Abstract
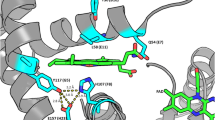
The crystal structure of Escherichia coli bacterioferritin has been solved to 1.9 Å, and shows the symmetrical binding of a haem molecule on the local twofold axis between subunits and a pair of metal atoms bound to each subunit at the ferroxidase centre. These metals have been identified as zinc by the analysis of the structure and X-ray data and confirmed by microfocused proton-induced X-ray emission experiments. For the first time the haem has been shown to be linked to both the internal and the external environments via a cluster of waters positioned above the haem molecule.






Similar content being viewed by others
References
Frolow F, Kalb A, Yariv J (1994) Nat Struct Biol 1:453–460
Dautant A, Meyer JB, Yariv J, Precigoux G, Sweet R, Kalb J, Frowlow F (1998) Acta Crystallogr D 54:16–24
van Eerde A, Wolterink-van Loo S, van der Oost J, Dijkstra BW (2006) Acta Crystallogr F 62:1061–1066
Stillman TJ, Hempstead PD, Artymiuk PJ, Andrews SC, Hudson AJ, Treffry A, Guest JR, Harrison PM (2001) J Mol Biol 307:587–603
Janowski R, Auerbach-Nevo T, Weiss MS (2008) Protein Sci 17:1138–1150
Guy J (2004) PhD thesis, University of Exeter
Laemmli U (1970) Nature 227:680–685
Cianci M, Antonyuk S, Bliss N, Bailey MW, Buffey SG, Cheung KC, Clarke JA, Derbyshire GE, Ellis MJ, Enderby MJ, Grant AF, Holbourn MP, Laundy D, Nave C, Ryder R, Stephenson P, Helliwell JR, Hasnain SS (2005) J Synchrotron Radiat 12:455–466
Otwinowski Z, Minor W (1997) Methods Enzymol 267:21839–21843
French GS, Wilson KS (1978) Acta Crystallogr A 34:517
Collaborative Computational Project, Number 4 (1994) Acta Crystallogr D 50:760–763
Cowtan K (1994) Joint CCP4/ESF-EACBM Newsl Protein Crystallogr 31:34–38
Grime GW (2004) In: Budnar M (ed) Tenth international conference on particle induced X-ray emission. Portoroz, Slovenia
Campbell JL, Hopman TL, Maxwell JA, Nejedly Z (2000) Nucl Instrum Methods Phys Res B 170:193–204
Garman EF, Grime GW (2005) Prog Biophys Mol Biol 89:173–205
Sheldrick GM (2008) Acta Crystallogr A 64:112–122
Emsley P, Cowtan K (2004) Acta Crystallogr D 60:2126–2132
Murshudov G, Vagin A, Dodson E (1997) Acta Crystallogr D 53:240–255
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) J Mol Biol 215:403–410
Vagin A, Teplyakov A (1997) J Appl Crystallogr 30:1022–1025
Kabsch W (1976) Acta Crystallogr A 32:922–923
Baaghil S, Thomson AJ, Moore GR, Le Brun NE (2002) J Chem Soc Dalton Trans 811–818
Baaghil S, Lewin A, Moore GR, Le Brun NE (2003) Biochemistry 42:14047–14056
Kadir FHA, Al-Massad FK, Moore GR (1992) Biochem J 282:867–870
Cromer DT (1983) J Appl Crystallogr 16:437–438
Le Brun NE, Andrews SC, Guest JR, Harrison PM, Moore GR, Thomson AJ (1995) Biochem J 312:385–392
Acknowledgments
We would like to thank J.E. Guy for cloning of the AKR enzyme and Inma Gomez-Morilla at the University of Surrey Ion Beam Centre for assistance with the microPIXE experiments. We would also like to thank Chirotech Technology Limited, Cambridge, UK, and the BBSRC for funding of a studentship to S.C.W. The Synchrotron Radiation Source, Daresbury, UK, is acknowledged for provision of data collection facilities on the NWSGC MAD10 beamline funded by BBSRC grants (719/B15474 and 719/REI20571) and an NWDA project award (N0002170). We thank the station scientist Mark Ellis for assistance with data collection.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Willies, S.C., Isupov, M.N., Garman, E.F. et al. The binding of haem and zinc in the 1.9 Å X-ray structure of Escherichia coli bacterioferritin. J Biol Inorg Chem 14, 201–207 (2009). https://doi.org/10.1007/s00775-008-0438-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-008-0438-8