Abstract
Introduction
Selenium (Se) as well as selenoproteins are vital for osteochondral system development. Se deficiency (SeD) has a definite impact on the expression and activity of histone deacetylases (HDACs). Abnormal expression of some HDACs affects cartilage development. This current study aims to explore the relationship between differentially expressed HDACs and cartilage development, especially extracellular matrix (ECM) homeostasis maintenance, under SeD conditions.
Materials and methods
Dark Agouti rats and C28/I2 cell line under SeD states were used to detect the differently expressed HDAC by RT-qPCR, western blotting and IHC staining. Meanwhile, the biological roles of the above HDAC in cartilage development and homeostasis maintenance were confirmed by siRNA transfection, western blotting, RNA sequence and inhibitor treatment experiments.
Results
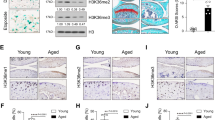
HDAC2 exhibited lower expression at protein level in both animals and chondrocytes during SeD condition. The results of cell-level experiments indicated that forkhead box O3A (FOXO3A), which was required to maintain metabolic homeostasis of cartilage matrix, was reduced by HDAC2 knockdown. Meanwhile, induced HDAC2 was positively associated with FOXO3A in rat SeD model. Meanwhile, knockdown of HDAC2 and FOXO3A led to an increase of intracellular ROS level, which activated NF-κB pathway. Se supplementary significantly inhibited the activation of NF-κB pathway with IL-1β treatment.
Conclusion
Our results suggested that low expression of HDAC2 under SeD condition increased ROS content by decreasing FOXO3A in chondrocytes, which led to the activation of NF-κB pathway and ECM homeostasis imbalance.






Similar content being viewed by others
Change history
02 January 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00774-022-01395-7
References
Labunskyy VM, Hatfield DL, Gladyshev VN (2014) Selenoproteins: molecular pathways and physiological roles. Physiol Rev 94:739–777
Guan F et al (2013) Histopathology of chondronecrosis development in knee articular cartilage in a rat model of Kashin-Beck disease using T-2 toxin and selenium deficiency conditions. Rheumatol Int 33:157–166
Mathieu F et al (1997) Clinical manifestations of Kashin-Beck disease in Nyemo valley. Tibet Int Orthop 21:151–156
Joseph J, Loscalzo J (2013) Selenistasis: epistatic effects of selenium on cardiovascular phenotype. Nutrients 5:340–358
Metes-Kosik N et al (2012) Both selenium deficiency and modest selenium supplementation lead to myocardial fibrosis in mice via effects on redox-methylation balance. Mol Nutr Food Res 56:1812–1824
Yang G et al (2014) TLR2-ICAM1-Gadd45alpha axis mediates the epigenetic effect of selenium on DNA methylation and gene expression in Keshan disease. Biol Trace Elem Res 159:69–80
Narayan V et al (2015) Epigenetic regulation of inflammatory gene expression in macrophages by selenium. J Nutr Biochem 26:138–145
Hu CF et al (2015) Upregulation of KLF4 by Methylseleninic acid in human esophageal squamous cell carcinoma cells: modification of histone h3 acetylation through hat/hdac interplay. Mol Carcinog 54:1051–1059
Gowda R et al (2012) Selenium-containing histone deacetylase inhibitors for melanoma management. Cancer Biol Ther 13:756–765
Kassam S et al (2011) Methylseleninic acid inhibits HDAC activity in diffuse large B-cell lymphoma cell lines. Cancer Chemother Pharmacol 68:815–821
Huang X et al (2021) Dietary selenium regulates microRNAs in metabolic disease: recent progress. Nutrients 13:1527
Li Y et al (2014) AF9 YEATS domain links histone acetylation to DOT1L-mediated H3K79 methylation. Cell 159:558–571
Carpio LR, Westendorf JJ (2016) Histone deacetylases in cartilage homeostasis and osteoarthritis. Curr Rheumatol Rep 18:52
Hong S et al (2009) A novel domain in histone deacetylase 1 and 2 mediates repression of cartilage-specific genes in human chondrocytes. FASEB J 23:3539–3552
Vega RB et al (2004) Histone deacetylase 4 controls chondrocyte hypertrophy during skeletogenesis. Cell 119:555–566
Lu J et al (2014) Histone deacetylase 4 alters cartilage homeostasis in human osteoarthritis. BMC Musculoskelet Disord 15:438
Shimizu E et al (2010) HDAC4 represses matrix metalloproteinase-13 transcription in osteoblastic cells, and parathyroid hormone controls this repression. J Biol Chem 285:9616–9626
Shimizu E et al (2014) Parathyroid hormone regulates histone deacetylase (HDAC) 4 through protein kinase a-mediated phosphorylation and dephosphorylation in osteoblastic cells. J Biol Chem 289:21340–21350
Higashiyama R et al (2010) Correlation between MMP-13 and HDAC7 expression in human knee osteoarthritis. Mod Rheumatol 20:11–17
Young DA et al (2005) Differential effects of histone deacetylase inhibitors on phorbol ester- and TGF-beta1 induced murine tissue inhibitor of metalloproteinases-1 gene expression. FEBS J 272:1912–1926
Young DA et al (2005) Histone deacetylase inhibitors modulate metalloproteinase gene expression in chondrocytes and block cartilage resorption. Arthritis Res Ther 7:R503–R512
Nishida K et al (2004) Histone deacetylase inhibitor suppression of autoantibody-mediated arthritis in mice via regulation of p16INK4a and p21(WAF1/Cip1) expression. Arthritis Rheum 50:3365–3376
Nasu Y et al (2008) Trichostatin A, a histone deacetylase inhibitor, suppresses synovial inflammation and subsequent cartilage destruction in a collagen antibody-induced arthritis mouse model. Osteoarthritis Cartilage 16:723–732
Bradley EW et al (2015) Histone deacetylases in bone development and skeletal disorders. Physiol Rev 95:1359–1381
Hazane-Puch F et al (2016) Sodium selenite decreased hdac activity, cell proliferation and induced apoptosis in three human glioblastoma cells. Anticancer Agents Med Chem 16:490–500
Min Z et al (2015) Abnormality of epiphyseal plate induced by selenium deficiency diet in two generation DA rats. APMIS 123:697–705
Yan J et al (2012) Selenium promotes proliferation of chondrogenic cell ATDC5 by increment of intracellular ATP content under serum deprivation. Cell Biochem Funct 30:657–663
Yan J et al (2013) Selenium effect on selenoprotein transcriptome in chondrocytes. Biometals 26:285–296
Salhotra A et al (2020) Mechanisms of bone development and repair. Nat Rev Mol Cell Biol 21:696–711
Huang X et al (2019) TMF inhibits miR-29a/Wnt/beta-catenin signaling through upregulating Foxo3a activity in osteoarthritis chondrocytes. Drug Des Devel Ther 13:2009–2019
Xu H et al (2021) NF-kappaB inducible miR-30b-5p aggravates joint pain and loss of articular cartilage via targeting SIRT1-FoxO3a-mediated NLRP3 inflammasome. Aging (Albany NY) 13:20774–20792
Zhao X et al (2014) Peroxisome proliferator-activated receptor gamma coactivator 1alpha and FoxO3A mediate chondroprotection by AMP-activated protein kinase. Arthritis Rheumatol 66:3073–3082
Peng S et al (2015) HDAC2 selectively regulates FOXO3a-mediated gene transcription during oxidative stress-induced neuronal cell death. J Neurosci 35:1250–1259
Alehagen U et al (2021) Impact of selenium on biomarkers and clinical aspects related to ageing A Review. Biomolecules 11:1478
Handy DE, Joseph J, Loscalzo J (2021) Selenium, a micronutrient that modulates cardiovascular health via redox enzymology. Nutrients 13:3238
Henning T, Weber D (2021) Redox biomarkers in dietary interventions and nutritional observation studies- From new insights to old problems. Redox Biol. https://doi.org/10.1016/j.redox.2021.101922
Razaghi A et al (2021) Selenium stimulates the antitumour immunity: Insights to future research. Eur J Cancer 155:256–267
Henrotin Y, Kurz B, Aigner T (2005) Oxygen and reactive oxygen species in cartilage degradation: friends or foes? Osteoarthritis Cartilage 13:643–654
Li D, Xie G, Wang W (2012) Reactive oxygen species: the 2-edged sword of osteoarthritis. Am J Med Sci 344:486–490
Gloire G, Legrand-Poels S, Piette J (2006) NF-kappaB activation by reactive oxygen species: fifteen years later. Biochem Pharmacol 72:1493–1505
Fairweather-Tait SJ et al (2011) Selenium in human health and disease. Antioxid Redox Signal 14:1337–1383
Stone R (2009) Diseases a medical mystery in middle China. Science 324:1378–1381
Zhou TY et al (2019) SIRT3 retards intervertebral disc degeneration by anti-oxidative stress by activating the SIRT3/FOXO3/SOD2 signaling pathway. Eur Rev Med Pharmacol Sci 23:9180–9188
Han MS et al (2020) Transglutaminase-2 regulates Wnt and FoxO3a signaling to determine the severity of osteoarthritis. Sci Rep 10:13228
Lin L, Hron JD, Peng SL (2004) Regulation of NF-kappaB, Th activation, and autoinflammation by the forkhead transcription factor Foxo3a. Immunity 21:203–213
Acknowledgements
The inbred Dark Agouti (DA) rats were provided by Prof. Rikard Holmdahl, Karolinska Institute, Sweden. The C28/I2 cell line is provided by Professor Junling Cao, from Institute of endemic diseases, Xi’an Jiaotong University Health Science Center.
Funding
This work was supported by National Natural Science Foundation of China (Project No. 82072432, 81970029).
Author information
Authors and Affiliations
Contributions
SL and JS conceived, designed and supervised the study. YZ performed most of the experiments, analyzed and interpreted the data and wrote the original manuscript. YG, MS, YZ, SH, HH, XH, YZ, FZ, YH, QN. and PX. helped the experiments. JS. and SL obtained the funding and critically revised the article for important intellectual content, and take responsibility for the integrity of the work as a whole.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The experiments were approved by Institutional Animal Ethics Committee of the University (Permission ID: XJ2006Y039; Xi'an Jiaotong University, Xi'an, China) and were in compliance with ARRIVE guidelines and European Community specifications regarding the use of laboratory animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised due to the funding note was missing and included in this version.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Zhao, Y., Guo, Y., Sun, M. et al. Selenium-sensitive histone deacetylase 2 is required for forkhead box O3A and regulates extracellular matrix metabolism in cartilage. J Bone Miner Metab 40, 914–926 (2022). https://doi.org/10.1007/s00774-022-01369-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-022-01369-9