Abstract.
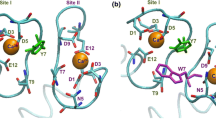
Peptides (33–34 amino acids long) corresponding to the helix-turn-helix (EF-hand) motif of the calcium binding site I of Paramecium tetraurelia calmodulin have been synthesized. The linear sequence was unable to acquire a native-like conformation and calcium binding. However, incorporation of a well-positioned disulfide bond bridging the two putative helical regions greatly improved the ordered structure and binding properties. Analyzed by electrospray mass spectrometry, circular dichroism and time-resolved laser-induced fluorescence, such a disulfide-stabilized peptide is shown to acquire a calcium-dependent helical conformation and exhibits native-like affinity for calcium, terbium and europium ions with 30±1, 3.5±0.6 and 0.6±0.1 µM dissociation constants, respectively. Comparable affinities were calculated within the biological construct comprising the entire domain I of Arabidopsis taliana calmodulin. Single sequence mutation (Glu25Asp) in the binding loop of the peptide abolishes calcium affinity, but preserves lanthanide affinity, showing that metal selectivity can be modulated by specific mutations. Such disulfide-stabilized peptides represent useful models to engineer metal specificity in new calmodulin proteins, facilitating the development of new systems to monitor metal pollution in biosensors and to increase metal binding capability of bacterial and plant cells in bioremediation techniques. Electronic supplementary material is available if you access this article at http://dx.doi.org/10.1007/s00775-002-0419-2. On that page (frame on the left side), a link takes you directly to the supplementary material.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Le Clainche, L., Plancque, G., Amekraz, B. et al. Engineering new metal specificity in EF-hand peptides. J Biol Inorg Chem 8, 334–340 (2003). https://doi.org/10.1007/s00775-002-0419-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00775-002-0419-2