Abstract
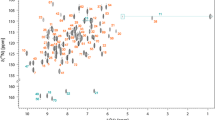
Desulfovibrio gigas ferredoxin II (DgFdII) is a small protein with a polypeptide chain composed of 58 amino acids, containing one Fe3S4 cluster per monomer. Upon studying the redox cycle of this protein, we detected a stable intermediate (FdIIint) with four 1H resonances at 24.1, 20.5, 20.8 and 13.7 ppm. The differences between FdIIox and FdIIint were attributed to conformational changes resulting from the breaking/formation of an internal disulfide bridge. The same 1H NMR methodology used to fully assign the three cysteinyl ligands of the [3Fe–4S] core in the oxidized state (DgFdIIox) was used here for the assignment of the same three ligands in the intermediate state (DgFdIIint). The spin-coupling model used for the oxidized form of DgFdII where magnetic exchange coupling constants of around 300 cm−1 and hyperfine coupling constants equal to 1 MHz for all the three iron centres were found, does not explain the isotropic shift temperature dependence for the three cysteinyl cluster ligands in DgFdIIint. This study, together with the spin delocalization mechanism proposed here for DgFdIIint, allows the detection of structural modifications at the [3Fe-4S] cluster in DgFdIIox and DgFdIIint.





Similar content being viewed by others
Abbreviations
- Dg :
-
Desulfovibrio gigas
- Fd:
-
Ferredoxin
- NOE:
-
Nuclear Overhauser effect
- NOESY:
-
Nuclear Overhauser enhancement spectroscopy
- SRB:
-
Sulfate-reducing bacteria
References
Holm RH (1992) Advances in inorganic chemistry, vol 38. Academic, San Diego
Johnson MK (1994) Encyclopedia of inorganic chemistry. In: King RB (ed) Wiley, Chichester, pp 1896–1915
Beinert H, Holm RH, Munck E (1997) Science 277:653–659
Beinert H (2000) J Biol Inorg Chem 5:2–15
Glaser T, Hedman B, Hodgson KO, Solomon EI (2000) Acc Chem Res 33:859–868
Noodleman L, Case DA (2000) Adv Inorg Chem 33:423–470
Calzolai L, Zhou Z-H, Adams MWW, LaMar GN (1996) J Am Chem Soc 118:2513–2514
Hunsicker-Wang LM, Heine A, Chen Y, Luna EP, Todaro T et al (2003) Biochemistry 42:7303–7317
Flint DH, Allen RM (1996) Chem Rev 96:2315–2334
Beinert H, Kennedy MC, Stout CD (1996) Chem Rev 96:2335–2373
Jarret JT (2003) Curr Opin Chem Biol 7:174–182
Cheek J, Broderick JB (2001) J Biol Inorg Chem 6:209–226
Dobbek H, Svetlitchnyi V, Gremer L, Huber R, Meyer O (2001) Science 293:1281–1285
Crane BR, Siegel LM, Getzoff ED (1995) Science 270:59–67
Doukov TI, Iverson TM, Seravalli J, Ragsdale SW, Drennan CL (2002) Science 298:567–572
Darnault C, Volbeda A, Kim EJ, Legrand P, Vernede X et al (2003) Nat Struct Biol 10:271–279
Svetlichnyi V, Dobbek H, Meyer-Klaucke W, Meins T, Thiele B et al (2004) Proc Natl Acad Sci USA 101:446–451
Nicolet Y, Cavazza C, Fontecilla-Camps JC (2002) J Inorg Biochem 91:1–8
Peters JW, Lanzilotta WN, Lemon BJ, Seefeldt LC (1998) Science 282:1853–1858
Thauer RK, Schönheit P (1982) Iron-sulfur proteins. In: Spiro TG (ed) Wiley-Interscience, New York, pp 329–341
Plank DW, Kennedy MC, Beinert H, Howard JB (1989) J Biol Chem 264:20385–20393
Golinelli MP, Chatelet C, Duin EC, Johnson MK, Meyer J (1998) Biochemistry 37:10429–10437
Mulholland SE, Gibney BR, Rabanal F, Dutton PL (1998) J Am Chem Soc 120:10296–10302
Cunningham RP, Asahara H, Bank JF, Scholes CP, Salerno JC et al (1989) Biochemistry 28:4450–4455
Kuo CF, McRee DE, Fisher CL, O’Handley SF, Cunningham RP, Tainer JA (1992) Science 258:434–440
Porello SL, Cannon MJ, David SS (1998) Biochemistry 37:6465–6475
Guan Y, Manuel RC, Arvai AS, Parikh SS, Mol CD et al (1998) Nat Struct Biol 5:1058–1064
Kiley PJ, Beinert H (2003) Curr Opin Microbiol 6:181–185
Demple B, Ding H, Jorgensen M (2002) Methods Enzymol 348:355–364
Alen C, Sonenshein AL (1999) Proc Natl Acad Sci USA 96:10412–10417
Tang Y, Guest JR (1999) Microbiology 145:3069–3079
Smith JL, Zaluzec EJ, Wery JP, Niu L, Switzer RL et al (1994) Science 264:1427–1433
Wu CK, Dailey HA, Rose JP, Burden A, Sellers VM, Wang BC (2001) Nat Struct Biol 8:156–160
Sellers VM, Johnson MK, Dailey HA (1996) Biochemistry 35:2699–2704
Dai S, Schwendtmayer C, Schurmann P, Ramaswamy S, Eklund H (2000) Science 287:655–658
Duin EC, Madadi-Kahkesh S, Hedderich R, Clay MD, Johnson MK (2002) FEBS Lett 512:263–268
Walters EM, Johnson MK (2004) Photosynth Res 79:249–264
Ugulave NB, Gibney BR, Jarret JT (2001) Biochemistry 40:8343–8351
Berkovitch F, Nicolet Y, Wan JT, Jarret JT, Drennan CL (2004) Science 303:76–79
Jameson GN, Cosper MM, Hernandez HL, Johnson MK, Huynh BH (2004) Biochemistry 43:2022–2031
Moura JJG, Macedo A, Palma PN (1994) Ferredoxins. Methods in enzymology. In: Peck HD Jr, LeGall J (eds) Inorganic microbial sulphur metabolism, vol 243. Academic, New York, chap 12
Bruschi M, Hatchikian C, LeGall J, Moura JJG, Xavier AV (1976) Biochem Biophys Acta 449:275–284
Bruschi M (1979) Biochim Biophys Res Commun 91:623
Kissinger CR, Sieker LC, Adman ET, Jensen JH (1991) J Mol Biol 219:693–715
Goodfellow BJ, Macedo AL, Rodrigues P, Wray V, Moura I, Moura JJG (1999) J Biol Inorg Chem 4:421–430
Macedo AL, Moura I, Surerus KK, Papaefthymiou V, Liu M, LeGall J, Münck E, Moura JJG (1994) J Biol Chem 269:8052–8058
Moura JJG, Moura I, Kent TA, Lipscomb JD, Huynh BH, LeGall J, Xavier AV, Münck E (1982) J Biol Chem 257:6259–6267
Moura I, Moura JJG, Münck E, Papaefthymiou V, LeGall J (1986) J Am Chem Soc 108:349–351
Butt JN, Sucheta A, Breton J, Thomson AJ, Hatchikian EC (1991) J Am Chem Soc 113:8948–8950
Conover RC, Park J-B, Adams MWW, Johnson MK (1990) J Am Chem Soc 112:4562–4564
Surerus KK, Münck E, Moura I, Moura JJG, LeGall J (1987) J Am Chem Soc 109:3805–3807
Moreno C, Macedo AL, Moura I, LeGall J, Moura JJG (1994) J Inorg Biochem 53:219–234
Hsieh YC, Liu MY, LeGall J, Chen CJ (2005) Acta Crystallogr D 61:780–783
Kent TA, Huynh BH, Münck E (1980) Proc Natl Acad Sci USA 77:6574
Emptage MH, Kent TA, Huynh BH, Rawlings J, Orme-Johnson WH, Münk E (1980) J Biol Chem 255:1793–1796
Xavier AV, Moura JJG, Moura I (1981) Struct Bonding 43:187–213
Armstrong FA, Butt JN, George SJ, Hatchikian EC, Thomson AJ (1989) FEBS Lett 259:15
Shen B, Martin LL, Butt JN, Armstrong FA, Stout CD, Jensen GM, Stephens PJ, LaMar GN, Gorst CM, Burgess BK (1993) J Biol Chem 268:25929–25939
Breton JL, Duff JL, Butt JN, Armstrong FA, Petillot Y, Forest E, Schafer G, Thomson AJ (1995) Eur J Biochem 233:937–946
Smith ET, Blamey JM, Zhou ZH, Adams MWW (1995) Biochemistry 53:219–234
Macedo AL, Palma N, Moura I, LeGall J, Wray V, Moura JJG (1993) Magn Res Chem 31:S59–S67
Macedo AL, Moura I, LeGall J, Huynh B, Moura JJG (1993) Inorg Chem 32:1101–1105
Bertini I, Ciurli S, Luchinat C (1995) Struct Bonding 83:1–53
Noodleman L (1988) Inorg Chem 27:3677–3679
Aono S, Bertini I, Cowan JA, Luchinat C, Rosato A, Viezolli MS (1996) J Biol Inorg Chem 1:523–528
Bertini I, Dikiy A, Luchinat C, Macinai R, Viezolli MS, Vincenzini M (1997) Biochemistry 36:3570–3579
Gorst CM, Zhou ZH, Ma K, Teng Q, Howard JB, Adams MWW, LaMar GN (1995) Biochemistry 34:8788–8795
Vold RL, Waugh JS, Klein MP, Phelps DE (1968) J Chem Phys 48:3831
Inubushi T, Becker EDJ (1983) J Magn Res 51:128–133
Bertini I, Briganti F, Luchinat C, Scozzafava A, Sola M (1991) J Am Chem Soc 113:1237–1245
Macura SR, Ernst RR (1980) Mol Phys 40:95–117
Kumar A, Ernst RR, Wüthrich K (1980) Biochem Biophys Res Commun 95:1
Busse SC, LaMar GN, Yu LP, Howard JB, Smith ET, Zhou ZH, Adams MWW (1992) Biochemistry 31: 11952–11962
Donaire A, Gorst CM, Zhou ZH, Adams MWW, LaMar GN (1994) J Am Chem Soc 116:6841–6849
Cheng H, Grohmann K, Sweeney W (1992) J Biol Chem 267:8073–8080
Davy SL, Osborne MJ, Breton J, Moore GR, Thomson AJ, Bertini I, Luchinat C (1995) FEBS Lett 363:199–204
Imai T, Matsumoto T, Ohta S, Ohmori D, Suzuki K, Tanaka J, Tsukioka M, Tobari J (1983) Biochim Biophys Acta 743:91–97
Trower MK, Marshall JE, Doleman MS, Emptage MH, Sariaslani FS (1990) Biochim Biophys Acta 1037:290–296
Bertini I, Capozzi F, Luchinat C, Piccioli M, Vila AJ (1994) J Am Chem Soc 116:651–660
Werth MT, Kurtz Jr DM, Moura I, LeGall J (1987) J Am Chem Soc 109:273–275
Rodrigues P, Graça F, Macedo AL, Moura I, Moura JJG (2001) Biochem Biophys Res Commun 289:630–633
Acknowledgements
We would like to thank Ludwig Krippahl for his help with the computer software provided by him and used in this work. This work was supported by Fundação para a Ciência e Tecnologia and COST D2 1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rodrigues, P.M., Macedo, A.L., Goodfellow, B.J. et al. Desulfovibrio gigas ferredoxin II: redox structural modulation of the [3Fe–4S] cluster. J Biol Inorg Chem 11, 307–315 (2006). https://doi.org/10.1007/s00775-005-0077-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-005-0077-2