Abstract
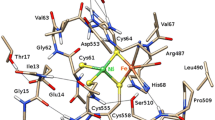
Catalytically inactive oxidized O2-sensitive [NiFe]-hydrogenases are characterized by a mixture of the paramagnetic Ni-A and Ni-B states. Upon O2 exposure, enzymes in a partially reduced state preferentially form the unready Ni-A state. Because partial O2 reduction should generate a peroxide intermediate, this species was previously assigned to the elongated Ni–Fe bridging electron density observed for preparations of [NiFe]-hydrogenases known to contain the Ni-A state. However, this proposition has been challenged based on the stability of this state to UV light exposure and the possibility of generating it anaerobically under either chemical or electrochemical oxidizing conditions. Consequently, we have considered alternative structures for the Ni-A species including oxidation of thiolate ligands to either sulfenate or sulfenic acid. Here, we report both new and revised [NiFe]-hydrogenases structures and conclude, taking into account corresponding characterizations by Fourier transform infrared spectroscopy (FTIR), that the Ni-A species contains oxidized cysteine and bridging hydroxide ligands instead of the peroxide ligand we proposed earlier. Our analysis was rendered difficult by the typical formation of mixtures of unready oxidized states that, furthermore, can be reduced by X-ray induced photoelectrons. The present study could be carried out thanks to the use of Desulfovibrio fructosovorans [NiFe]-hydrogenase mutants with special properties. In addition to the Ni-A state, crystallographic results are also reported for two diamagnetic unready states, allowing the proposal of a revised oxidized inactive Ni-SU model and a new structure characterized by a persulfide ion that is assigned to an Ni-‘Sox’ species.




Similar content being viewed by others
Abbreviations
- Av :
-
Allochromatium vinosum
- Df :
-
Desulfovibrio fructosovorans
- DvH:
-
Desulfovibrio vulgaris Hildenborough
- DvMF:
-
Desulfovibrio vulgaris Miyazaki F
- MSR:
-
Methionine sulfoxide reductase
- WT:
-
Wild type
References
Vignais PM, Billoud B (2007) Chem Rev 107:4206–4272. doi:10.1021/cr050196r
Fritsch J, Scheerer P, Frielingsdorf S, Kroschinsky S, Friedrich B, Lenz O, Spahn CMT (2011) Nature 479:249–252. doi:10.1038/nature10505
Shomura Y, Yoon KS, Nishihara H, Higuchi Y (2011) Nature 479:253–256. doi:10.1038/nature10504
Volbeda A, Amara P, Darnault C, Mouesca JM, Parkin A, Roessler MM, Armstrong FA, Fontecilla-Camps JC (2012) Proc Natl Acad Sci USA 109:5305–5310. doi:10.1073/pnas.1119806109
Evans RM, Parkin A, Roessler MM, Murphy BJ, Adamson H, Lukey MJ, Sargent F, Volbeda A, Fontecilla-Camps JC, Armstrong FA (2013) J Am Chem Soc 135:2694–2707. doi:10.1021/ja311055d
Mouesca J-M, Fontecilla-Camps JC, Amara P (2013) Angew Chem Int Ed Engl 52:2002–2006. doi:10.1073/pnas.1302304110
Lauterbach L, Lenz O (2013) J Am Chem Soc 135:17897–17905. doi:10.1021/ja408420d
Volbeda A, Amara P, Iannello M, DeLacey AL, Cavazza C, Fontecilla-Camps JC (2013) Chem Comm 49:7061–7063. doi:10.1039/c3cc43619e
Fontecilla-Camps JC, Volbeda A, Cavazza C, Nicolet Y (2007) Chem Rev 107:4273–4303. doi:10.1021/cr050195z
Volbeda A, Martin L, Cavazza C, Matho M, Faber BW, Roseboom W, Albracht SPJ, Garcin E, Rousset M, Fontecilla-Camps JC (2005) J Biol Inorg Chem 10:239–249. doi:10.1007/s00775-005-0632-x
Fernandez VM, Hatchikian EC, Cammack R (1985) Biochim Biophys Acta 832:69–79. doi:10.1016/0167-4838(85)90175-X
Van Gastel M, Stein M, Brecht M, Schröder O, Lendzian F, Bittl R, Ogata H, Higuchi Y, Lubitz W (2006) J Biol Inorg Chem 11:41–51. doi:10.1007/s00775-005-0048-7
Lamle SE, Albracht SPJ, Armstrong FA (2004) J Am Chem Soc 126:14899–14909. doi:10.1021/ja047939v
Van der Zwaan JW, Coremans JM, Bouwens EC, Albracht SPJ (1990) Biochim Biophys Acta 1041:101–110. doi:10.1016/0167-4838(90)90051-G
Carepo M, Tierney DL, Brondino CD, Yang TC, Pamplona A, Telser J, Moura I, Moura JJG, Hoffman BM (2002) J Am Chem Soc 124:281–286. doi:10.1021/ja010204v
Ogata H, Hirota S, Nakahara A, Komori H, Shibata N, Kato T, Kano K, Higuchi Y (2005) Structure 13:1635–1642. doi:10.1016/j.str.2005.07.018
Söderhjelm P, Ryde U (2006) J Mol Struct (THEOCHEM) 770:199–219. doi:10.1016/j.theochem.2006.06.008
Ogata H, Kellers P, Lubitz W (2010) J Mol Biol 420:428–444. doi:10.1016/j.jmb.2010.07.041
AbouHamdan A, Burlat B, Gutiérrez-Sanz O, Liebgott P-P, Baffert C, De Lacey AL, Rousset M, Guigliarelli B, Léger C, Dementin S (2013) Nat Chem Biol 9:15–17. doi:10.1038/nchembio.1110
De Lacey AL, Hatchikian EC, Volbeda A, Frey M, Fontecilla-Camps JC, Fernandez VM (1997) J Am Chem Soc 119:7181–7189. doi:10.1021/ja963802w
Ravelli RBG, Garman EF (2006) Curr Opin Struct Biol 16:624–629. doi:10.1016/j.sbi.2006.08.001
Bleijlevens B, Van Broekhuizen FA, De Lacey AL, Roseboom W, Fernandez VM, Albracht SPJ (2004) J Biol Inorg Chem 9:743–752. doi:10.1007/s00775-004-0570-z
Vincent KA, Belsey NA, Lubitz W, Armstrong FA (2006) J Am Chem Soc 128:7448–7449. doi:10.1021/ja061732f
De Lacey AL, Fernández VM, Rousset M, Cavazza C, Hatchikian EC (2003) J Biol Inorg Chem 8:129–134. doi:10.1007/s00775-002-0397-4
Rousset M, Montet Y, Guigliarelli B, Forget N, Asso M, Bertrand P, Fontecilla-Camps JC, Hatchikian EC (1998) Proc Natl Acad Sci USA 95:11625–11630. doi:10.1073/pnas.95.20.11625
Hatchikian CE, Traore AS, Fernandez VM, Cammack R (1990) Eur J Biochem 187:635–643. doi:10.1111/j.1432-1033.1990.tb15347.x
Dementin S, Burlat B, De Lacey AL, Pardo A, Adryanczyk-Perrier G, Guigliarelli B, Fernandez VM, Rousset M (2004) J Biol Chem 279:10508–10513. doi:10.1074/jbc.M312716200
Leroux F, Dementin S, Burlat B, Cournac L, Volbeda A, Champ S, Martin L, Guigliarelli B, Bertrand P, Fontecilla-Camps JC, Rousset M, Léger C (2008) Proc Natl Acad Sci USA 105:11188–11193. doi:10.1073/pnas.0803689105
Liebgott P-P, Leroux F, Burlat B, Dementin S, Baffert C, Lautier T, Fourmond V, Ceccaldi P, Cavazza C, Meynial-Salles I, Soucaille P, Fontecilla-Camps JC, Guigliarelli B, Bertrand P, Rousset M, Léger C (2010) Nat Chem Biol 6:63–70. doi:10.1038/nchembio.276
Vernède X, Fontecilla-Camps JC (1999) J Appl Cryst 32:505–509. doi:10.1107/S0021889899002678
Kabsch W (2010) Acta Crystallogr Sect D Biol Crystallogr 66:125–132. doi:10.1107/S0907444909047337
Karplus PA, Diederichs K (2012) Science 336:1030–1033. doi:10.1126/science.1218231
Winn MD, Ballard CC, Cowtan KD, Dodson EJ, Emsley P, Evans PR, Keegan RM, Krissinel EB, Leslie AG, McCoy A, McNicholas SJ, Murshudov GN, Pannu NS, Potterton EA, Powell HR, Read RJ, Vagin A, Wilson KS (2011) Acta Crystallogr Sect D Biol Crystallogr 67:235–242. doi:10.1107/S0907444910045749
McCoy AJ, Grosse-Kunstleve RW, Adams PD, Winn MD, Storoni LC, Read RJ (2007) J Appl Cryst 40:658–674. doi:10.1107/S0021889807021206
Murshudov GN, Skubák P, Lebedev AA, Pannu NS, Steiner RA, Nicholls RA, Winn MD, Long F, Vagin AA (2011) Acta Crystallogr Sect D Biol Crystallogr 67:355–367. doi:10.1107/S0907444911001314
Emsley P, Lohkamp B, Scott WG, Cowtan K (2010) Acta Crystallogr Sect D Biol Crystallogr 66:486–501. doi:10.1107/S0907444910007493
De Lacey AL, Fernández VM, Rousset M, Cammack R (2007) Chem Rev 107:4304–4330. doi:10.1021/cr0501947
Van Gastel M (2010) Appl Magn Reson 37:207–218. doi:10.1007/s00723-009-0044-0
Wood PM (1988) Biochem J 253:287–289
Volbeda A, Montet Y, Vernède X, Hatchikian EC, Fontecilla-Camps JC (2002) Int J Hydrogen Energy 27:1449–1461. doi:10.1016/S0360-3199(02)00072-1
Kumar M, Colpas GJ, Day RO, Maroney MJ (1989) J Am Chem Soc 111:8323–8325. doi:10.1021/ja00203a068
Farmer PJ, Verpeaux J-N, Amatore C, Darensbourg MY, Musie G (1994) J Am Chem Soc 116:9355–9356. doi:10.1021/ja00099a073
Marques MC, Coelho R, Pereira IAC, Matias PM (2013) Int J Hydrogen Energy 38:8664–8682. doi:10.1016/j.ijhydene.2013.04.132
Pandelia M-E, Ogata H, Lubitz W (2010) Chem Phys Chem 11:1127–1140. doi:10.1002/cphc.200900950
Claiborne A, Yeh JI, Mallet TC, Luba J, Crane EJ, Charrier V, Parsonage D (1999) Biochemistry 38:15407–15416. doi:10.1021/bi992025k
Boschi-Muller S, Gand A, Branlant G (2008) Arch Biochem Biophys 474:266–273. doi:10.1016/j.abb.2008.02.007
Tarrago L, Laugier E, Zaffagnini M, Marchand CH, Le Maréchal P, Lemaire SD, Rey P (2010) J Biol Chem 285:14964–14972. doi:10.1074/jbc.M110.108373
Nagahara N, Katayama A (2005) J Biol Chem 280:34569–34576. doi:10.1074/jbc.M505643200
Liebgott P-P, de Lacey AL, Burlat B, Cournac L, Richaud P, Brugna M, Fernandez VM, Guigliarelli B, Rousset M, Léger C, Dementin S (2011) J Am Chem Soc 133:986–997. doi:10.1021/ja108787s
Marques MC, Coelho R, De Lacey AL, Pereira IAC, Matias PM (2010) J Mol Biol 396:893–907. doi:10.1016/j.jmb.2009.12.013
Brunger AT (2010) Nature 355:473–475. doi:10.1038/355472a0
Acknowledgments
We thank the Commissariat à l’Energie Atomique et aux Energies Alternatives (CEA) and the Centre National de la Recherche Scientifique (CNRS) for institutional funding, the Direction des Sciences du Vivant of the CEA for funding through the Bioenergy Program, the Agence Nationale de Recherche for further financial support (project Hyliox, contract n° ANR-07-BIOE-010) and the staff at the used ESRF beamlines for assistance with X-ray data collections. ALDL and OG-S thank the Spanish MINECO for financial support (project CTQ2012-32448) and a FPI grant, respectively. In addition, P-PL, SD and MR thank the Pôle de Compétitivité Capénergies for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
An Interactive 3D Complement page in Proteopedia is available at: http://proteopedia.org/wiki/index.php/Journal:JBIC:28
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Volbeda, A., Martin, L., Barbier, E. et al. Crystallographic studies of [NiFe]-hydrogenase mutants: towards consensus structures for the elusive unready oxidized states. J Biol Inorg Chem 20, 11–22 (2015). https://doi.org/10.1007/s00775-014-1203-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-014-1203-9