Abstract
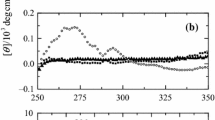
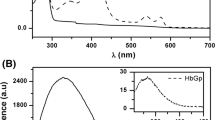
Human serum albumin (HSA) participates in heme scavenging, the bound heme turning out to be a reactivity center and a powerful spectroscopic probe. Here, the reversible unfolding of heme–HSA has been investigated by 1H-NMR relaxometry, circular dichroism, and absorption spectroscopy. In the presence of 6 equiv of myristate (thus fully saturating all available fatty acid binding sites in serum heme–albumin), 1.0 M guanidinium chloride induces some unfolding of heme–HSA, leading to the formation of a folding intermediate; this species is characterized by increased relaxivity and enhanced dichroism signal in the Soret region, suggesting a more compact heme pocket conformation. Heme binds to the folding intermediate with K d = (1.2 ± 0.1) × 10−6 M. In the absence of myristate, the conformation of the folding intermediate state is destabilized and heme binding is weakened [K d = (3.4 ± 0.1) × 10−5 M]. Further addition of guanidinium chloride (up to 5 M) brings about the usual denaturation process. In conclusion, myristate protects HSA from unfolding, stabilizing a folding intermediate state in equilibrium with the native and the fully unfolded protein, envisaging a two-step unfolding pathway for heme–HSA in the presence of myristate.





Similar content being viewed by others
Abbreviations
- B:
-
Basic
- CD:
-
Circular dichroism
- F:
-
Fast migrating
- FA:
-
Fatty acid
- GnCl:
-
Guanidinium chloride
- HSA:
-
Human serum albumin
- N:
-
Neutral
References
Carter DC, Ho JX (1994) Adv Protein Chem 45:153–203
Peters T Jr (1996) All about albumin: biochemistry, genetics and medical applications. Academic Press, Orlando
Curry S (2002) Vox Sang 83:315–319
Fasano M, Curry S, Terreno E, Galliano M, Fanali G, Narciso P, Notari S, Ascenzi P (2005) IUBMB Life 57:787–796
Ascenzi P, Bocedi A, Notari S, Fanali G, Fesce R, Fasano M (2006) Mini Rev Med Chem 6:483–489
Spector AA (1975) J Lipid Res 16:165–179
Hamilton JA (2004) Prog Lipid Res 43:177–199
Simard JR, Zunszain PA, Ha CE, Yang JS, Bhagavan NV, Petitpas I, Curry S, Hamilton JA (2005) Proc Natl Acad Sci USA 102:17958–17963
Kragh-Hansen U, Watanabe H, Nakajou K, Iwao Y, Otagiri M (2006) J Mol Biol 363:702–712
Simard JR, Zunszain PA, Hamilton JA, Curry S (2006) J Mol Biol 361:336–351
Fasano M, Baroni S, Vannini A, Ascenzi P, Aime S (2001) J Biol Inorg Chem 6:650–658
Wardell MZ, Wang JX, Ho J, Robert J, Rüker F, Ruble J, Carter DC (2002) Biochem Biophys Res Commun 291:813–819
Zunszain PA, Ghuman J, Komatsu T, Tsuchida E, Curry S (2003) Struct Biol 3:6
Fasano M, Fanali G, Leboffe L, Ascenzi P (2007) IUBMB Life 59:436–440
Fasano M, Fanali G, Fesce R, Ascenzi P (2008) In: Bolognesi M, di Prisco G, Verde C (eds) Dioxygen binding and sensing proteins. Springer, Heidelberg, pp 121–131
Baroni S, Mattu M, Vannini A, Cipollone R, Aime S, Ascenzi P, Fasano M (2001) Eur J Biochem 268:6214–6220
Mattu M, Vannini A, Coletta M, Fasano M, Ascenzi P (2001) J Inorg Biochem 84:293–296
Fasano M, Mattu M, Coletta M, Ascenzi P (2002) J Inorg Biochem 91:487–490
Monzani E, Curto M, Galliano M, Minchiotti L, Aime S, Baroni S, Fasano M, Amoresano A, Salzano AM, Pucci P, Casella L (2002) Biophys J 83:2248–2258
Fanali G, Fesce R, Agrati C, Ascenzi P, Fasano M (2005) FEBS J 272:4672–4683
Fanali G, Bocedi A, Ascenzi P, Fasano M (2007) FEBS J 274:4491–4502
Wyman J Jr (1964) Adv Protein Chem 19:223–286
Yamasaki K, Maruyama T, Yoshimoto K, Tsutsumi Y, Narazaki R, Fukuhara A, Kragh-Hansen U, Otagiri M (1999) Biochim Biophys Acta 1432:313–323
Ascenzi P, Bocedi A, Notari S, Menegatti E, Fasano M (2005) Biochem Biophys Res Commun 334:481–486
Dill K, Alonso DOV, Hutchinson K (1989) Biochemistry 28:5439–5449
Kosa T, Maruyama T, Sakai N, Yonemura N, Yahara S, Otagiri M (1998) Pharm Res 15:592–598
Farruggia B, Rodriguez F, Rigatuso R, Fidelio G, Picò G (2001) J Protein Chem 20:81–89
Santra MK, Banerjee A, Krishnakumar SS, Rahaman O, Panda D (2004) Eur J Biochem 271:1789–1797
Santra MK, Banerjee A, Rahaman O, Panda D (2005) Int J Biol Macromol 37:200–204
Ahmad B, Ankita, Khan RH (2005) Arch Biochem Biophys 437:159–167
Rezaei-Tavirani M, Moghaddamnia SH, Ranjbar B, Amani M, Marashi SA (2006) J Biochem Mol Biol 39:530–536
Fanali G, Ascenzi P, Fasano M (2007) Biophys Chem 129:29–35
Bradford MM (1976) Anal Biochem 72:248–254
Boffi A, Das TK, Della Longa S, Spagnuolo C, Rousseau DL (1999) Biophys J 77:1143–1149
Koenig SH, Brown RDIII (1990) Progr NMR Spectrosc 22:487–567
Bertini I, Luchinat C (1986) NMR of paramagnetic molecules in biological systems. Benjamin/Cummings, Menlo Park
Banci L, Bertini I, Luchinat C (1991) Nuclear and electron relaxation. VCH, Weinheim
Pace CN (1986) Methods Enzymol 131:266–280
Sugio S, Kashima A, Mochizuki S, Noda M, Kobayashi K (1999) Protein Eng 12:439–446
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng EC, Ferrin TE (2004) J Comput Chem 25:1605–1612
La Mar GN, Yamamoto Y, Jue T, Smith KM, Pandey RK (1985) Biochemistry 24:3826–3831
Santucci R, Ascoli F, La Mar GN, Pandey RK, Smith KM (1993) Biochim Biophys Acta 1164:133–137
Manning MC, Woody RW (1989) Biochemistry 28:8609–8613
Santucci R, Polizio F, Desideri A (1999) Biochimie 81:745–751
Ascenzi P, Bocedi A, Visca P, Altruda F, Tolosano E, Beringhelli T, Fasano M (2005) IUBMB Life 57:749–759
Myer YP, Pande A (1978) In: Dolphin D (ed) The porphyrins. Academic Press, New York, pp 271–322
Goto Y, Fink AL (1994) Methods Enzymol 232:3–15
Aime S, Dastrù W, Fasano M, Arnelli A, Castagnola M, Giardina B, Ascenzi P (1992) Clin Chem 38:2401–2404
Aime S, Fasano M, Paoletti S, Arnelli A, Ascenzi P (1995) Magn Reson Med 33:827–831
Aime S, Fasano M, Paoletti S, Cutruzzolà F, Desideri A, Bolognesi M, Rizzi M, Ascenzi P (1996) Biophys J 70:482–488
Fasano M, Baroni S, Aime S, Mattu M, Ascenzi P (2003) J Inorg Biochem 95:64–67
Muzammil S, Kumar Y, Tayyab S (1999) Eur J Biochem 266:26–32
Sattarahmady N, Moosavi-Movahedi AA, Ahmad F, Hakimelahi GH, Habibi-Rezaei M, Saboury AA, Sheibani N (2007) Biochim Biophys Acta 1770:933–942
Acknowledgment
The authors thank Giorgio Pariani for technical assistance. Grant MiUR FIRB RBNE03PX83 to M.C. is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fanali, G., De Sanctis, G., Gioia, M. et al. Reversible two-step unfolding of heme–human serum albumin: a 1H-NMR relaxometric and circular dichroism study. J Biol Inorg Chem 14, 209–217 (2009). https://doi.org/10.1007/s00775-008-0439-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-008-0439-7