Abstract
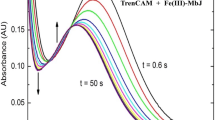
Aspergillus fumigatus is an opportunistic fungal pathogen that causes life-threatening infections in immunocompromised patients. Despite low levels of free iron, A. fumigatus grows in the presence of human serum in part because it produces high concentrations of siderophores. The most abundant siderophores produced by A. fumigatus are N′,N′′,N′′′-triacetylfusarinine C (TAF) and ferricrocin, both of which have thermodynamic iron binding constants that theoretically allow them to remove transferrin (Tf)-bound iron. Urea–polyacrylamide gel electrophoresis was used to measure the change in concentration of Tf species incubated with TAF or ferricrocin. The rate of removal of iron from diferric Tf by both siderophores was measured, as were the individual microscopic rates of iron removal from each Tf species (diferric Tf, N-terminal monoferric Tf and C-terminal monoferric Tf). TAF removed iron from all Tf species at a faster rate than ferricrocin. Both siderophores showed a preference for removing C-terminal iron, evidenced by the fact that k1C and k2C were much larger than k1N and k2N. Cooperativity in iron binding was observed with TAF, as the C-terminal iron was removed by TAF much faster from monoferric than from diferric Tf. With both siderophores, C-terminal monoferric Tf concentrations remained below measurable levels during incubations. This indicates that k2C and k1C are much larger than k1N. TAF and ferricrocin both removed Tf-bound iron with second-order rate constants that were comparable to those of the siderophores of several bacterial pathogens, indicating they may play a role in iron uptake in vivo and thereby contribute to the virulence of A. fumigatus.









Similar content being viewed by others
References
Howard DH (1999) Clin Microbiol Rev 12:394–404
Byers BR, Arceneaux JE (1998) Met Ions Biol Syst 35:37–66
Matzanke BF, Bill E, Trautwein AX, Winkelmann G (1987) J Bacteriol 169:5873–5876
Govindaraju K, Cowley EA, Eidelman DH, Lloyd DK (1998) J Chromatogr B Biomed Sci Appl 705:223–230
Baker EN, Baker HM, Kidd RD (2002) Biochem Cell Biol 80:27–34
Ratledge C, Dover LG (2000) Annu Rev Microbiol 54:881–941
Bullen JJ (1981) Rev Infect Dis 3:1127–1138
Torres AG, Redford P, Welch RA, Payne SM (2001) Infect Immun 69:6179–6185
Wertheimer AM, Verweij W, Chen Q, Crosa LM, Nagasawa M, Tolmasky ME, Actis LA, Crosa JH (1999) Infect Immun 67:6496–6509
Litwin CM, Rayback TW, Skinner J (1996) Infect Immun 64:2834–2838
Sokol PA, Darling P, Woods DE, Mahenthiralingam E, Kooi C (1999) Infect Immun 67:4443–4455
Meyer JM, Neely A, Stintzi A, Georges C, Holder IA (1996) Infect Immun 64:518–523
Latge JP (2001) Trends Microbiol 9:382–389
Hissen AH, Chow JM, Pinto LJ, Moore MM (2004) Infect Immun 72:1402–1408
Diekmann H, Krezdorn E (1975) Arch Microbiol 106:191–194
Nilius AM, Farmer SG (1990) J Med Vet Mycol 28:395–403
Moore RE, Emery T (1976) Biochemistry 15:2719–2723
Adjimani JP, Emery T (1987) J Bacteriol 169:3664–3668
Haselwandter K, Winkelmann G (2002) Biometals 15:73–77
Bartholdy BA, Berreck M, Haselwandter K (2001) Biometals 14:33–42
Hordt W, Romheld V, Winkelmann G (2000) Biometals 13:37–46
Ohra J, Morita K, Tsujino Y, Tazaki H, Fujimori T, Goering M, Evans S, Zorner P (1995) Biosci Biotechnol Biochem 59:113–114
Matzanke BF, Bill E, Trautwein AX, Winkelmann G (1988) Biol Met 1:18–25
Bentley MD, Anderegg RJ, Szaniszlo PJ, Davenport RF (1986) Biochemistry 25:1455–1457
Frederick CB, Szaniszlo PJ, Vickrey PE, Bentley MD, Shive W (1981) Biochemistry 20:2432–2436
Eisendle M, Oberegger H, Zadra I, Haas H (2003) Mol Microbiol 49:359–375
Konopka K, Bindereif A, Neilands JB (1982) Biochemistry 21:6503–6508
Harris WR, Carrano CJ, Cooper SR, Sofen SR, Avdeef AE, McArdle JV, Raymond KN (1979) J Am Chem Soc 101(20):6097–6104
Aisen P, Leibman A, Zweier J (1978) J Biol Chem 253:1930–1937
Harris WR (1983) Biochemistry 22:3920–3926
Pollack S, Vanderhoff G, Lasky F (1977) Biochim Biophys Acta 497:481–487
Wong GB, Kappel MJ, Raymond KN, Matzanke B, Winkelmann G (1983) J Am Chem Soc 105:810–815
Makey DG, Seal US (1976) Biochim Biophys Acta 453:250–256
Payne SM (1994) Meth Enzymol 235:329–344
Wiebe C, Winkelmann G (1975) J Bacteriol 123:837–842
Turcot I, Stintzi A, Xu J, Raymond KN (2000) J Biol Inorg Chem 5:634–641
Bali PK, Harris WR (1990) Arch Biochem Biophys 281:251–256
Baldwin DA, de Sousa DM (1981) Biochem Biophys Res Commun 99:1101–1107
Stintzi A, Raymond KN (2000) J Biol Inorg Chem 5:57–66
Wolz C, Hohloch K, Ocaktan A, Poole K, Evans RW, Rochel N, Albrecht-Gary AM, Abdallah MA, Doring G (1994) Infect Immun 62:4021–4027
Howard DH, Rafie R, Tiwari A, Faull KF (2000) Infect Immun 68:2338–2343
Ford S, Cooper RA, Evans RW, Hider RC, Williams PH (1988) Eur J Biochem 178:477–481
Leibman A, Aisen P (1979) Blood 53:1058–1065
Konopka K, Neilands JB (1984) Biochemistry 23:2122–2127
Warner PJ, Williams PH, Bindereif A, Neilands JB (1981) Infect Immun 33:540–545
Boelaert JR, de Locht M, Van Cutsem J, Kerrels V, Cantinieaux B, Verdonck A, Van Landuyt HW, Schneider YJ (1992) Clin Infect Dis 14:66–74
Schrettl M, Bignell E, Kragl C, Joechl C, Rogers T, Arst HN Jr, Haynes K, Haas H (2004) J Exp Med 200:1213–1219
Wendenbaum S, Demange P, Dell A, Meyer JM, Abdallah MA (1983) Tetrahedron Lett 24:4877–4880
Harris WR, Wang Z, Brook C, Yang B, Islam A (2003) Inorg Chem 42:5880–5889
Acknowledgements
The authors wish to thank Jurgen Hissen for assistance in deriving equations and for valuable discussions. Financial support from the Natural Sciences and Engineering Research Council of Canada is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hissen, A.H.T., Moore, M.M. Site-specific rate constants for iron acquisition from transferrin by the Aspergillus fumigatus siderophores N′,N′′,N′′′-triacetylfusarinine C and ferricrocin. J Biol Inorg Chem 10, 211–220 (2005). https://doi.org/10.1007/s00775-005-0630-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-005-0630-z