Abstract
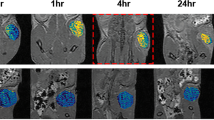
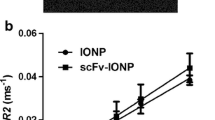
A novel magnetic resonance imaging (MRI) contrast agent containing Herceptin is reported. The surfaces of superparamagnetic iron oxide nanoparticles were modified with dextran and conjugated with Herceptin (Herceptin–nanoparticles) to improve their dispersion, magnetization, and targeting of the specific receptors on cells. From analytical results, we found that Herceptin–nanoparticles were well dispersed in solutions of various pH range, and had no hysteresis, high saturation magnetization (80 emu/g), and low cytotoxicity to a variety of cells. Notably, the magnetic resonance enhancements for the different breast cancer cell lines (BT-474, SKBR-3, MDA-MB-231, and MCF-7) are proportional to the HER2/neu expression level in vitro. When Herceptin–nanoparticles were administered to mice bearing breast tumor allograft by intravenous injection, the tumor site was detected in T 2-weighted magnetic resonance images as a 45% enhancement drop, indicating a high level of accumulation of the contrast agent within the tumor sites. Therefore, targeting of cancer cells was observed by in vitro and in vivo MRI studies using Herceptin–nanoparticles contrast agent. In addition, Herceptin–nanoparticles enhancing the magnetic resonance signal intensity were sufficient to detect the cell lines with a low level of HER2/neu expression.







Similar content being viewed by others
References
Sun C, Veiseh O, Gunn J, Fang C, Hansen S, Lee D, Sze R, Ellenbogen RG, Olson J, Zhang M (2008) Small 4:240–249
Schellenberger E, Schnorr J, Reutelingsperger C, Ungethüm L, Meyer W, Taupitz M, Hamm B (2008) Small 4:225–230
Li Z, Tan B, Allix M, Cooper AI, Rosseinsky MJ (2008) Small 4:231–239
Kalambur VS, Longmire EK, Bischof JC (2007) Langmuir 23:12329–12336
Gupta AK, Gupta M (2005) Biomaterials 26:3995–4021
Yigit MV, Mazumdar D, Kim H-K, Lee JH, Odintsov B, Lu Y (2007) Chembiochem 8:1675–1678
Paul KG, Frigo TB, Groman JY, Groman EV (2004) Bioconjug Chem 15:194–401
Koch AM, Reynolds F, Kircher MF, Merkle HP, Weissleder R, Josephson L (2003) Bioconjug Chem 14:1115–1121
Josephson L, Kircher MF, Mahmood U, Tang Y, Weissleder R (2002) Bioconjug Chem 13:554–560
Högemann D, Josephson L, Weissleder R, Basilion JP (2000) Bioconjug Chem 11:941–946
Lee H, Yu MK, Park S, Moon S, Min JJ, Jeong YY, Kang H-W, Jon S (2007) J. Am Chem Soc 129:12739–12745
Choi H, Choi SR, Zhou R, Kung HF, Chen IW (2004) Acad Radiol 11:996–1004
Tran T, Engfeldt T, Orlova A, Sandström M, Feldwisch J, Abrahmsén L, Wennborg A, Tolmachev V, Karlström AE (2007) Bioconjug Chem 18:1956–1964
Germershaus O, Merdan T, Bakowsky U, Behe M, Kissel T (2006) Bioconjug Chem 17:1190–1199
Shukla R, Thomas TP, Peters JL, Desai AM, Kukowska-Latallo J, Patri AK, Kotlyar A, Baker JR (2006) Bioconjug Chem 17:1109–1115
Hilger I, Leistner Y, Berndt A, Fritsche C, Haas KM, Kosmehl H, Kaiser WA (2004) Eur Radiol 14:1124–1129
Pan M-H, Lin C-C, Lin J-K, Chen W-J (2007) J Agric Food Chem 55:5030–5037
Suna B, Ranganathana B, Feng S-S (2008) Biomaterials 29:475–486
Bergman I, Whitaker-Dowling P, Gao Y, Griffin JA, Watkinse SC (2003) Virology 316:337–347
Colbern GT, Hiller AJ, Musterer RS, Working PK, Henderson IC (1999) J Inorg Biochem 77:117–120
Meares CF, Chmura AJ, Orton MS, Corneillie TM, Awhetstone P (2003) J Mol Recognit 16:255–259
Garmestania K, Milenica DE, Plascjakb PS, Brechbiel MW (2002) Nucl Med Biol 29:599–606
Chen T-J, Cheng T-H, Hung Y-C, Lin K-T, Liuc G-C, Wang Y-M (2008) J Biomed Mater Res A 87:165–175
Taktak S, Sosnovik D, Cima MJ, Weissleder R, Josephson L (2007) Anal Chem 79:8863–8869
Casula MF, Y-w Jun, Zaziski DJ, Chan EM, Corrias A, Alivisatos AP (2006) J Am Chem Soc 128:1675–1682
Park J, Lee E, Hwang N-M, Kang M, Kim SC, Hwang Y, Park J-G, Noh H-J, Kim J-Y, Park J-H, Hyeon T (2005) Angew Chem Int Ed 44:2872–2877
Sun S, Zeng H (2002) J Am Chem Soc 124:8204–8205
Weissleder R, Elizondo G, Wittenberg J, Rabito CA, Bengele HH, Josephson L (1990) Radiology 175:485–493
Lee H, Lee E, Kim DK, Jang NK, Jeong YY, Jon S (2006) J Am Chem Soc 128:7383–7389
Cheng F-Y, Su C-H, Yang Y-S, Yeh C-S, Tsai C-Y, Wu C-L, Wu M-T, Shieh D-B (2005) Biomaterials 26:729–738
Shieh D-B, Cheng F-Y, Su C-H, Yeh C-S, Wu M-T, Wu Y-N, Tsai C-Y, Wu C-L, Chen D-H, Chou C-H (2005) Biomaterials 26:7183–7191
Varanda LC, Jafelicci M, Tartaj P, O’Grady K, González-Carreño T, Morales MP, Muñoz T, Serna CJ (2002) J Appl Phys 92:2079–2085
Jun Y-W, Huh Y-M, Choi J-S, Lee J-H, Song H-T, Kim S, Yoon S, Kim K-S, Shin J-S, Suh J-S, Cheon J (2005) J Am Chem Soc 127:5732–5733
Huh Y-M, Jun Y-W, Song H-T, Kim S, Choi J-S, Lee J-H, Yoon S, Kim K-S, Shin J-S, Suh J-S, Cheon J (2005) J Am Chem Soc 127:12387–12391
Horák D, Babič M, Jendelová P, Herynek V, Trchova M, Pientka Z, Pollert E, Hájek M, Syková E (2007) Bioconjug Chem 8:635–644
Sturla SJ, Irwin JJ, Loeppky RN, Mulvihill MJ, Searcey M (2007) ACS Chem Biol 2:286–292
Lee J-H, Huh Y-M, Jun Y-W, Seo J-W, Jang J-T, Song H-T, Kim S, Cho E-J, Yoon H-G, Suh J-S, Cheon J (2006) Nat Med 13:95–99
Acknowledgements
We are grateful to the National Science Council of the Republic of China for financial support under contracts no. NSC 96-2627-M-037-001 and NSC 97-2623-7-037-001-NU. This research was also supported in part by grants from National Health Research Institutes under contract no. NHRI-EX-95-9424EI.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, TJ., Cheng, TH., Chen, CY. et al. Targeted Herceptin–dextran iron oxide nanoparticles for noninvasive imaging of HER2/neu receptors using MRI. J Biol Inorg Chem 14, 253–260 (2009). https://doi.org/10.1007/s00775-008-0445-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-008-0445-9