KS, Sato M, Ushida T, Mochida J. Characteristics of a scaffold-free articular chondrocyte plate grown in rotational culture. Tissue Eng Part A. 2008;14:1183–93.
Nagai T, Sato M, Furukawa KS, Kutsuna T, Ohta N, Ushida T, Mochida J. Optimization of allograft implantation using scaffold-free chondrocyte plates. Tissue Eng Part A. 2008;14:1225–35.
Kitahara S, Nakagawa K, Sah RL. In vivo maturation of scaffold-free engineered articular cartilage on hydroxyapatite. Tissue Eng Part A. 2008;14:1905–13.
Waldman SD, Grynpas MD, Pilliar RM, Kandel RA. Characterization of cartilagenous tissue formed on calcium polyphosphate substrates in vitro. J Biomed Mater Res. 2002;62:323–30.
Acknowledgment
This study was partly supported by the Northern Advancement Center for Science and Technology, Japan and Graduate School of Engineering, Hokkaido University.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Niyama, K., Ide, N., Onoue, K. et al. Construction of osteochondral-like tissue graft combining β-tricalcium phosphate block and scaffold-free centrifuged chondrocyte cell sheet. J Orthop Sci 16, 613–621 (2011). https://doi.org/10.1007/s00776-011-0120-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00776-011-0120-9