Abstract.
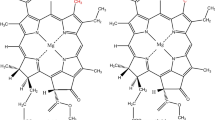
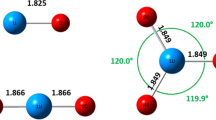
The structural characteristics of oxy- and deoxy-hemocyanins have been investigated using X-ray absorption spectroscopy both in the near-edge (XANES) and for the first shell contribution in the EXAFS region. Several arthropodan and molluscan hemocyanins have been studied in order to trace the inter- and intra-phyla differences. The XANES spectra of oxy-hemocyanins of the different species are remarkably similar, consistent with a very strongly conserved co-ordination geometry of the copper active site. In contrast, small but significant differences are observed between the deoxy-forms of arthropodan and molluscan proteins. In particular, the XANES spectra of deoxy-arthropodan hemocyanins (with the exception of L. polyphemus Hc) show a more intense edge feature at approximately 8983 eV. This difference is tentatively assigned to a more planar geometry of the copper-ligands system in the arthropodan rather than in the molluscan proteins.
The first shell analysis of the EXAFS modulation is consistent with the presence of n=3Nε 2 imidazole nitrogens at an average distance of 1.92±0.03 Å from copper in all the deoxy-hemocyanins investigated.
Binding of dioxygen results for all hemocyanins in the increase of the number of first shell back-scattering atoms to n=5 with average distances of 1.93 Å. Alternatively, by separating the contribution of Nε 2 imidazole nitrogens and of peroxide O-atoms, n=3 ligands at 1.98±0.03 Å and n=2 ligands at 1.87±0.03 Å are found.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Sabatucci, A., Ascone, I., Bubacco, L. et al. Comparison of the X-ray absorption properties of the binuclear active site of molluscan and arthropodan hemocyanins. J Biol Inorg Chem 7, 120–128 (2002). https://doi.org/10.1007/s007750100272
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s007750100272