Abstract
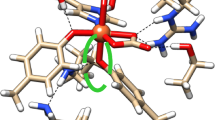
Density functional theory computations of heme with ionized propionic acid groups, axially coordinated with two imidazoles, were performed for different mutual orientations of the imidazole planes. Environmental influences from water or protein were considered with a continuum dielectric medium by solving the Poisson equation. In vacuum, optimized geometries yielded imidazole–heme conformations where the NH groups of imidazoles are oriented toward the heme propionic groups in agreement with data from crystal structures of heme proteins. Conformational free-energy dependencies of the mutual orientation of axially ligated imidazoles calculated in protein (ε=10) and water (ε=80) environments confirmed the vacuum results, albeit the energy difference between the preferred and the 180° opposite orientations of the imidazole ligand decreased from 3.84 kcal/mol in vacuum to 2.35 and 2.40 kcal/mol in protein and water, respectively. Two main factors determine the imidazole orientation: (1) the direct intramolecular electrostatic interactions of propionic groups with the polar NH groups of imidazole and (2) the electrostatic interaction of the total dipole moment of the imidazole–heme complex with the reaction field. In vacuum, only the first type of interaction is present, while in a dielectric medium the latter effect becomes competitive at high dielectric constant, resulting in a decrease of the orientational preference. Interestingly, the orientational preference of the imidazole axially ligated to heme becomes even more pronounced, if the negatively charged propionates are neutralized by counter charges that mimic salt bridges or protonation of the propionates.








Similar content being viewed by others
Abbreviations
- DFT:
-
Density functional theory
- ESP:
-
Electrostatic potential
- PDB:
-
Protein Data Bank
- RESP:
-
Restraint electrostatic potential
- vdW:
-
van der Waals
References
Bytheway I, Hall MB (1994) Chem Rev 94:639–658
Vangberg A, Ghosh A (1999) J Am Chem Soc 121:12154–12160
Ghosh A, Bocian DF (1996) J Phys Chem 100:6363–6367
Jewsbury P, Yamamoto S, Minato T, Saito M, Kitagawa T (1994) J Am Chem Soc 116:11586–11587
Harris D, Loew G, Waskell L (1998) J Am Chem Soc 120:4308–4318
Rovira C, Kunc K, Hutter J, Ballone P, Parinello M (1997) J Phys Chem 101:8914–8925
Rovira C, Parinello M (1999) Chem Eur J 5:250–263
Popovic DM, Zarić SD, Rabenstein B, Knapp E (2001) J Am Chem Soc 123:6040–6053
Smith DMA, Dupuis M, Vorpagel ER, Straatsma TP (2003) J Am Chem Soc 125:2711–2717
Iakovleva O, Reiner M, Rau H, Haehnel W, Parak F (2002) Phys Chem Chem Phys 4:655–660
Walker FA (1999) Coord Chem Rev 185–186:471–534
Safo MK, Walker FA, Raitsimring AM, Walters WP, Dolota DP, Debrunner PG, Scheidt WR (1994) J Am Chem Soc 116:7760–7770
Nakamura M, Ikeue T, Neya N, Fanasaki N, Nakamura N (1996) Inorg Chem 35:3731–3732
Nakamura M, Ikeue T, Ikezaki A, Ohgo Y, Fujii H (1999) Inorg Chem 38:3857–3863
Wiesemann F, Wonnemann R, Krebs B, Keutel H, Jager E (1994) Angew Chem Int Ed Engl 33:1363–1367
Wolowiec S, Latos-Gra6zynski L, Toronto D, Marchon JC (1998) Inorg Chem 37:724–732
Pilard MA, Guillemot M, Toupet L, Jordanov J, Simonneaux G (1997) Inorg Chem 36:6307–6314
Walker FA, Nasri H, Turowska-Tyrk I, Mohanrao K, Watson CT, Shokhirev NV, Debrunner PG, Scheidt WR (1996) J Am Chem Soc 118:12109–12118
Shokhirev NV, Walker FA (1995) J Phys Chem 99:17795–17804
Raitsimring AM, Walker FA (1998) J Am Chem Soc 120:991–1002
Shokhirev NV, Walker FA (1998) J Am Chem Soc 120:981–990
Nesset MJM, Shokhirev NV, Enemark PD, Jacobson SE, Walker FA (1996) Inorg Chem 35:5188–5200
Nasri H, Ellison MK, Chen S, Huynh BH, Scheidt WR (1997) J Am Chem Soc 119:6374–6383
Grodzicki M, Flint H, Winkler H, Walker FA, Trautwein A (1997) J Phys Chem 101:4202–4207
Walker FA, Huynh BH, Scheidt WR, Osvath S (1986) J Am Chem Soc 108:5288–5297
Menyhard DK, Keseru GM (1998) J Am Chem Soc 120:7991–7992
Zarić SD, Popović DM, Knapp EW (2001) Biochemistry 40:7914–7928
Munro OQ, Serth-Guzzo JA, Turkovska-Tyrk I, Mohanrao K, Shokhireva TKh, Walker FA, Debrunner PG, Scheidt W (1999) J Am Chem Soc 121:11144–11155
Safo MK, Nesset MJM, Walker FA, Debrunner PG, Scheidt WR (1997) J Am Chem Soc 119:9438–9448
Berman HM, Westbrok J, Feng Z, Gilliland G, Bhat TN, Weissig H, Shindyalov IN, Bourne PE (2000) The Protein Data Bank. Nucleic Acids Res 28:235–242
Simoes P, Matias PM, Morais J, Wilson K, Dauter Z, Carrondo MA (1998) Inorg Chim Acta 273:213–224
Slater JC (1974) Quantum theory of molecules and solids, vol 4. McGraw-Hill, New York
Becke AD (1988) Phys Rev A 38:3098–3100
Vosko SH, Wilk L, Nusair M (1980) Can J Phys 58:1200–1211
Lee C, Yang W, Paar RG (1988) Phys Rev B 46:785–789
Miehlich B, Savin A, Stoll H, Pruess H (1989) Chem Phys Lett 157:200–206
Zarić SD, Popović DM, Knapp EW (2000) Chem Eur J 6:3935–3942
Ghosh A, Gonzales E, Vangberg T (1999) J Phys Chem 103:1363–1367
Ziegler T (1995) Can J Chem 73:743–761
Strout DL, Zarić SD, Niu S, Hall MB (1996) J Am Chem Soc 118:6068–6069
Siegbahn PEM, Crabtree RH (1997) J Am Chem Soc 119:3103–3113
Niu S, Hall MB (1998) J Am Chem Soc 120:6169–6170
Zarić SD, Hall MB (1998) J Phys Chem 102:1963–1964
Zarić SD (1999) Chem Phys Lett 311:77–80
JAGUAR 4.2, Schrödinger, Inc, Portland, OR, 1991–2000
Tannor DJ, Marten B, Murphy R, Friesner RA, Sitkoff D, Nicholls A, Ringnalda M, Goddard III WA, Honig B (1994) J Am Chem Soc 116:11875–11882
Marten B, Kim K, Cortis C, Friesner RA, Murphy RB, Ringnalda MN, Sitkoff D, Honig B (1996) J Phys Chem 100:11775–11788
Hay PJ, Wadt WR (1985) J Chem Phys 82:299–310
Bayly C, Cieplak P, Cornell W, Kollman P (1993) J Phys Chem 97:10269–10280
Cornell W, Cieplak P, Bayly C, Kollman P (1993) J Am Chem Soc 115:9630–9631
Popović DM, Zarić SD, Rabenstein B, Knapp EW (2001) J Am Chem Soc 123:6040–6053
Schmidt am Busch M, Knapp EW (2004) Chem Phys Chem 5:1513–1522
Gilson M, Honig B (1986) Biopolymers 25:2097–2119
Rosen D (1963) Trans Faraday Soc 59:2178–2191
Takashima S, Schwan HP (1965) J Phys Chem 69:4176–4182
Schutz CN, Warshel A (2001) Proteins 44:400–417
Bashford D, Gerwert K (1992) J Mol Biol 224:473–486
Bashford D, (1997) An object-oriented programming suite for electrostatic effects in biological molecules. In: Yutaka I, Rodney RO, John VWR, Marydell T (eds) Scientific computing in object-oriented parallel environments, v. 1343 of Lecture notes in computer science, ISCOPE97. Springer Berlin Heidelberg, New York pp 233–240
Rocchia W, Alexov E, Honig B (2001) J Phys Chem 105:6507–6514
Rocchia W, Sridharan S, Nicholls A, Alexov E, Chiabrera A, Honig B (2002) J Comp Chem 23:128–137
ACD/ChemSketch, 1994–2001 Advanced Chemistry Development Inc
Voigt P, Knapp EW (2003) J Biol Chem 278:51993–52001
Momot KI, Walker FA (1997) J Phys Chem 101:2787–2795
Shokhirev NV, Shokhireva TKh, Polam JR, Watson CT, Raffii K, Simons U, Walker FA (1997) J Phys Chem 101:2778–2786
Nakamura M, Tajima K, Tada K, Ishizu K, Nakamura N (1994) Inorg Chim Acta 224:113–124
Polam JR, Shokhireva TKh, Raffii K, Simons U, Walker FA (1997) Inorg Chim Acta 263:109–117
Medacović V, Zarić SD (2003) Inorg Chim Acta 349:1–5
Humphrey W, Dalke A, Schulten K (1996) “VMD–visual molecular dynamics”. J Mol Graphics 14:33–38
Flükiger P, Lüthi HP, Portmann S, Weber J 2000–2001 MOLEKEL 4.3, Swiss Center for Scientific Computing, Manno Switzerland
Topol IA, Tawa GJ, Burt SK, Rashin AA (1997) J Phys Chem A 101:10075–10081
Rashin AA, Namboodiri K (1987) J Phys Chem 91:6003–6012
Miertuš S, Tomasi J (1982) Chem Phys 65:239–245
Chen JL, Noodleman L, Case DA, Bashford D (1994) J Phys Chem 98:11059–11068
Lovell T, Himo F, Han WG, Noodleman L (2003) Coor Chem Rev 238-239:211–232
Acknowledgements
We thank Donald Bashford and Martin Karplus for providing the programs MEAD and CHARMM, respectively. S.D.Z. is grateful for generous support by the Humboldt foundation. This work was supported by the Deutsche Forschungsgemeinschaft (SFB 498 Project A5; Forschergruppe 475; GRK 80/2; GRK 268; GRK 788/1).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Galstyan, A.S., Zarić, S.D. & Knapp, EW. Computational studies on imidazole heme conformations. J Biol Inorg Chem 10, 343–354 (2005). https://doi.org/10.1007/s00775-005-0642-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-005-0642-8