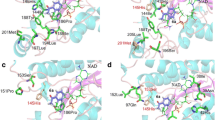
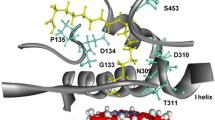
Abstract
CYP102A1 is a highly active water-soluble bacterial monooxygenase that contains both substrate-binding heme and diflavin reductase subunits, all in a single polypeptide that has been called a “self-sufficient enzyme.” Several years ago we developed a procedure called “scanning chimeragenesis,” where we focused on residues 73–82 of CYP102A1, which contact approximately 40% of the substrates palmitoleic acid and N-palmitoylglycine [Murataliev et al. (2004) Biochemistry 43:1771–1780]. These residues were replaced with the homologous residues of CYP4C7. In the current work, that study has been expanded to include residue 87. Phenylalanine 87 of wild-type CYP102A1 was replaced with the homologous residue of CYP4C7, leucine, as well as with alanine. The full-sized chimeric proteins C(73–78, F87L), C(73–78, F87A), C(75–80, F87L), C(75–80, F87A), C(78–82, F87L) and C(78–82, F87A) have been purified and characterized. Wild-type CYP102A1 is most active toward fatty acids (both lauric and palmitic acids produce ω-1, ω-2, and ω-3 hydroxylated fatty acids), but it also catalyzes the oxidation of farnesol to three products (2, 3- and 10,11-epoxyfarnesols and 9-hydroxyfarnesol). All of the F87-mutant chimeric proteins show dramatic decreases in activities with the natural CYP102A1 substrates. In contrast, C(78–82, F87A) and C(78–82, F87L) have markedly increased activities with farnesol, with the latter showing a 5.7-fold increase in catalytic activity as compared to wild-type CYP102A1. C(78–82, F87L) produces 10,11-epoxyfarnesol as the single primary metabolite. The results show that chimeragenesis involving only the second half of SRS-1 plus F87 is sufficient to change the substrate selectivity of CYP102A1 from fatty acids to farnesol and to produce a single primary product.





Similar content being viewed by others
Abbreviations
- CYP102A1:
-
Cytochrome P450 from Bacillus megaterium, also called P450-BM3
- CYP4C7:
-
A membrane-bound cytochrome P450 from the cockroach, Diploptera punctata
References
Miura Y, Fulco AJ (1974) Biochim Biophys Acta 249:1880–1888
Miura Y, Fulco AJ (1975) Biochim Biophys Acta 388:305–317
Narhi LO, Fulco AJ (1986) J Biol Chem 261:7160–7169
Fulco AJ (1991) Annu Rev Pharmacol Toxicol 31:177–203
Klein ML, Fulco AJ (1993) J Biol Chem 268:7553–7561
Klein ML, Fulco AJ (1994) Biochim Biophys Acta 1201:245–250
Yeom H, Sligar SG, Li H, Poulos TL, Fulco AJ (1995) Biochemistry 34:14733–14740
Murataliev MB, Feyereisen R, Walker FA (2004) Biochim Biophys Acta 1698:1–26
Capdevila JH, Wei S, Helvig C, Fulco JR, Belosludtsev Y, Truan G, Graham-Lorence SE, Peterson JA (1996) J Biol Chem 271:22663–22671
Munro AW, Leys DG, Mclean KJ, Marshall KR, Ost TW, Daff S, Miles CS, Chapman SK, Lysek DA, Moser CC, Page CC, Dutton PL (2002) Trends Biochem Sci 27:250–257
Gilardi G, Meharenna YT, Tsothou GE, Sadeghi SJ, Fairhead M, Giannini S (2002) Biosens Bioelectron 17:133–145
Fairhead M, Giannini S, Gillam EM, Gilardi G (2005) J Biol Inorg Chem 10:842–853
Dodhia VR, Fantuzzi A, Gilardi G (2006) J Biol Inorg Chem 11:903–916
Miles CS, Ost TW, Noble MA, Munro AW, Chapman SK (2000) Biochim Biophys Acta 1543:383–407
Li QS, Schwaneberg U, Fisher P, Schmitt J, Pleiss J, Lutz-Wahl S, Schmid RD (2001) Biochim Biophys Acta 1545:114–121
Li QS, Ogawa J, Schmid RD, Shimizu S (2001) Appl Environ Microbiol 67:5735–5739
Cowart LA, Falck JR, Capdevila JH (2001) Arch Biochem Biophys 387:117–124
Li H, Poulos TL (1995) Acta Crystalogr D51:21–32
Li H, Poulos TL (1997) Nat Struct Biol 4:140–146
Sevrioukova IF, Li H, Zhang H, Peterson JA, Poulos TL (1999) Proc Natl Acad Sci USA 99:1863–1868
Haines DC, Tomchick DR, Machius M, Peterson JA (2001) Biochemistry 40:13456–13465
Joyce MG, Girvan HM, Munro AW, Leys D (2004) J Biol Chem 279:23287–23293
Kuper J, Wong TS, Roccatano D, Wilmanns M, Schwaneberg U (2007) J Am Chem Soc 129:5786–5787
Modi S, Primrose WU, Boyle JMB, Gibson CF, Lian L-Y, Roberts GCK (1995) Biochemistry 34:8982–8988
Modi S, Sutcliffe MJ, Primrose WY, Lian L-Y, Roberts GCK (1996) Nat Struct Biol 3:414–417
Graham-Lorence S, Truan G, Peterson JA, Falck JR, Wei S, Helvig C, Capdevila JH (1997) J Biol Chem 272:1127–1135
Shao Z, Arnold FH (1996) Curr Opin Struct Biol 6:513–518
Oliver CF, Modi S, Sutcliffe MJ, Primrose WU, Lian LY, Roberts GC (1997) Biochemistry 36:1567–1572
Joo H, Lin Z, Arnold FH (1999) Nature 399:670–673
Noble MA, Miles CS, Chapman SK, Lysek DA, Mackay AC, Reid GA, Hanzlik RP, Munro AW (1999) Biochem J 339:371–379
Li QS, Schwaneberg U, Fisher P, Schmid RD (2000) Chem Eur J 6:1531–1536
Loughran PA, Roman LJ, Aitken AE, Miller RT, Masters BSS (2000) Biochemistry 39:15110–15120
Arnold FH, Wintrode PL, Miyazaki K, Gershenson A (2001) Trends Biochem Sci 26:100–106
Farinas ET, Schwaneberg U, Glieder A, Arnold FH (2001) Adv Synth Catal 343:601–606
Glieder A, Farinas ET, Arnold FH (2002) Nat Biotechnol 20:1135–1139
Salazar O, Cirino PC, Arnold FH (2003) Chembiochem 4:891–893
Kumar S, Chen CS, Waxman DJ, Halpert JR (2005) J Biol Chem 280:19569–19575
Meinhold P, Peters MW, Chen MM, Takahashi K, Arnold FH (2005) Chembiochem 6:1765–1768
Kumar S, Halpert JR (2005) Biochem Biophys Res Commun 338:456–464
Otey CR, Bandara G, Lalonde J, Takahashi K, Arnold FH (2006) Biotechnol Bioeng 93:494–499
Kumar S, Sun L, Liu H, Muralidhara BK, Halpert JR (2006) Protein Eng Des Sel 19:547–554
Kumar S, Liu H, Halpert JR (2006) Drug Metab Dispos 34:1958–1965
Sun L, Chen CS, Waxman DJ, Liu H, Halpert JR, Kumar S (2007) Arch Biochem Biophys 458:167–174
Zhao Y, Halpert JR (2007) Biochim Biophys Acta 1770:402–412
Peters MW, Meinhold P, Glieder A, Arnold FH (2003) J Am Chem Soc 125:13442–13450
Murataliev MB, Trinh LN, Moser LV, Bates RB, Feyereisen R, Walker FA (2004) Biochemistry 43:1771–1780
Sutherland TD, Unnithan GC, Andersen JF, Evans PH, Murataliev MB, Szabo LZ, Mash EA, Feyereisen R (1998) Proc Natl Acad Sci USA 95:12884–12889
Baker FC, Mauchamp B, Tsai LW, Schoolery DA (1983) J Lipid Res 24:1586–1594
Bede J, Goodman WG, Tobe S (1999) Pure Appl Chem 70:1–9
Yu SJ (2000) Zool Stud 39:243–249
Helvig C, Koener JF, Unnithan GC, Feyereisen R (2004) Proc Natl Acad Sci USA 101:4024–4029
Raner GM, Muir AQ, Lowry CW, Davis BA (2002) Biochem Biophys Res Commun 293:1–6
Barchuk AR, Maleszka R, Simoes LP (2004) Insect Mol Biol 13:459–467
Boddupalli SS, Pramanik BC, Slaughter CA, Estabrook RW, Peterson JA (1992) Arch Biochem Biophys 292:20–28
Pearson WR (1996) Meth Enzymol 266:227–258
Gotoh O (1992) J Biol Chem 267:83–90
Li H, Poulos TL (1999) Biochim Biophys Acta 1441:141–149
Murataliev MB, Feyereisen R (1996) Biochemistry 35:15029–15037
Morrison HG, Desrosiers RC (1993) Biotechniques 14:454–457
Bongers J, Walton CD, Richardson DE, Bell JU (1988) Anal Chem 60:2683–2686
Omura T, Sato R (1964) J Biol Chem 239:2379–2385
O’Keeffe DH, Ebel RE, Peterson JA (1978) Methods Enzymol 52:151–157
Nicolaides N, Soukup VG, Ruth EC (1983) Biomed Mass Spectrom 10:441–449
Guex N, Peitsch MC (1997) Electrophoresis 18:2714–2723
van Gunsteren WF et al (1996) Biomolecular simulation: the GROMOS96 manual and user guide. vdf Hochschulverlag AG an der ETH Zürich, Zürich/BIOMOS BV, Groningen
Luthy R, Bowie JU, David Eisenberg D (1992) Nature 356:83–85
Melo F, Feytmans E (1998) J Mol Biol 277:1141–1152
Davis CE, Bailey JL, Lockner JW, Coates RM (2003) J Org Chem 68:75–82
Hanzlik RP (1988) Org Synth Coll 6:560–565
Hanzlik RP (1977) Org Synth Coll 56:112–115
Acknowledgments
We thank Professor Armand Fulco of the University of California Los Angeles for the original E. coli strain pbsBM-3 with the plasmid carrying the CYP102A1 gene, Professor Paul Ortiz de Montellano for helpful comments, Dr. Markus Knipp for assistance in the modeling of the chimeras, Dr. Marat B. Murataliev (University of Alberta, Edmonton, AB, Canada) for technical advice on protein purification, fatty acid and farnesol metabolite identification, and Mary T. Flores for preparing the NMR figures. This research was supported by the Walker Research Gift Fund.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, CK.J., Shokhireva, T.K., Berry, R.E. et al. The effect of mutation of F87 on the properties of CYP102A1-CYP4C7 chimeras: altered regiospecificity and substrate selectivity. J Biol Inorg Chem 13, 813–824 (2008). https://doi.org/10.1007/s00775-008-0368-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-008-0368-5