Abstract
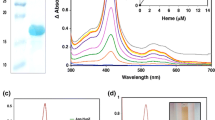
Thermotoga maritima grows optimally at 80 °C by fermenting carbohydrates to organic acids, CO2, and H2. The production of H2 is catalyzed by a cytoplasmic, heterotrimeric (αβγ) Fe-hydrogenase. This is encoded by three genes, hydC (γ), hydB (β) and hydA (α), organized within a single operon that contains five additional open reading frames (ORFs). The recombinant form of the first ORF of the operon, TM1420, was produced in Escherichia coli. It has a molecular mass of 8537±3 Da as determined by mass spectrometry, in agreement with the predicted amino acid sequence. Purified TM1420 is red in color, has a basic pI (8.8), and contains 1.9 Fe atoms/mol that are present as a single [2Fe-2S] cluster, as determined by UV-visible absorption and EPR spectroscopy. The protein contains five cysteine residues, but their arrangement is characteristic of a subunit or domain rather than of a ferredoxin-type protein. The reduction potential of the [2Fe-2S] cluster (−233 mV at pH 6.5 and 25 °C) is pH independent but decreases linearly with temperature to −296 mV (−1.15 mV/°C) at 80 °C. TM1420 is not reduced, in vitro, by the Fe-hydrogenase nor by a pyruvate ferredoxin oxidoreductase. The protein was unstable at 70 °C under anaerobic conditions with a half-life of ~30 min. The basic nature of TM1420, its instability at the growth temperature of T. maritima, and the unusual spacing of its cysteine residues suggest that this protein does not function as a ferredoxin-type electron carrier for the Fe-hydrogenase. Instead, TM1420 is more likely part of a thermostable multi-protein complex that is involved in metal cluster assembly of the hydrogenase holoenzyme.







Similar content being viewed by others
Abbreviations
- CHES:
-
2-(N-cyclohexylamino)-1-propanesulfonic acid
- DTT:
-
dithiothreitol
- EPPS:
-
N-(2-hydroxyethyl)piperazine-N'-(3-propanesulfonic acid)
- EPR:
-
electron paramagnetic resonance
- Fd:
-
ferredoxin
- HEPES:
-
N-(2-hydroxyethyl)piperazine-N'-(2-ethanesulfonic acid)
- ICP:
-
inductively coupled plasma emission spectroscopy
- MES:
-
2-(N-morpholino)ethanesulfonic acid
- ORF:
-
open reading frame
- PCR:
-
polymerase chain reaction
- SDS-PAGE:
-
sodium dodecyl sulfate-polyacrylamide gel electrophoresis
- SHE:
-
standard hydrogen electrode
References
Adams MWW (1990) Biochim Biophys Acta 1020:115–145
Albracht SPJ (1994) Biochim Biophys Acta 1188:167–204
Vignais PM, Billoud B, Meyer J (2001) FEMS Microbiol Rev 25:455–501
Horner DS, Heil B, Happe T, Embley TM (2002) Trends Biochem Sci 27:148–153
Peters JW, Lanzilotta WN, Lemon BJ, Seefeldt LC (1998) Science 282:1853–1858
Nicolet Y, Piras C, Legrand P, Hatchikian CE, Fontecilla-Camps JC (1999) Structure 7:13–23
Schönheit P, Schäfer T (1995) World J Microbiol Biotech 11:26–57
Verhagen MFJM, O'Rourke T, Adams MWW (1999) Biochim Biophys Acta 1412:212–229
Verhagen MFJM, O'Rourke T, Menon AL, Adams MWW (2001) Biochim Biophys Acta 1505:209–219
Nelson KE, Clayton RA, Gill SR, Gwinn ML, Dodson RJ, Haft DH, Hickey EK, Peterson JD, Nelson WC, Ketchum KA, McDonald L, Utterback TR, Malek JA, Linher KD, Garrett MM, Stewart AM, Cotton MD, Pratt MS, Phillips CA, Richardson D, Heidelberg J, Sutton GG, Fleischmann RD, Eisen JA, White O, Salzberg SL, Smith HO, Venter JC, Fraser CM (1999) Nature 399:323–329
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Laemmli UK (1970) Nature 227:680–685
Goa J (1953) Scan J Clin Lab Invest 5:218–222
Bensadoun A, Weinstein D (1976) Anal Biochem 70:241–250
Lovenberg W, Buchanan BB, Rabinowitz JC (1963) J Biol Chem 238:3899–3913
Brereton, PS, Verhagen MFJM, Zhou ZH, Adams MWW (1998) Biochemistry 37:7351–7362
Menon AL, Hendrix H, Hutchins A, Verhagen MFJM, Adams, MWW (1998) Biochemistry 37:12838–12846
Matsubara H, Saeki K (1992) Adv Inorg Chem 38:223–280
Meyer J, Fujinaga J, Gaillard J, Lutz M (1994) Biochemistry 33:13642–13650
Johnson MK (1994) In: King RB (ed) Encyclopedia of inorganic chemistry. Wiley, New York, pp 1896–1915
Bertini I, Luchinat C, Provenzani A, Rosato A, Vasos PR (2002) Proteins Struct Funct Genet 46:110–127
Xu XM, Matsuno-Yagi A, Yagi T (1991) Biochemistry 30:8678–8684
Weidner U, Geier S, Ptock A, Freidrich T, Leif H, Weiss H (1993) J Mol Biol 233:109–122
Yano T, Sled VD, Ohnishi T, Yagi T (1994) Biochemistry 33:494–499
De Luca G, Asso M, Bélaïch J-P, Dermoun Z (1998) Biochemistry 37:2660–2665
Chatelet C, Meyer J (1999) J Biol Inorg Chem 4:311–317
Meyer J (2001) FEBS Lett 509:1–5
Chatelet C, Gaillard J, Pétillot Y, Louwagie M, Meyer J (1999) Biochem Biophys Res Commun 261:885–889
Yeh AP, Chatelet C, Soltis SM, Kuhn P, Meyer J, Rees DC (2000) J Mol Biol 300:587–595
Gollinelli M-P, Chatelet C, Duin EC, Johnson MK, Meyer J (1998) Biochemistry 37:10429–10437
Heering HA, Bulsink YBM, Hagen WR, Meyer TE (1995) Biochemistry 34:14675–14686
Voordouw G, Hagen WR, Krüse-Wolters KM, van Berkel-Arts A, Veeger C (1987) Eur J Biochem 162:31-36
Atta M, Meyer J (2000) Biochim Biophys Acta 1476:368–371
Dean DR, Bolin JT, Zheng L (1993) J Bacteriol 175:6737–6744
Acknowledgements
We wish to thank Ish Dhawan for assistance with the EPR and Marc Verhagen for many helpful discussions. This research was supported by a grant from the National Science Foundation (MCB 9904624).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pan, G., Menon, A.L. & Adams, M.W.W. Characterization of a [2Fe-2S] protein encoded in the iron-hydrogenase operon of Thermotoga maritima . J Biol Inorg Chem 8, 469–474 (2003). https://doi.org/10.1007/s00775-002-0439-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-002-0439-y