Abstract
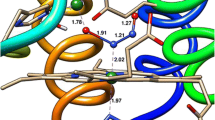
Quercetin 2,3-dioxygenase (QDO) is an enzyme which accepts various transition metal ions as cofactors, and cleaves the heterocyclic ring of quercetin with consumption of dioxygen and release of carbon monoxide. QDO from B. subtilis that binds Mn(II) displays an unprecedented nitroxygenase activity, whereby nitroxyl (HNO) is incorporated into quercetin cleavage products instead of dioxygen. Interestingly, the reaction proceeds with high regiospecificity, i.e., nitrogen and oxygen atoms of HNO are incorporated into specific fragments of the cleavage product. A nonenzymatic base-catalyzed reaction, which occurs in pH above 7.5, yields the same reaction products. Herein, we report results of quantum chemical studies on the mechanisms of the nitroxygenase reaction of Mn-QDO. Density functional method with dispersion correction (B3LYP-D3) was applied to the Mn-QDO active site model and the reactants of the nonenzymatic reaction. Co(II)- and Fe(II)-variants of the active site were also considered. Analysis of reaction energy profiles suggests that the regiospecificity of the reaction is an inherent property of the reactants, whereas the unique reactivity of Mn-QDO, as opposed to Co- or Fe-QDO that do not catalyze nitroxygenation, stems from weak HNO binding and lack of strong preference for coordination of HNO through the nitrogen atom. Moreover, the enzyme activates quercetin through deprotonation and the proton acceptor—Glu69 needs to reorient for the reaction to proceed.



















Similar content being viewed by others
References
van Acker SA, van den Berg DJ, Tromp MN, Griffioen DH, van Bennekom WP, van der Vijgh WJ, Bast A (1996) Free Radic Biol Med 20:331–342
Li M, Xu Z (2008) Arch Pharm Res 31:640–644
Plaper A, Golob M, Hafner I, Oblak M, Solmajer T, Jerala R (2003) Biochem Biophys Res Commun 306:530–536
Gellert M, Mizuuchi K, O’Dea MH, Nash HA (1976) Proc Natl Acad Sci USA 73:3872–3876
Oka T, Simpson FJ (1971) Biochem Biophys Res Commun 43:1–5
Dunwell JM (1998) Biotechnol Genet Eng Rev 15:1–32
Bowater L, Fairhurst SA, Just VJ, Bornemann S (2004) FEBS Lett 557:45–48
Gopal B, Madan LL, Betz SF, Kossiakoff AA (2005) Biochemistry 2005(44):193–201
Saito T, Kawakami T, Yamanaka S, Okumura M (2015) J Phys Chem B 119:6952–6962
Siegbahn PEM (2004) Inorg Chem 43:5944–5953
Kumar MR, Zapata A, Ramirez AJ, Bowen SK, Francisco WA, Farmer PJ (2011) Proc Natl Acad Sci 108:18926–18931
Steiner RA, Kalk KH, Dijkstra BW (2002) Proc Natl Acad Sci USA 99:16625–16630
Lemanska K, Szymusiak H, Tyrakowska B, Zieliński R, Soffers AE, Rietjens IM (2001) Free Radic Biol Med 31:869–881
Dutton AS, Fukuto JM, Houk KN (2005) Inorg Chem 44:4024–4028
Shafirovich V, Lymar SV (2002) Proc Natl Acad Sci USA 99:7340–7345
Shafirovich V, Lymar SV (2003) J Am Chem Soc 125:6547–6552
Parr R, Yang W (1994) Density-functional theory of atoms and molecules international series of monographs on chemistry. Oxford University Press, USA
Grimme S, Ehrlich S, Goerigk L (2011) J Comput Chem 32:1456–1465
Ditchfield R, Hehre WJ, Pople JA (1971) J Chem Phys 54:724
Hehre WJ, Ditchfield R, Pople J (1972) J Chem Phys 56:2257
Miertuš S, Scrocco JE, Tomasi (1981) Chem Phys 55:117–129
Raghavachari K, Binkley J, Seeger R, Pople J (1980) J Chem Phys 72:650–654
Frisch M, Pople J, Binkley J (1984) J Chem Phys 80:3265–3269
Yamaguchi K, Fukui H, Fueno T (1986) Chem Lett 15:625–628
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery Jr JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Revision D.01. Gaussian, Inc. Wallingford, CT
Fukui KT, Yonezawa T, Shingu H (1952) J Chem Phys 20:722
Parr R, Yang W (1984) J Am Chem Soc 106:4049
Han X, Kumar MR, Farmer PJ (2016) Tetrahedron Lett 57:399–402
Marcus R, Sutin N (1985) Biochim Biophys Acta BBA Rev Bioenerg 811:265–322
Schaab MR, Barney BM, Francisc WA (2006) Biochemistry 45:1009–1016
Matuz A, Giorgi M, Speier G, Kaizer J (2013) Polyhedron 63:41–49
Shook RL, Gunderson WA, Greaves J, Ziller JW, Hendrich MP, Borovik AS (2008) J Am Chem Soc 130:8888–8889
Copeland DM, Soares AS, West AH, Richter-Addo GB (2006) J Inorg Biochem 100:1413–1425
Zahran ZN, Chooback L, Copeland DM, West AH, Richter-Addo GB (2008) J Inorg Biochem 102:216–233
Carducci MD, Pressprich MR, Coppens P (1997) J Am Chem Soc 119:2669–2678
Morioka Y, Takeda S, Tomizawa H, Miki EI (1998) Chem Phys Lett 292:625–630 (cited By 15)
Kurtikyan TS, Hovhannisyan AA, Hakobyan ME, Patterson JC, Iretskii A, Ford PC (2007) J Am Chem Soc 129:3576–3585
Berto TC, Lehnert N (2011) Inorg Chem 50:7361–7363
Perissinotti LL, Marti MA, Doctorovich F, Luque FJ, Estrin DA (2008) Biochemistry 47:9793–9802
Linder DP, Rodgers KR (2005) Inorg Chem 44:8259–8264
Merkens H, Kappl R, Jakob RP, Schmid FX, Fetzner S (2008) Biochemistry 47:12185–12196
Mbughuni MM, Chakrabarti M, Hayden JA, Bominaar EL, Hendrich MP, Münck E, Lipscomb JD (2010) Proc Natl Acad Sci USA 107:16788–16793
Chiang CW, Kleespies ST, Stout HD, Meier KK, Li PY, Bominaar EL, Que L Jr, Münck E, Lee WZ (2014) J Am Chem Soc 136:10846–10849
Bonner F, Hughes M (1988) Comments Inorg Chem 7:215–234
Ruud K, Helgaker T, Uggerud E (1997) J Mol Struct (Thoechem) 393:59–71
Sun YJ, Huang QQ, Li P, Zhang JJ (2015) Dalton Trans 44:13926–13938
Acknowledgments
The authors thank Dr. Mariusz Radoń for helpful suggestions. This research was supported in part by PL-Grid Infrastructure. This research project was supported by Grant No. UMO-2011/03/B/NZ1/04999 from the National Science Centre, Poland.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
775_2016_1356_MOESM1_ESM.pdf
Two reaction paths investigating the role of switch of Glu69 H-bond and optimized Cartesian coordinates of all obtained stationary points are available
Rights and permissions
About this article
Cite this article
Wojdyła, Z., Borowski, T. DFT study of the mechanism of manganese quercetin 2,3-dioxygenase: quest for origins of enzyme unique nitroxygenase activity and regioselectivity. J Biol Inorg Chem 21, 475–489 (2016). https://doi.org/10.1007/s00775-016-1356-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-016-1356-9