Abstract
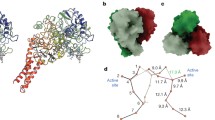
The facultative sulfate/nitrate-reducing bacterium Desulfovibrio desulfuricans ATCC 27774 harbours a split-Soret cytochrome c. This cytochrome is a homodimeric protein, having two bis-histidinyl c -type haems per monomer. It has an unique architecture at the haem domain: each haem has one of the coordinating histidines provided by the other monomer, and in each monomer the haems are parallel to each other, almost in van der Waals contact. This work reports the cloning and sequencing of the gene encoding for this cytochrome and shows, by transcriptional analysis, that it is more expressed in nitrate-grown cells than in sulfate-grown ones. In addition, the gene-deduced amino acid sequence revealed two new cysteine residues that could be involved in the binding of a non-haem iron centre. Indeed, the presence of a novel type of an iron-sulfur centre (possibly of the [2Fe-2S] type) was demonstrated by EPR spectroscopy, and putative models for its localization and structure in the cytochrome molecule are proposed on the basis of the so-far-known 3D crystallographic structure of the aerobically purified split-Soret cytochrome, which lacks this centre.








Similar content being viewed by others
References
Liu MC, Costa C, Coutinho IB, Moura JJ, Moura I, Xavier AV, LeGall J (1988) J Bacteriol 170:5545–5551
Pereira IAC, Teixeira M, Xavier AV (1998) Struct Bonding 91:65–89
Coelho AV, Matias P, Sieker LC, Morais J, Carrondo MA, Lampreia J, Costa C, Moura JJ, Moura I, LeGall J (1996) Acta Crystallogr Sect D 52:1202–1208
Coutinho IB, Xavier AV (1994) Methods Enzymol 243:119–140
Arciero DM, Collins MJ, Haladjian J, Bianco P, Hooper AB (1991) Biochemistry 30:11459–11465
Pereira MM, Carita JN, Teixeira M (1999) Biochemistry 38:1268–1275
Matias PM, Morais J, Coelho AV, Meijers R, Gonzalez A, Thompson AW, Sieker L, LeGall J, Carrondo MA (1997) J Biol Inorg Chem 2:507–514
Devreese B, Costa C, Demol H, Papaefthymiou V, Moura I, Moura JJ, Van Beeumen J (1997) Eur J Biochem 248:445–451
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (1995) Current protocols in molecular biology. Greene/Wiley, New York
Stokkermans JP, Pierik AJ, Wolbert RB, Hagen WR, Van Dongen WM, Veeger C (1992) Eur J Biochem 208:435–442
Liu MC, Peck HD Jr (1981) J Biol Chem 256:13159–13164
Corpet F (1988) Nucleic Acids Res 16:10881–10890
Reese MG, Harris NL, Eeckman FH (1996) In: Hunter L, Klein TE (eds) Biocomputing: proceedings of the 1996 Pacific symposium. World Scientific, Singapore, pp 737–738
Thompson JD, Higgins DG, Gibson TJ (1994) Nucleic Acids Res 22:4673–4680
Klenk HP, Clayton RA, Tomb J-F, White O, Nelson KE, Ketchum KA, Dodson RJ, Gwinn M, Hickey EK, Peterson JD, Richardson DL, Kerlavage AR, Graham DE, Kyrpides NC, Fleischmann RD, Quackenbush J, Lee NH, Sutton GG, Gill S, Kirkness EF, Dougherty BA, McKenney K, Adams MD, Loftus B, Peterson S, Reich CI, McNeil LK, Badger JH, Glodek A, Zhou L, Overbeek R, Gocayne JD, Weidman JF, McDonald L, Utterback T, Cotton MD, Spriggs T, Artiach P, Kaine BP, Sykes SM, Sadow PW, D'Andrea KP, Bowman C, Fujii C, Garland SA, Mason TM, Olsen GJ, Fraser CM, Smith HO, Woese CR, Venter JC (1997) Nature 390: 364–370
Peitsch MC (1995) Bio/Technology 13:658–660
Peitsch MC (1996) Biochem Soc Trans 24:274–279
Guex N, Peitsch MC (1997) Electrophoresis 18:2714–2723
Chothia C, Lesk AM (1986) EMBO J 5:823–826
Harrison RW, Chatterjee D, Weber IT (1995) Proteins Struct Funct Genet 2:463–471
Bajorath J, Stenkamp R, Aruffo A (1993) Protein Sci 2:1798–1810
LeGall J, Liu MY, Gomes CM, Braga V, Pacheco I, Regalla M, Xavier AV, Teixeira M (1998) FEBS Lett 429:295–298
Berry EA, Trumpower BL (1987) Anal Biochem 161:1–15
Fischer DS, Price DC (1964) Clin Chem 10:21–25
Aasa R, Vanngard VT (1975) J Magn Reson 19:308–315
Brünger AT (1992) X-PLOR, version 3.1: a system for crystallography and NMR. Yale University Press, New Haven
Brünger AT (1992) Nature 355:472–474
Roussel A, Fontecilla-Camps JC, Cambillau C (1990) In: XV IUCR congress abstracts, Bordeaux, France, pp 66–67
Read RJ (1986) Acta Crystallogr Sect A 42:140–149
Berman HM, Westbrook J, Feng Z, Gilliland G, Bhat TN, Weissig H, Shindyalov IN, Bourne PE (2000) Nucleic Acids Res 28:235–242
Saraiva LM, da Costa PN, Conte C, Xavier AV, LeGall J (2001) Biochim Biophys Acta 1520:63–70
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) J Appl Crystallogr 26:283–291
Ramachandran GN, Sasisekharan V (1968) Adv Protein Chem 23: 283–437
Kraulis PJ (1991) J Appl Crystallogr 24:946–950
Merritt EA, Murphy MEP (1994) Acta Crystallogr Sect D 50:869–873
Iverson TM, Arciero DM, Hooper AB, Rees DC (2001) J Biol Inorg Chem 6:390–397
Collaborative Computational Project Number 4 (1994) Acta Crystallogr Sect D 50:760–763
Esnouf RM (1999) Acta Crystallogr Sect D 55:938–940
Sanner MF, Spehner J-C, Olson AJ (1996) Biopolymers 38:305–320
Liang J, Edelsbrunner H, Woodward C (1998) Protein Sci 7:1884–1897
Eisenhaber F, Lijnzaad P, Argos P, Sander C, Scharf M (1995) J Comput Chem 16:273–284
Yuvaniyama P, Agar JN, Cash VL, Johnson MK, Dean DR (2000) Proc Natl Acad Sci USA 97:599–604
Fujita T (1966) J Biochem (Tokyo) 60:204–215
Kajie S-I, Anraku Y (1986) Eur J Biochem 154:457–463
Pereira IAC, Abreu IA, Xavier AV, LeGall J, Teixeira M (1996) Biochem Biophys Res Commun 224:611–618
Acknowledgements
We would like to thank Isabel Pacheco and Célia V. Romão for their help in the purification of the Dd27k split-Soret cytochrome c. This work was supported by Fundação da Ciência e Tecnologia projects POCTI/1999/BME/36558 (to M.T.) and POCTI/1999/BME/35021 (to A.V.X.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abreu, I.A., Lourenço, A.I., Xavier, A.V. et al. A novel iron centre in the split-Soret cytochrome c from Desulfovibrio desulfuricans ATCC 27774. J Biol Inorg Chem 8, 360–370 (2003). https://doi.org/10.1007/s00775-002-0426-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-002-0426-3