Abstract
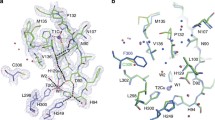
The cytochrome P450 heme monooxygenases commonly use an acid-alcohol pair of residues, within the I-helix, to activate iron-bound dioxygen. This work aims to clarify conflicting reports on the importance of the alcohol functionality in this process. Mutants of the P450, CYP199A4 (CYP199A4D251N and CYP199A4T252A), were prepared, characterised and their crystal structures were solved. The acid residue of CYP199A4 is not part of a salt bridge network, a key feature of paradigmatic model system P450cam. Instead, there is a direct proton delivery network, via a chain of water molecules, extending to the surface. Nevertheless, CYP199A4D251N dramatically reduced the activity of the enzyme consistent with a role in proton delivery. CYP199A4T252A decreased the coupling efficiency of the enzyme with a concomitant increase in the hydrogen peroxide uncoupling pathway. However, the effect of this mutation was much less pronounced than reported with P450cam. Its crystal structures revealed fewer changes at the I-helix, compared to the P450cam system. The structural changes observed within the I-helix of P450cam during oxygen activation do not seem to be required in this P450. These differences are due to the presence of a second threonine residue at position 253, which is absent in P450cam. This threonine forms part of the hydrogen bonding network, resulting in subtle structural changes and is also present across the majority of the P450 superfamily. Overall, the results suggest that while the acid-alcohol pair is important for dioxygen activation this process and the method of proton delivery can differ across P450s.
Graphic abstract







Similar content being viewed by others
References
Ortiz de Montellano PR (2010) Chem Rev 110:932–948
Poulos TL (2014) Chem Rev 114:3919–3962
Ogliaro F, de Visser SP, Cohen S, Sharma PK, Shaik S (2002) J Am Chem Soc 124:2806–2817
Rittle J, Green MT (2010) Science 330:933–937
Sharma PK, De Visser SP, Shaik S (2003) J Am Chem Soc 125:8698–8699
Meunier B, de Visser SP, Shaik S (2004) Chem Rev 104:3947–3980
Shaik S, Cohen S, Wang Y, Chen H, Kumar D, Thiel W (2010) Chem Rev 110:949–1017
Groves JT, McClusky GA (1976) J Am Chem Soc 98:859–861
Huang X, Groves JT (2017) J Biol Inorg Chem 22:185–207
Sarkar MR, Houston SD, Savage GP, Williams CM, Krenske EH, Bell SG, De Voss JJ (2019) J Am Chem Soc 141:19688-19699
Stok JE, Yamada S, Farlow AJ, Slessor KE, De Voss JJ (2013) Biochim Biophys Acta 1834:688–696
Cupp-Vickery JR, Han O, Hutchinson CR, Poulos TL (1996) Nat Struct Biol 3:632–637
Nagano S, Poulos TL (2005) J Biol Chem 280:31659–31663
Raag R, Martinis SA, Sligar SG, Poulos TL (1991) Biochemistry 30:11420–11429
Vidakovic M, Sligar SG, Li H, Poulos TL (1998) Biochemistry 37:9211–9219
Schlichting I, Berendzen J, Chu K, Stock AM, Maves SA, Benson DE, Sweet RM, Ringe D, Petsko GA, Sligar SG (2000) Science 287:1615–1622
Imai M, Shimada H, Watanabe Y, Matsushima-Hibiya Y, Makino R, Koga H, Horiuchi T, Ishimura Y (1989) Proc Natl Acad Sci USA 86:7823–7827
Gerber NC, Sligar SG (1994) J Biol Chem 269:4260–4266
Martinis SA, Atkins WM, Stayton PS, Sligar SG (1989) J Am Chem Soc 111:9252–9253
Gerber NC, Sligar SG (1992) J Am Chem Soc 114:8742–8743
Ishigooka M, Shimizu T, Hiroya K, Hatano M (1992) Biochemistry 31:1528–1531
Yeom H, Sligar SG (1997) Arch Biochem Biophys 337:209–216
Lee-Robichaud P, Akhtar ME, Akhtar M (1998) Biochem J 330(Pt 2):967–974
Zhou DJ, Korzekwa KR, Poulos T, Chen SA (1992) J Biol Chem 267:762–768
Kimata Y, Shimada H, Hirose T, Ishimura Y (1995) Biochem Biophys Res Commun 208:96–102
Bell SG, Hoskins N, Xu F, Caprotti D, Rao Z, Wong LL (2006) Biochem Biophys Res Commun 342:191–196
Bell SG, Tan AB, Johnson EO, Wong LL (2010) Mol Biosyst 6:206–214
Bell SG, Yang W, Tan AB, Zhou R, Johnson EO, Zhang A, Zhou W, Rao Z, Wong LL (2012) Dalton Trans 41:8703–8714
Bell SG, Zhou R, Yang W, Tan AB, Gentleman AS, Wong LL, Zhou W (2012) Chem Eur J 18:16677–16688
Coleman T, Chao RR, Bruning JB, De Voss J, Bell SG (2015) RSC Adv 5:52007–52018
Chao RR, De Voss JJ, Bell SG (2016) RSC Adv 6:55286–55297
Coleman T, Wong SH, Podgorski MN, Bruning JB, De Voss JJ, Bell SG (2018) ACS Catal 8:5915–5927
Bell SG, Xu F, Forward I, Bartlam M, Rao Z, Wong L-L (2008) J Mol Biol 383:561–574
Omura T, Sato R (1964) J Biol Chem 239:2370–2378
Coleman T, Chao RR, De Voss J, Bell SG (2016) Biochim Biophys Acta Proteins Proteom 1864:667–675
Maddigan NK, Bell SG (2017) Arch Biochem Biophys 615:15–21
Williams JW, Morrison JF (1979) Methods Enzymol 63:437–467
Xu F, Bell SG, Rao Z, Wong LL (2007) Protein Eng Des Sel 20:473–480
Cowieson NP, Aragao D, Clift M, Ericsson DJ, Gee C, Harrop SJ, Mudie N, Panjikar S, Price JR, Riboldi-Tunnicliffe A, Williamson R, Caradoc-Davies T (2015) J Synchrotron Radiat 22:187–190
McPhillips TM, McPhillips SE, Chiu HJ, Cohen AE, Deacon AM, Ellis PJ, Garman E, Gonzalez A, Sauter NK, Phizackerley RP, Soltis SM, Kuhn P (2002) J Synchrotron Radiat 9:401–406
Battye TG, Kontogiannis L, Johnson O, Powell HR, Leslie AG (2011) Acta Crystallogr D Biol Crystallogr 67:271–281
Evans PR, Murshudov GN (2013) Acta Crystallogr D Biol Crystallogr 67:1204–1214
Winn MD, Ballard CC, Cowtan KD, Dodson EJ, Emsley P, Evans PR, Keegan RM, Krissinel EB, Leslie AG, McCoy A, McNicholas SJ, Murshudov GN, Pannu NS, Potterton EA, Powell HR, Read RJ, Vagin A, Wilson KS (2011) Acta Crystallogr D Biol Crystallogr 67:235–242
McCoy AJ, Grosse-Kunstleve RW, Adams PD, Winn MD, Storoni LC, Read RJ (2007) J Appl Crystallogr 40:658–674
Emsley P, Lohkamp B, Scott WG, Cowtan K (2010) Acta Crystallogr D Biol Crystallogr 66:486–501
Adams PD, Afonine GBPV, Chen VB, Davis IW, Echols N, Headd JJ, Hung L-W, Kapral GJ, Grosse-Kunstleve RW, McCoy AJ, Moriarty NW, Oeffner R, Read RJ, Richardson DC, Richardson JS, Terwilliger TC, Zwart PH (2010) Acta Crystallogr D Biol Crystallogr 66:213–221
Jefcoate CR (1978) Methods Enzymol 52:258–279
Dawson JH, Andersson LA, Sono M (1982) J Biol Chem 257:3606–3617
Dawson JH, Andersson LA, Sono M (1983) J Biol Chem 258:13637–13645
Podgorski MN, Coleman T, Chao RR, De Voss JJ, Bruning JB, Bell SG (2020) J Inorg Biochem 203:110913
Batabyal D, Poulos TL (2013) Biochemistry 52:8898–8906
Hasemann CA, Kurumbail RG, Boddupalli SS, Peterson JA, Deisenhofer J (1995) Structure 3:41–62
Guengerich FP (2001) Chem Res Toxicol 14:611–650
Yeom H, Sligar SG, Li H, Poulos TL, Fulco AJ (1995) Biochemistry 34:14733–14740
Truan G, Peterson JA (1998) Arch Biochem Biophys 349:53–64
Cryle MJ, De Voss JJ (2008) ChemBioChem 9:261–266
Hishiki T, Shimada H, Nagano S, Egawa T, Kanamori Y, Makino R, Park SY, Adachi S, Shiro Y, Ishimura Y (2000) J Biochem 128:965–974
Kim D, Heo YS, Ortiz de Montellano PR (2008) Arch Biochem Biophys 474:150–156
Dhers L, Ducassou L, Boucher JL, Mansuy D (2017) Cell Mol Life Sci 74:1859–1869
Vaz AD, Pernecky SJ, Raner GM, Coon MJ (1996) Proc Natl Acad Sci USA 93:4644–4648
Vaz AD, McGinnity DF, Coon MJ (1998) Proc Natl Acad Sci USA 95:3555–3560
Lau ICK, Feyereisen R, Nelson DR, Bell SG (2019) Arch Biochem Biophys 669:11–21
Child SA, Bradley JM, Pukala TL, Svistunenko DA, Le Brun NE, Bell SG (2018) Chem Sci 9:7948–7957
Slessor KE, Farlow AJ, Cavaignac SM, Stok JE, De Voss JJ (2011) Arch Biochem Biophys 507:154–162
Coelho PS, Brustad EM, Kannan A, Arnold FH (2013) Science 339:307–310
Acknowledgements
This work was supported by ARC Grant DP140103229 (to JJDV and SGB). SGB acknowledges the ARC for a Future Fellowship (FT140100355). The authors also acknowledge the award of Australian Government Research Training Program Scholarships (PhD to TC and MPhil to MNP). We would like to thank the scientists at MX1 beamline for help with data collection. We acknowledge ANSTO for financial support and in providing the facility used in this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Coleman, T., Stok, J.E., Podgorski, M.N. et al. Structural insights into the role of the acid-alcohol pair of residues required for dioxygen activation in cytochrome P450 enzymes. J Biol Inorg Chem 25, 583–596 (2020). https://doi.org/10.1007/s00775-020-01781-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-020-01781-4