Abstract
The aim of this work was to clarify the effect of the position of the hydroxyl group on the antioxidant capacity of hydroxyferrocifen by means of a chemical kinetic method. Propionylferrocene and benzoylferrocene condensed with 4-hydroxydiphenylketone, 3,4-dihydroxydiphenylketone, and 4,4′-dihydroxydiphenylketone to form FP3, FP4, FB3, and FB4 with a single hydroxyl group and FP34, FP44, FB34, and FB44 with two hydroxyl groups. These hydroxyferrocifens were applied in Cu2+/glutathione (GSH)-induced, hydroxyl radical (·OH)-induced, and 2,2′-azobis(2-amidinopropane hydrochloride) (AAPH)-induced oxidation of DNA, and in trapping 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonate) cationic radical (ABTS+·). It was found that these hydroxyferrocifens acted as prooxidants in Cu2+/GSH-induced oxidation of DNA and exhibited very weak effects on ·OH-induced oxidation of DNA. FP3, FP4, FB3, and FB4 can only retard the rate of AAPH-induced oxidation of DNA, whereas FP44, FB44, FB34, and FP34 can trap 11.9, 7.1, 6.2, and 4.9 radicals, respectively, in AAPH-induced oxidation of DNA. The ability to trap ABTS+· followed the order FB4 > FP44 > FB34 > FB44 > FP34. It was concluded that two hydroxyl groups at the para position of two benzene rings rather than at the ortho position in the same benzene ring were beneficial for hydroxyferrocifen to increase the antioxidant capacity.
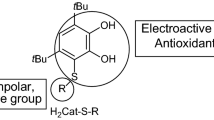
Graphical abstract
Antioxidant effectiveness of hydroxyferrocifen derivatives were evaluated in the experimental systems of Cu2+/glutathione-induced, ·OH-induced, and 2,2′-azobis(2-amidinopropane hydrochloride) induced oxidation of DNA and of trapping 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonate) cationic radical








Similar content being viewed by others
References
Allardyce CS, Dorcier A, Scolaro C, Dyson PJ (2005) Appl Organomet Chem 19:1–10
van Staveren DR, Metzler-Nolte N (2004) Chem Rev 104:5931–5985
Fouda MFR, Abd-Elzaher MM, Abdelsamaia RA, Labib AA (2007) Appl Organomet Chem 21:613–625
Top S, Tang J, Vessières A, Carrez D, Provot C, Jaouen G (1996) Chem Commun 955–956
Top S, Vessières A, Leclercq G, Quivy J, Tang J, Vaissermann J, Huché M, Jaouen G (2003) Chem Eur J 9:5223–5236
Top S, Vessières A, Cabestaing C, Laïos I, Leclercq G, Provot C, Jaouen G (2001) J Organomet Chem 637–639:500–506
Hillard EA, Vessières A, Jaouen G (2010) Top Organomet Chem 32:81–117
Pigeon P, Top S, Vessières A, Huché M, Hillard EA, Salomon E, Jaouen G (2005) J Med Chem 48:2814–2821
Nikitin K, Ortin Y, Müller-Bunz H, Plamont M-A, Jaouen G, Vessières A, McGlinchey MJ (2010) J Organomet Chem 695:595–608
Zanellato I, Heldt J-M, Vessières A, Jaouen G, Osella D (2009) Inorg Chim Acta 362:4037–4042
Vessières A, Corbet C, Heldt JM, Lories N, Jouy N, Laïos I, Leclercq G, Jaouen G, Toillon R-A (2010) J Inorg Biochem 104:503–511
Hamels D, Dansette PM, Hillard EA, Top S, Vessiéres A, Herson P, Jaouen G, Mansuy D (2009) Angew Chem Int Ed 48:9124–9126
Hillard EA, Pigeon P, Vessières A, Amatore C, Jaouen G (2007) Dalton Trans 5073–5081
Buriez O, Heldt JM, Labbé E, Vessiéres A, Jaouen G, Amatore C (2008) Chem Eur J 14:8195–8203
Bowry VW, Stocker R (1993) J Am Chem Soc 115:6029–6044
Li Y-F, Liu Z-Q, Luo X-Y (2010) J Agric Food Chem 58:4126–4131
Munoz-Munoz JL, Garcia-Molina F, Varon R, Tudela J, García-Cánovas F, Rodriguez-Lopez JN (2010) J Agric Food Chem 58:2062–2070
Top S, Dauer B, Vaissermann J, Jaouen G (1997) J Organomet Chem 541:355–361
Allard E, Passirani C, Garcion E, Pigeon P, Vessières A, Jaouen G, Benoit J-P (2000) C R Acad Sci Ser IIc 3:89–93
Reed CJ, Douglas KT (1991) Biochem J 275:601–608
Janicek MF, Haseltine WA, Henner WD (1985) Nucleic Acids Res 13:9011–9029
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Free Radic Biol Med 26:1231–1237
Maity B, Roy M, Banik B, Majumdar R, Dighe RR, Chakravarty AR (2010) Organometallics 29:3632–3641
Prütz WA (1994) Biochem J 302:373–382
Battin EE, Brumaghim JL (2008) J Inorg Biochem 102:2036–2042
Li P-Z, Liu Z-Q (2011) Eur J Med Chem 46:1821–1826
Zheng L-F, Dai F, Zhou B, Yang L, Liu Z-L (2008) Food Chem Toxicol 46:149–156
Hillard E, Vessiéres A, Thouin L, Jaouen G, Amatore C (2006) Angew Chem 118:291–296
Zhu B-Z, Kitrossky N, Chevion M (2000) Biochem Biophys Res Commun 270:942–946
Vessières A, Top S, Pigeon P, Hillard E, Boubeker L, Spera D, Jaouen G (2005) J Med Chem 48:3937–3940
Zhang P, Omaye ST (2001) Food Chem Toxicol 39:239–246
Li G-X, Liu Z-Q, Wu D (2009) J Phys Org Chem 22:883–887
Acknowledgments
Financial support from the Jilin Provincial Foundation for Natural Science, China (201115017), is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, F., Zhao, C. & Liu, ZQ. Synthesis of hydroxyferrocifen and its abilities to protect DNA and to scavenge radicals. J Biol Inorg Chem 16, 1169–1176 (2011). https://doi.org/10.1007/s00775-011-0805-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-011-0805-8