Abstract
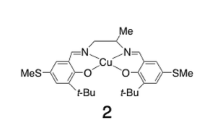
Copper(II) complexes derived from the tripodal ligand bis(3′-t–butyl-2′-hydroxybenzyl)(2-pyridylmethyl)amine (LH2) have been studied in order to mimic the redox active site of the free radical-containing copper metalloenzyme galactose oxidase. In non-coordinating solvents such as dichloromethane, only an EPR-silent dimeric complex was obtained (L2Cu2). The crystal structure of L2Cu2 revealed a "butterfly" design of the [Cu(μOR)2Cu] unit, which is not flattened and leads to a short Cu–Cu distance, the t–butyl groups being localized on the same side of the [Cu(μOR)2Cu] unit. The dimeric structure was broken down by acetonitrile or by alcohols, leading quantitatively to a brown mononuclear copper(II) complex. UV-visible and EPR data indicated the coordination of the solvent in these mononuclear complexes. Electrochemical as well as chemical (silver acetate) one-electron oxidation of acetonitrile solutions of the monomeric complex led to a yellow-green solution. Based on EPR, UV-visible and resonance Raman spectroscopy, the one-electron oxidation product was identified as a cupric phenoxyl radical system. It slowly decomposes into a product where the ligand has been substituted (dimerization) in the para position of the hydroxyl group, for one of the phenolic groups. The data for the one-electron oxidized species provides strong evidence for a free-radical copper (II) complex.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 19 July 1996 / Accepted: 16 October 1996
Rights and permissions
About this article
Cite this article
Zurita, D., Gautier-Luneau, I., Ménage, S. et al. A first model for the oxidized active form of the active site in galactose oxidase: a free-radical copper complex. JBIC 2, 46–55 (1997). https://doi.org/10.1007/s007750050105
Issue Date:
DOI: https://doi.org/10.1007/s007750050105