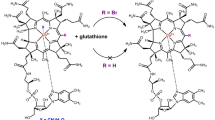
Abstract
Hypochlorous acid (HOCl) is a strong oxidant produced by myeloperoxidase. Previous work suggested that HOCl modifies the corrin ring of cobalamins to yield chlorinated species via mechanisms that are incompletely understood. Herein, we report a mechanistic study on the reaction between cyanocobalamin (CNCbl, vitamin B12) and HOCl. Under weakly acidic, neutral and weakly alkaline conditions, the reaction produces the c-lactone derivative of CNCbl chlorinated at the C10-position of corrin ring (C10–Cl–CNCbl-c-lactone). Formation of C10–Cl–CNCbl-c-lactone was not observed at pH ≥ 9.9. The chlorination of CNCbl by HOCl proceeds via two pathways involving one and two HOCl molecules: the reaction is initiated by the very fast formation of a complex between CNCbl and HOCl, which either undergoes slow transformation to chlorinated species, or rapidly reacts with a second HOCl molecule to produce C10–Cl–CNCbl. Subsequent reaction of C10–Cl–CNCbl with HOCl proceeds rapidly toward lactone ring formation by H-atom abstraction at position C8. This work uncovered mechanisms and products of the reaction of a biologically active and therapeutically used cobalamin, CNCbl and the endogenous oxidant HOCl. Binding and reactivity studies of C10–Cl–CNCbl and C10–Cl–CNCbl-c-lactone with relevant proteins of the cobalamin pathway and with cultured cells are necessary to elucidate the potential physiological effects of these species.
Graphic abstract








Similar content being viewed by others
Abbreviations
- C10–Cl–CNCbl:
-
CNCbl chlorinated at C10-position of corrin ring
- C10–Cl–CNCbl-c-lactone:
-
C-lactone derivative of C10–Cl–CNCbl
- Cbl:
-
Cobalamin
- CNCbl:
-
Cyanocobalamin
- GSCbl:
-
Glutathionylcobalamin
- H2OCbl:
-
Aquacobalamin
- HSQC:
-
Heteronuclear single quantum coherence
References
Dereven’kov IA, Salnikov DS, Silaghi-Dumitrescu R, Makarov SV, Koifman OI (2016) Coord Chem Rev 309:68–83
ó Proinsias K, Giedyk M, Gryko D (2013) Chem Soc Rev 42:6605–6619
Malech HL, Gallin JI (1987) N Engl J Med 317:687–694
Davies MJ (2011) J Clin Biochem Nutr 48:8–19
Pullar JM, Vissers MC, Winterbourn CC (2000) IUBMB Life 50:259–266
Peskin AV, Winterbourn CC (2001) Free Radic Biol Med 30:572–579
Pattison D, Davies MJ (2001) Chem Res Toxicol 14:1453–1464
Storkey C, Davies MJ, Pattison DI (2014) Free Radic Biol Med 73:60–66
Balasubramanian PN, Gould ES (1984) Inorg Chem 23:3689–3693
Dassanayake RS, Farhath MM, Shelley JT, Basu S, Brasch NE (2016) J Inorg Biochem 163:81–87
Dereven’kov IA, Shpagilev NI, Valkai L, Salnikov DS, Horváth AK, Makarov SV (2017) J Biol Inorg Chem 22:453–459
Dereven’kov IA, Makarov SV, Shpagilev NI, Salnikov DS, Koifman OI (2017) Biometals 30:757–764
Abu-Soud HM, Maitra D, Byun J, Souza CEA, Banerjee J, Saed GM, Diamond MP, Andreana PR, Pennathur S (2012) Free Radic Biol Med 52:616–625
Prinsloo FF, Breet ELJ, van Eldik R (1995). J Chem Soc Dalton Trans 685–688. https://doi.org/10.1039/dt9950000685
Okamoto N, Bito T, Hiura N, Yamamoto A, Iida M, Baba Y, Fujita T, Ishihara A, Yabuta Y, Watanabe F (2020) ACS Omega 5:6207–6214
Harwood DT, Kettle AJ, Winterbourn CC (2006) Biochem J 399:161–168
Maitra D, Ali I, Abdulridha RM, Shaeib F, Khan SN, Saed GM, Pennathur S, Abu-Soud HM (2014). PLoS ONE. https://doi.org/10.1371/journal.pone.0110595
Brown KL, Cheng S, Zou X, Zubkowski JD, Valente EJ, Knapton L, Marques HM (1997) Inorg Chem 36:3666–3675
Knapton L, Marques HM (2005). Dalton Trans 889–895. https://doi.org/10.1039/b416083e
Wagner F, Bernhauer K (1964) Ann NY Acad Sci 112:580–589
Ghadimi N, Perry CB, Fernandes MA, Govender PP, Marques HM (2015) Inorg Chim Acta 436:29–38
Wierzba AJ, Wincenciuk A, Karczewski M, Vullev VI, Gryko D (2018) Chem Eur J 24:10344–10356
Prieto L, Rossier J, Derszniak K, Dybas J, Oetterli RM, Kottelat E, Chlopicki S, Zelder F, Zobi F (2017) Chem Commun 53:6840–6843
Ashby MT, Carlson AC, Scott MJ (2004) J Am Chem Soc 126:15976–15977
Palmer AG III, Cavanagh J, Wright PE, Rance M (1991) J Magn Reson (1969) 93:151–170
Kay LE, Keifer P, Saarinen T (1992) J Am Chem Soc 114:10663–10665
Schleucher J, Schwendinger M, Sattler M, Schmidt P, Schedletzky O, Glaser SJ, Sørensen OW, Griesinger C (1994) J Biomol NMR 4:301–306
Calafat AM, Marzilli LG (1993) J Am Chem Soc 115:9182–9190
Navizet I, Perry CB, Govender PP, Marques HM (2012) J Phys Chem B 116:8836–8845
Khodov IA, Alper GA, Mamardashvili GM, Mamardashvili NZh (2015) J Mol Struct 1099:174–180
Efimov SV, Dubinin MV, Kobchikova PP, Zgadzay YuO, Khodov IA, Belosludtsev KN, Klochkov VV (2020) Biochem Biophys Res Commun 526:1054–1060
Maltceva O, Mamardashvili G, Khodov I, Lazovskiy D, Khodova V, Krest’yaninov M, Mamardashvili N, Dehaen W (2017) J Supramol Chem 29:360–369
Kormányos B, Nagypál I, Peintler G, Horváth AK (2008) Inorg Chem 47:7914–7920
Brown KL (2005) Chem Rev 105:2075–2149
Hannibal L, DiBello PM, Jacobsen DW (2013) Clin Chem Lab Med 51:477–488
Kim J, Hannibal L, Gherasim C, Jacobsen DW, Banerjee К (2009) J Biol Chem 284:33418–33424
Wingert V, Mukherjee S, Esser AJ, Behringer S, Tanimowo S, Klenzendorf M, Dereven’kov IA, Makarov SV, Jacobsen DW, Spiekerkoetter U, Hannibal L (2021) Biochimie 83:108–125
Banerjee R (2006) ACS Chem Biol 1:149–159
Acknowledgements
This work was supported by the Russian Science Foundation (Project no. 19-73-00147) to IAD. The authors thank researcher D.V. Tyurin of the Shared Scientific Resource Center, Ivanovo State University of Chemistry and Technology, Ivanovo, Russia, for his help in analyzing the products of the reaction between cyanocobalamin and hypochlorous acid using MALDI-mass-spectrometry. The NMR spectroscopy experiment was performed using the molecular fluid spectroscopy facility (http://www.ckp-rf.ru/usu/503933/) of G.A. Krestov Institute of Solution Chemistry of the Russian Academy of Sciences (ISC RAS) (Russia).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Raw data on mass-spectrometry and 2D-NMR results presented in our study are available to colleagues upon reasonable request. All requests should be directed to the corresponding author. The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dereven’kov, I.A., Osokin, V.S., Hannibal, L. et al. Mechanism of cyanocobalamin chlorination by hypochlorous acid. J Biol Inorg Chem 26, 427–434 (2021). https://doi.org/10.1007/s00775-021-01869-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-021-01869-5