Abstract
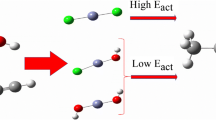
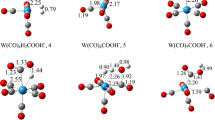
The reaction mechanism of the hydration of acetylene to acetaldehyde catalyzed by [WIVO(mnt)2]2− (where mnt2− is 1,2-dicyanoethylenedithiolate) is studied using density functional theory. Both the uncatalyzed and the catalyzed reaction are considered to find out the origin of the catalysis. Three different models are investigated, in which an aquo, a hydroxo, or an oxo coordinates to the tungsten center. A first-shell mechanism is suggested, similarly to recent calculations on tungsten-dependent acetylene hydratase. The acetylene substrate first coordinates to the tungsten center in an η2 fashion. Then, the tungsten-bound hydroxide activates a water molecule to perform a nucleophilic attack on the acetylene, resulting in the formation of a vinyl anion and a tungsten-bound water molecule. This is followed by proton transfer from the tungsten-bound water molecule to the newly formed vinyl anion intermediate. Tungsten is directly involved in the reaction by binding and activating acetylene and providing electrostatic stabilization to the transition states and intermediates. Three other mechanisms are also considered, but the associated energetic barriers were found to be very high, ruling out those possibilities.










Similar content being viewed by others
References
Rosner BM, Schink B (1995) J Bacteriol 177:5767–5772
Schink B (1985) Arch Microbiol 142:295–301
Einsle O, Niessen H, Abt DJ, Seiffert G, Schink B, Huber R, Messerschmidt A, Kroneck PMH (2005) Acta Crystallogr F 61:299–301
Seiffert GB, Ullmann GM, Messerschmidt A, Schink B, Kroneck PMH, Einsle O (2007) Proc Natl Acad Sci USA 104:3073–3077
Antony S, Bayse CA (2009) Organometallics 28:4938–4944
Vincent MA, Hillier IH, Periyasamy G, Burton NA (2010) Dalton Trans 39:3816–3822
Liao RZ, Yu JG, Himo F (2010) Proc Natl Acad Sci USA 107:22523–22527
Gavrilova AL, Bosnich B (2004) Chem Rev 104:349
Das SK, Biswas D, Maiti R, Sarkar S (1996) J Am Chem Soc 118:1387–1397
Sugimoto H, Tsukube H (2008) Chem Soc Rev 37:2609–2619
Das SK, Biswas D, Chaudhury PK, Sarkar S (1994) J Am Chem Soc 116:9061
Ueyama N, Oku H, Nakamura A (1992) J Am Chem Soc 114:7310–7311
Yadav J, Das SK, Sarkar S (1997) J Am Chem Soc 119:4315–4316
Frisch MJ et al (2003) Gaussian 03, revision D.01. Gaussian, Pittsburgh
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Ghosh P, Bill E, Weyhermuller T, Neese F, Wieghardt K (2003) J Am Chem Soc 125:1293
Siegbahn PEM (2003) Q Rev Biophys 36:91–145
Noodleman L, Lovell T, Han WG, Li J, Himo F (2004) Chem Rev 104:459–508
Neese F (2003) Curr Opin Chem Biol 7:125
Borowski T, Bassan A, Siegbahn PEM (2004) Inorg Chem 43:3277–3291
Bassan A, Blomberg MRA, Siegbahn PEM, Que L Jr (2005) Angew Chem Int Ed 44:2939–2941
Lundberg M, Siegbahn PEM (2005) J Comput Chem 26:661–667
Bergner A, Dolg M, Kuechle W, Stoll H, Preuss H (1993) Mol Phys 80:1431
Barone V, Cossi M (1998) J Phys Chem A 102:1995–2001
Cammi R, Mennucci B, Tomasi J (1999) J Phys Chem A 103:9100
Klamt A, Schmmrmann G (1993) J Chem Soc Perkin Trans 2:799–805
Tomasi J, Mennucci B, Cammi (2005) Chem Rev 105:2999–3094
Gonzalez C, Schlegel HB (1989) J Chem Phys 90:2154–2161
Gonzalez C, Schlegel HB (1990) J Phys Chem 94:5523–5527
Zhao Y, Lynch BJ, Truhlar DG (2004) J Phys Chem A 108:2715–2719
Zhao Y, Truhlar DG (2008) Theor Chem Acc 120:215–241
Zhao Y, Truhlar DG (2008) Acc Chem Res 41:157–167
Frisch MJ et al (2009) Gaussian 09, revision A.02, Gaussian, Wallingford
Liao RZ, Ding WJ, Yu JG, Fang WH, Liu RZ (2007) J Phys Chem A 111:3184–3190
Guo JX, Ho JJ (1999) J Phys Chem A 103:6433–6441
Kim Y, Lim S, Kim HJ, Kim Y (1999) J Phys Chem A 103:617–624
Sun Y, Li H, Liang WC, Han SJ (2005) J Phys Chem B 109:5919–5926
Hu XB, Li HR, Liang WC, Han SJ (2004) J Phys Chem B 108:12999–13007
Li QS, Fang WH, Yu JG (2005) J Phys Chem A 109:3983–3990
Xue Y, Kim CK, Guo Y, Xie DQ, Yan GS (2005) J Comput Chem 26:994–1005
Sun XM, Wei XG, Wu XP, Ren Y, Wong NB, Li WK (2010) J Phys Chem A 114:595–602
Jaramillo P, Coutinho K, Canuto S (2009) J Phys Chem A 113:12485–12495
Catak S, Monard G, Aviyente V, Ruiz-López MF (2009) J Phys Chem A 113:1111–1120
Nguyen MT, Matus MH, Jackson VE, Ngan VT, Rustad JR, Dixon DA (2008) J Phys Chem A 112:10386–10398
Erdtman E, Eriksson LA (2008) J Phys Chem A 112:4367–4374
Lima MCP, Coutinho K, Canuto S, Rocha WR (2006) J Phys Chem A 110:7253–7261
Chen HT, Chang JG, Chen HL (2008) J Phys Chem A 112:8093–8099
Liao RZ, Yu JG, Raushel FM, Himo F (2008) Chem Eur J 14:4287–4292
Liao RZ, Himo F, Yu JG, Liu RZ (2010) J Inorg Biochem 104:37–46
Leopoldini M, Toscano M, Russo N (2007) J Am Chem Soc 129:7776–7784
Szaleniec M, Borowski T, Schühle K, Witko M, Heider J (2010) Am Chem Soc 132:6014–6024
Metz S, Wang DQ, Thiel W (2009) J Am Chem Soc 131:628–4640
Metz S, Thiel W (2005) J Am Chem Soc 131:14885–14902
Yang L, Liao RZ, Yu JG, Liu RZ (2009) J Phys Chem B 113:6505–6510
Prabhakar R, Morokuma K, Musaev DG (2006) J Comput Chem 27:438–1445
Acknowledgments
We gratefully acknowledge Margareta Blomberg (Stockholm University) for her critical comments on this manuscript. We appreciate Sven de Marothy (Stockholm University) providing the XYZ viewer which helped us to create all the figures of the molecule models. This work was supported by grants from the National Natural Science Foundation of China (grants 20573011, 20733002, and 20873008) and Major State Basic Research Development Programs (grant 2004CB719903).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, YF., Liao, RZ., Ding, WJ. et al. Theoretical investigation of the first-shell mechanism of acetylene hydration catalyzed by a biomimetic tungsten complex. J Biol Inorg Chem 16, 745–752 (2011). https://doi.org/10.1007/s00775-011-0775-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-011-0775-x