Abstract
Introduction
This study aimed to assess the effectiveness of calcimimetics in reducing the risk of fractures in dialysis patients with secondary hyperparathyroidism (SHPT).
Material and methods
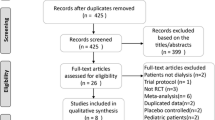
A comprehensive literature search was conducted using PubMed, Embase, and Cochrane Library for articles published through December 9, 2023. The quality of each trial was evaluated using the Cochrane Collaboration tool. Meta-analysis was performed using a random-effects model, and effect measures across studies were synthesized. The risk ratio (RR) and 95% confidence interval (CI) were used to quantify the risk of fracture.
Results
We identified seven studies involving 6481 dialysis patients with SHPT. The administration of calcimimetics reduced fracture incidence compared to placebo or conventional treatment (RR: 0.50, 95% CI 0.29–0.88, p = 0.02). Calcimimetics demonstrated a low number needed to treat (NNT) to prevent an incident fracture (NNT: 47).
Conclusion
The use of calcimimetics offers a significant benefit in reducing the risk of fractures in patients undergoing dialysis with SHPT.




Similar content being viewed by others
References
Wakasugi M, Kazama JJ, Taniguchi M, Wada A, Iseki K, Tsubakihara Y, Narita I (2013) Increased risk of hip fracture among Japanese hemodialysis patients. J Bone Miner Metab 31:315–321. https://doi.org/10.1007/s00774-012-0411-z
Alem AM, Sherrard DJ, Gillen DL, Weiss NS, Beresford SA, Heckbert SR, Wong C, Stehman-Breen C (2000) Increased risk of hip fracture among patients with end-stage renal disease. Kidney Int 58:396–399. https://doi.org/10.1046/j.1523-1755.2000.00178.x
Coco M, Rush H (2000) Increased incidence of hip fractures in dialysis patients with low serum parathyroid hormone. Am J Kidney Dis 36:1115–1121. https://doi.org/10.1053/ajkd.2000.19812
Tentori F, McCullough K, Kilpatrick RD, Bradbury BD, Robinson BM, Kerr PG, Pisoni RL (2014) High rates of death and hospitalization follow bone fracture among hemodialysis patients. Kidney Int 85:166–173. https://doi.org/10.1038/ki.2013.279
Tentori F, Wang M, Bieber BA, Karaboyas A, Li Y, Jacobson SH, Andreucci VE, Fukagawa M, Frimat L, Mendelssohn DC, Port FK, Pisoni RL, Robinson BM (2015) Recent changes in therapeutic approaches and association with outcomes among patients with secondary hyperparathyroidism on chronic hemodialysis: the DOPPS study. Clin J Am Soc Nephrol: CJASN 10:98–109. https://doi.org/10.2215/CJN.12941213
Taniguchi M, Fukagawa M, Fujii N, Hamano T, Shoji T, Yokoyama K, Nakai S, Shigematsu T, Iseki K, Tsubakihara Y, Committee of Renal Data Registry of the Japanese Society for Dialysis T (2013) Recent changes in therapeutic approaches and association with outcomes among patients with secondary hyperparathyroidism on chronic hemodialysis: the DOPPS study. Ther Apher Dial: Off Peer-Rev J Int Soc Apher Jpn Soc Apher Jan Soc Dial Ther 17:228. https://doi.org/10.1111/1744-9987.12030
Block GA, Kilpatrick RD, Lowe KA, Wang W, Danese MD (2013) CKD-mineral and bone disorder and risk of death and cardiovascular hospitalization in patients on hemodialysis. Clin J Am Soc Nephrol: CJASN 8:2132–2140. https://doi.org/10.2215/CJN.04260413
Fernandez-Martin JL, Martinez-Camblor P, Dionisi MP, Floege J, Ketteler M, London G, Locatelli F, Gorriz JL, Rutkowski B, Ferreira A, Bos WJ, Covic A, Rodriguez-Garcia M, Sanchez JE, Rodriguez-Puyol D, Cannata-Andia JB, group C, (2015) Improvement of mineral and bone metabolism markers is associated with better survival in haemodialysis patients: the COSMOS study. Nephrol, Dial, Transplant : Off Publ Eur Dial Transplant Assoc - Eur Ren Assoc 30:1542–1551. https://doi.org/10.1093/ndt/gfv099
Wakasugi M, Kazama JJ, Kikuchi K, Yasuda K, Wada A, Hamano T, Masakane I, Narita I (2019) Hemodialysis product and hip fracture in hemodialysis patients: a nationwide cohort study in Japan. Ther Apher Dial: Off Peer-Rev J Int Soc Apher, Jpn Soc Apher, Jpn Society Dial Ther 23:507–517. https://doi.org/10.1111/1744-9987.12807
Jadoul M, Albert JM, Akiba T, Akizawa T, Arab L, Bragg-Gresham JL, Mason N, Prutz KG, Young EW, Pisoni RL (2006) Incidence and risk factors for hip or other bone fractures among hemodialysis patients in the Dialysis Outcomes and Practice Patterns Study. Kidney Int 70:1358–1366. https://doi.org/10.1038/sj.ki.5001754
Custodio MR, Koike MK, Neves KR, dos Reis LM, Graciolli FG, Neves CL, Batista DG, Magalhaes AO, Hawlitschek P, Oliveira IB, Dominguez WV, Moyses RM, Jorgetti V (2012) Parathyroid hormone and phosphorus overload in uremia: impact on cardiovascular system. Nephrol, Dial, Transplant: Off Publ Eur Dial Transplant Assoc – Eur Ren Assoc 27:1437–1445. https://doi.org/10.1093/ndt/gfr447
Block GA, Martin KJ, de Francisco AL, Turner SA, Avram MM et al (2004) Cinacalcet for secondary hyperparathyroidism in patients receiving hemodialysis. N Engl J Med 350:1516–1525. https://doi.org/10.1056/NEJMoa031633
Investigators ET, Chertow GM, Block GA, Correa-Rotter R, Drueke TB, Floege J, Goodman WG, Herzog CA, Kubo Y, London GM, Mahaffey KW, Mix TC, Moe SM, Trotman ML, Wheeler DC, Parfrey PS (2012) Effect of cinacalcet on cardiovascular disease in patients undergoing dialysis. N Engl J Med 367:2482–2494. https://doi.org/10.1056/NEJMoa1205624
Diaz-Tocados JM, Rodriguez-Ortiz ME, Almaden Y, Pineda C, Martinez-Moreno JM et al (2019) Calcimimetics maintain bone turnover in uremic rats despite the concomitant decrease in parathyroid hormone concentration. Kidney Int 95:1064–1078. https://doi.org/10.1016/j.kint.2018.12.015
Koiwa F, Tokunaga S, Asada S, Endo Y, Fukagawa M, Akizawa T (2021) Efficacy of evocalcet in previously cinacalcet-treated secondary hyperparathyroidism patients. Kidney Int Rep 6:2830–2839. https://doi.org/10.1016/j.ekir.2021.08.020
Block GA, Bushinsky DA, Cheng S, Cunningham J, Dehmel B, Drueke TB, Ketteler M, Kewalramani R, Martin KJ, Moe SM, Patel UD, Silver J, Sun Y, Wang H, Chertow GM (2017) Effect of etelcalcetide vs cinacalcet on serum parathyroid hormone in patients receiving hemodialysis with secondary hyperparathyroidism: a randomized clinical trial. JAMA 317:156–164. https://doi.org/10.1001/jama.2016.19468
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Moe SM, Abdalla S, Chertow GM, Parfrey PS, Block GA, Correa-Rotter R, Floege J, Herzog CA, London GM, Mahaffey KW, Wheeler DC, Dehmel B, Goodman WG, Drueke TB, Evaluation of Cinacalcet HTtLCETI (2015) Effects of cinacalcet on fracture events in patients receiving hemodialysis: the evolve trial. J Am Soc Nephrol 26:1466–1475. https://doi.org/10.1681/ASN.2014040414
Kazama JJ, Koiwa F, Yokoyama K, Fukagawa M, Asano K, Honda D, Akizawa T (2022) First-in-patient phase i/ii study of upacicalcet in Japanese patients with secondary hyperparathyroidism undergoing hemodialysis: pharmacokinetic and pharmacodynamic properties. Clin Pharmacokinet 61:1271–1284. https://doi.org/10.1007/s40262-022-01139-w
Cunningham J, Danese M, Olson K, Klassen P, Chertow GM (2005) Effects of the calcimimetic cinacalcet HCl on cardiovascular disease, fracture, and health-related quality of life in secondary hyperparathyroidism. Kidney Int 68:1793–1800. https://doi.org/10.1111/j.1523-1755.2005.00596.x
Block GA, Chertow GM, Sullivan JT, Deng H, Mather O, Tomlin H, Serenko M (2019) An integrated analysis of safety and tolerability of etelcalcetide in patients receiving hemodialysis with secondary hyperparathyroidism. PLoS ONE 14:e0213774. https://doi.org/10.1371/journal.pone.0213774
Shigematsu T, Koiwa F, Isaka Y, Fukagawa M, Hagita K, Watanabe YS, Honda D, Akizawa T (2023) Efficacy and safety of upacicalcet in hemodialysis patients with secondary hyperparathyroidism: a randomized placebo-controlled trial. Clin J Am Soc Nephrol : CJASN 18:1300–1309. https://doi.org/10.2215/CJN.0000000000000253
Itano Y, Kato S, Tsuboi M, Kasuga H, Tsuruta Y, Sato F, Hishida M, Ishimoto T, Kosugi T, Ando M, Kuwatsuka Y, Maruyama S (2020) A prospective, randomized clinical trial of etelcalcetide in patients receiving hemodialysis with secondary hyperparathyroidism (the DUET trial). Kidney Int Rep 5:2168–2177. https://doi.org/10.1016/j.ekir.2020.09.010
El-Shafey EM, Alsahow AE, Alsaran K, Sabry AA, Atia M (2011) Cinacalcet hydrochloride therapy for secondary hyperparathyroidism in hemodialysis patients. Ther Apher Dial: Off Peer-Rev J Int Soc Apher, Jpn Soc Apher, Jpn Soc Dial Ther 15:547–555. https://doi.org/10.1111/j.1744-9987.2011.00994.x
Yeung WG, Palmer SC, Strippoli GFM, Talbot B, Shah N, Hawley CM, Toussaint ND, Badve SV (2023) Vitamin D therapy in adults with ckd: a systematic review and meta-analysis. Am J Kidney Dis. https://doi.org/10.1053/j.ajkd.2023.04.003
Komaba H, Zhao J, Karaboyas A, Yamamoto S, Dasgupta I, Hassan M, Zuo L, Christensson A, Combe C, Robinson BM, Fukagawa M (2023) Active vitamin D use and fractures in hemodialysis patients: results from the international DOPPS. J Bone Miner Res: Off J Am Soc Bone Miner Res 38:1577–1585. https://doi.org/10.1002/jbmr.4913
Rudser KD, de Boer IH, Dooley A, Young B, Kestenbaum B (2007) Fracture risk after parathyroidectomy among chronic hemodialysis patients. J Am Soc Nephrol 18:2401–2407. https://doi.org/10.1681/ASN.2007010022
Ishani A, Liu J, Wetmore JB, Lowe KA, Do T, Bradbury BD, Block GA, Collins AJ (2015) Clinical outcomes after parathyroidectomy in a nationwide cohort of patients on hemodialysis. Clin J Am Soc Nephrol : CJASN 10:90–97. https://doi.org/10.2215/CJN.03520414
Behets GJ, Spasovski G, Sterling LR, Goodman WG, Spiegel DM, De Broe ME, D’Haese PC (2015) Bone histomorphometry before and after long-term treatment with cinacalcet in dialysis patients with secondary hyperparathyroidism. Kidney Int 87:846–856. https://doi.org/10.1038/ki.2014.349
Khairallah P, Cherasard J, Sung J, Agarwal S, Aponte MA, Bucovsky M, Fusaro M, Silberzweig J, Frumkin GN, El Hachem K, Schulman L, McMahon D, Allen MR, Metzger CE, Surowiec RK, Wallace J, Nickolas TL (2023) Changes in bone quality after treatment with etelcalcetide. Clin J Am Soc Nephrol : CJASN. https://doi.org/10.2215/CJN.0000000000000254
Li X, Yu L, Asuncion F, Grisanti M, Alexander S, Hensley K, Han CY, Niu QT, Dwyer D, Villasenor K, Stolina M, Dean C Jr, Ominsky MS, Ke HZ, Tomlinson JE, Richards WG (2017) Etelcalcetide (AMG 416), a peptide agonist of the calcium-sensing receptor, preserved cortical bone structure and bone strength in subtotal nephrectomized rats with established secondary hyperparathyroidism. Bone 105:163–172. https://doi.org/10.1016/j.bone.2017.08.026
Al-Dujaili SA, Koh AJ, Dang M, Mi X, Chang W, Ma PX, McCauley LK (2016) Calcium sensing receptor function supports osteoblast survival and acts as a co-factor in pth anabolic actions in bone. J Cell Biochem 117:1556–1567. https://doi.org/10.1002/jcb.25447
Disease K (2011) Improving global outcomes ckdmbduwg (2017) kdigo 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int Suppl 7:1–59. https://doi.org/10.1016/j.kisu.2017.04.001
Fukagawa M, Yokoyama K, Koiwa F, Taniguchi M, Shoji T et al (2013) Clinical practice guideline for the management of chronic kidney disease-mineral and bone disorder (in eng). Ther Apher And Dial: Off Peer-Rev J Int Soc Apher, Jpn Soc Apher, Jpn Soc Dial Ther 17:247–288. https://doi.org/10.1111/1744-9987.12058
Harbord RM, Egger M, Sterne JA (2006) A modified test for small-study effects in meta-analyses of controlled trials with binary endpoints. Stat Med 25:3443–3457. https://doi.org/10.1002/sim.2380
Author information
Authors and Affiliations
Contributions
Conception, study design, and interpretation of the data were performed by T.W., S.Y., K.M., M.T., T.H., M.F. and J.K. Systematic reviews were performed by T.W., S.Y., and K.M. Meta-analyses were performed by T.W. and S.Y. The manuscript was drafted by T. W. and S. Y. Approval of the final version of the manuscript was provided by T. W., S. Y., K. M., M. T., T. H., M.F., and J. K.
Corresponding author
Ethics declarations
Conflict of interest
S.Y. has received honoraria from Kyowa Kirin, and research findings from Toray Medical Co., Ltd and Kaneka Medix Co., Ltd. M.T. received honoraria from Kyowa Kirin, Bayer Yakuhin Ltd., Mitsubishi Tanabe Pharma Corporation, Kissey Pharmaceutical Co., Ltd., Ono Pharmaceutical Co., Ltd., Torii Pharmaceutical Co., Ltd.,and Daiichi Sankyo Company Ltd. T.H. has received honoraria, consulting fees, and grant support from Kissei Pharmaceutical, Kyowa Hakko Kirin, Ono Pharmaceutical, and Sanwa Kagaku Kenkyusho. M.F. has received honoraria, consulting fees, and/or grant support from Kyowa Kirin, Ono Pharmaceutical, Sanwa Kagaku Kenkyusho, and Kissei Pharmaceutical. The other authors declare no conflicts of interest. J.K. has received honoraria from Kyowa Kirin.
Ethical approval
This article is a meta-analysis and does not contain any studies with human participants performed by the authors.
Informed consent
No informed consent is available.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Wakamatsu, T., Yamamoto, S., Matsuo, K. et al. Effectiveness of calcimimetics on fractures in dialysis patients with secondary hyperparathyroidism: meta-analysis of randomized trials. J Bone Miner Metab (2024). https://doi.org/10.1007/s00774-024-01500-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00774-024-01500-y