Abstract
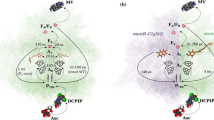
Interactions of charged peptides, such as aspartic acid peptides (Aspptds) and lysine peptides (Lysptds), with cytochrome c (cyt c) or plastocyanin (PC) have been studied by measuring electron transfer between [Fe(CN)6]4– and cyt c or PC in the presence of these peptides. Aspptds, up to penta-aspartic acid, served as competitive inhibitors of electron transfer from [Fe(CN)6]4– to oxidized cyt c, while Lysptds, up to penta-lysine, promoted electron transfer from [Fe(CN)6]4– to oxidized PC. The electron transfer inhibitory effects of Aspptds are explained as competitive inhibition due to neutralization of the positively charged amino acid residues at the surface of cyt c by electrostatic interactions, whereas the electron transfer promoting effects of Lysptds may be due to formation of PC·Lysptd or Lysptd·[Fe(CN)6]4– complexes subsequently forming an electron transferring complex, PC·Lysptd·[Fe(CN)6]4–, without repulsion of the negative charges. The inhibitory effect of Aspptds and promotional effect of Lysptds became significant as the net charge or concentration of the peptides increased. The promotional effects of Lysptds decreased as the net charge of the PC negative patch was decreased by mutagenesis. Thus, charged peptides may serve as a probe for investigation of the molecular recognition character of proteins.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 19 May 1998 / Accepted: 27 July 1998
Rights and permissions
About this article
Cite this article
Hirota, S., Endo, M., Tsukazaki, T. et al. Effects of charged peptides on electron transfer from [Fe(CN)6]4– to cytochrome c or plastocyanin. JBIC 3, 563–569 (1998). https://doi.org/10.1007/s007750050269
Issue Date:
DOI: https://doi.org/10.1007/s007750050269